Abstract
Objectives
This in vivo animal study aimed to develop a murine model of pulpitis induced by pulp exposure with or without application of zymosan in Naval Medical Research Institute (NMRI) mice and observe expressions of Toll-like receptor (TLR)-2, TLR-4, Dectin-1, Osteopontin (OPN), tumor necrosis factor alpha (TNF-α), interleukin (IL)-6, and IL-1ß.
Material and methods
A total of 168 NMRI mice were divided into two groups, i.e., group A (n = 84) (pulpitis induced by pulp exposure only) and group B (n = 84) (pulpitis induced by pulp exposure and zymosan application). Right maxillary molar pulps were exposed with ¼ round bur, and animals were sacrificed at 0, 6, 9, 12, 24, 48, and 72 h. The exposed teeth were obtained for real-time polymerase chain reaction (qRT-PCR) analysis and histological and immunohistochemistry (IHC) analysis.
Results
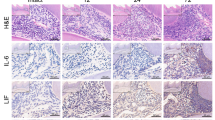
Histological evaluation revealed a time-dependent steady increase in inflammation. Similar time-dependent increase in the expression of inflammatory cytokines was noted. Group A exhibited an increase in TLR-4, Dectin-1, and OPN at 6 h, while TLR-2 was expressed at 24 h. Group B expressed TLR-2, Dectin-1, and OPN at 9, 48, and 72 h, respectively (p ≤ 0.05). Expression of OPN and TNF-α exhibited a similar pattern in both groups. IHC also detected expression of TLR-2, Dectin-1, TLR4, and CD68 in some cells at 6 and 9 h.
Conclusions
NMRI mice provided for a stable pulp inflammation model. Zymosan may be used to develop pulp inflammation model and study inflammatory response towards fungal antigens. Dental pulp expressed Dectin-1 receptor. OPN and TNF-α exhibited a similar expression pattern.
Clinical relevance
Innate immunity of dental pulp is capable of detecting fungal pathogens.





Similar content being viewed by others
References
Farges JC, Alliot-Licht B, Renard E, Ducret M, Gaudin A, Smith AJ, Cooper PR (2015) Dental pulp defence and repair mechanisms in dental caries. Mediators Inflamm 2015:230251. https://doi.org/10.1155/2015/230251
Mutoh N, Tani-Ishii N, Tsukinoki K, Chieda K, Watanabe K (2007) Expression of toll-like receptor 2 and 4 in dental pulp. J Endod 33:1183–1186. https://doi.org/10.1016/j.joen.2007.05.018
Mutoh N, Watabe H, Chieda K, Tani-Ishii N (2009) Expression of Toll-like receptor 2 and 4 in inflamed pulp in severe combined immunodeficiency mice. J Endod 35:975–980. https://doi.org/10.1016/j.joen.2009.04.005
Keller JF, Carrouel F, Colomb E, Durand SH, Baudouin C, Msika P, Bleicher F, Vincent C, Staquet MJ, Farges JC (2010) Toll-like receptor 2 activation by lipoteichoic acid induces differential production of pro-inflammatory cytokines in human odontoblasts, dental pulp fibroblasts and immature dendritic cells. Immunobiol 215:53–59. https://doi.org/10.1016/j.imbio.2009.01.009
Kikuchi T, Matsuguchi T, Tsuboi N, Mitani A, Tanaka S, Matsuoka M, Yamamoto G, Hishikawa T, Noguchi T, Yoshikai Y (2001) Gene expression of osteoclast differentiation factor is induced by lipopolysaccharide in mouse osteoblasts via Toll-like receptors. J Immunol 166:3574–3579. https://doi.org/10.4049/jimmunol.166.5.3574
Marcato LG, Ferlini AP, Bonfim RC, Ramos-Jorge ML, Ropert C, Afonso LF, Vieira LQ, Sobrinho AP (2008) The role of Toll-like receptors 2 and 4 on reactive oxygen species and nitric oxide production by macrophage cells stimulated with root canal pathogens. Oral Microbiol Immunol 23:353–359. https://doi.org/10.1111/j.1399-302X.2008.00432.x
Carrouel F, Staquet MJ, Keller JF, Baudouin C, Msika P, Bleicher F, Alliot-Licht B, Farges JC (2013) Lipopolysaccharide-binding protein inhibits toll-like receptor 2 activation by lipoteichoic acid in human odontoblast-like cells. J Endod 39:1008–1014. https://doi.org/10.1016/j.joen.2013.04.020
da Silva RA, Ferreira PD, De Rossi A, Nelson-Filho P, Silva LA (2012) Toll-like receptor 2 knockout mice showed increased periapical lesion size and osteoclast number. J Endod 38:803–813. https://doi.org/10.1016/j.joen.2012.03.017
Desai SV, Love RM, Rich AM, Seymour GJ (2011) Toll-like receptor 2 expression in refractory periapical lesions. Int Endod J 44:907–916. https://doi.org/10.1111/j.1365-2591.2011.01893.x
Egan MW, Spratt DA, Ng YL, Lam JM, Moles DR, Gulabivala K (2002) Prevalence of yeasts in saliva and root canals of teeth associated with apical periodontitis. Int Endod J 35:321–329. https://doi.org/10.1046/j.1365-2591.2002.00478.x
Miranda TT, Vianna CR, Rodrigues L, Monteiro AS, Rosa CA, Correa A Jr (2009) Diversity and frequency of yeasts from the dorsum of the tongue and necrotic root canals associated with primary apical periodontitis. Int Endod J 42:839–844. https://doi.org/10.1111/j.1365-2591.2009.01601.x
Peciuliene V, Reynaud AH, Balciuniene I, Haapasalo M (2001) Isolation of yeasts and enteric bacteria in root-filled teeth with chronic apical periodontitis. Int Endod J 34:429–434. https://doi.org/10.1046/j.1365-2591.2001.00411.x
Waltimo TM, Sen BH, Meurman JH, Orstavik D, Haapasalo MP (2003) Yeasts in apical periodontitis. Crit Rev Oral Biol Med 14:128–137. https://doi.org/10.1177/154411130301400206
Waltimo TMT, Haapasalo M, Zehnder M, Meyer J (2004) Clinical aspects related to endodontic yeast infections. Endod Top 9:66–78. https://doi.org/10.1111/j.1601-1546.2004.00086.x
Diaz PI, **e Z, Sobue T, Thompson A, Biyikoglu B, Ricker A, Ikonomou L, Dongari-Bagtzoglou A (2012) Synergistic interaction between Candida albicans and commensal oral streptococci in a novel in vitro mucosal model. Infect Immun 80:620–632. https://doi.org/10.1128/IAI.05896-11
Falsetta ML, Klein MI, Colonne PM, Scott-Anne K, Gregoire S, Pai CH, Gonzalez-Begne M, Watson G, Krysan DJ, Bowen WH, Koo H (2014) Symbiotic relationship between Streptococcus mutans and Candida albicans synergizes virulence of plaque biofilms in vivo. Infect Immun 82:1968–1981. https://doi.org/10.1128/IAI.00087-14
Koo H, Bowen WH (2014) Candida albicans and Streptococcus mutans: a potential synergistic alliance to cause virulent tooth decay in children. Future Microbiol 9:1295–1297. https://doi.org/10.2217/fmb.14.92
Hahn CL, Liewehr FR (2007) Relationships between caries bacteria, host responses, and clinical signs and symptoms of pulpitis. J Endod 33:213–219. https://doi.org/10.1016/j.joen.2006.11.008
Patidar A, Mahanty T, Raybarman C, Sarode AY, Basak S, Saha B, Bhattacharjee S (2020) Barley beta-Glucan and Zymosan induce Dectin-1 and Toll-like receptor 2 co-localization and anti-leishmanial immune response in Leishmania donovani-infected BALB/c mice. Scand J Immunol 92:e12952. https://doi.org/10.1111/sji.12952
Hsu PH, Chiang PC, Liu CH, Chang YW (2015) Characterization of cell wall proteins in Saccharomyces cerevisiae clinical isolates elucidates Hsp150p in virulence. PLoS ONE 10:e0135174. https://doi.org/10.1371/journal.pone.0135174
Klis FM, de Groot P, Hellingwerf K (2001) Molecular organization of the cell wall of Candida albicans. Med Mycol 39(Suppl 1):1–8
Gantner BN, Simmons RM, Canavera SJ, Akira S, Underhill DM (2003) Collaborative induction of inflammatory responses by dectin-1 and Toll-like receptor 2. J Exp Med 197:1107–1117. https://doi.org/10.1084/jem.20021787
Monroe LL, Armstrong MG, Zhang X, Hall JV, Ozment TR, Li C, Williams DL, Hoover DB (2016) Zymosan-induced peritonitis: effects on cardiac function, temperature regulation, translocation of bacteria, and role of dectin-1. Shock 46:723–730. https://doi.org/10.1097/SHK.0000000000000669
Olynych TJ, Jakeman DL, Marshall JS (2006) Fungal zymosan induces leukotriene production by human mast cells through a dectin-1-dependent mechanism. J Allergy Clin Immunol 118:837–843. https://doi.org/10.1016/j.jaci.2006.06.008
Rosenzweig HL, Clowers JS, Nunez G, Rosenbaum JT, Davey MP (2011) Dectin-1 and NOD2 mediate cathepsin activation in zymosan-induced arthritis in mice. Inflamm Res 60:705–714. https://doi.org/10.1007/s00011-011-0324-7
Cho HJ, Cho HJ, Kim HS (2009) Osteopontin: a multifunctional protein at the crossroads of inflammation, atherosclerosis, and vascular calcification. Curr Atheroscler Rep 11:206–213. https://doi.org/10.1007/s11883-009-0032-8
Shinohara ML, Kim HJ, Kim JH, Garcia VA, Cantor H (2008) Alternative translation of osteopontin generates intracellular and secreted isoforms that mediate distinct biological activities in dendritic cells. Proc Natl Acad Sci U S A 105:7235–7239. https://doi.org/10.1073/pnas.0802301105
Wang KX, Denhardt DT (2008) Osteopontin: role in immune regulation and stress responses. Cytokine Growth Factor Rev 19:333–345. https://doi.org/10.1016/j.cytogfr.2008.08.001
Ashkar S, Weber GF, Panoutsakopoulou V, Sanchirico ME, Jansson M, Zawaideh S, Rittling SR, Denhardt DT, Glimcher MJ, Cantor H (2000) Eta-1 (osteopontin): an early component of type-1 (cell-mediated) immunity. Science 287:860–864. https://doi.org/10.1126/science.287.5454.860
Nau GJ, Liaw L, Chupp GL, Berman JS, Hogan BL, Young RA (1999) Attenuated host resistance against Mycobacterium bovis BCG infection in mice lacking osteopontin. Infect Immun 67:4223–4230. https://doi.org/10.1128/IAI.67.8.4223-4230.1999
Dong M, Yu X, Chen W, Guo Z, Sui L, Xu Y, Shang Y, Niu W, Kong Y (2018) Osteopontin promotes bone destruction in periapical periodontitis by activating the NF-κB pathway. Cell Physiol Biochem 49:884–898. https://doi.org/10.1159/000493219
Rittling SR, Zetterberg C, Yagiz K, Skinner S, Suzuki N, Fujimura A, Sasaki H (2010) Protective role of osteopontin in endodontic infection. Immunology 129:105–114. https://doi.org/10.1111/j.1365-2567.2009.03159.x
Haddad M, Lefranc G, Aftimos G (2003) Local application of IGF1 on dental pulp mechanically exposed; in vivo study on rabbit. Bull Group Int Rech Sci Stomatol Odontol 45:12–17
Huang J, Lv Y, Fu Y, Ren L, Wang P, Liu B, Huang K, Bi J (2015) Dynamic regulation of delta-opioid receptor in rat trigeminal ganglion neurons by lipopolysaccharide-induced acute pulpitis. J Endod 41:2014–2020. https://doi.org/10.1016/j.joen.2015.09.011
Shahravan A, Ghoddusi J, Eslami B, Rategar AF (2010) A histopathological study of the pulp of dogs’ teeth after induction of experimental pulp inflammation for different periods of time. J Microsc 237:119–121. https://doi.org/10.1111/j.1365-2818.2009.03312.x
Balto K, White R, Mueller R, Stashenko P (2002) A mouse model of inflammatory root resorption induced by pulpal infection. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 93:461–468
De Rossi A, Rocha LB, Rossi MA (2008) Interferon-gamma, interleukin-10, Intercellular adhesion molecule-1, and chemokine receptor 5, but not interleukin-4, attenuate the development of periapical lesions. J Endod 34:31–38. https://doi.org/10.1016/j.joen.2007.09.021
Song M, Kim S, Kim T, Park S, Shin KH, Kang M, Park NH, Kim R (2017) Development of a direct pulpcap** model for the evaluation of pulpal wound healing and reparative dentin formation in mice. J Vis Exp 119:54973. https://doi.org/10.3791/54973
Sasaki H, Furusho H, Rider DB, Dobeck JM, Kuo WP, Fujimura A, Yoganathan S, Hirai K, Xu S, Sasaki K, Stashenko P (2019) Endodontic infection-induced inflammation resembling osteomyelitis of the jaws in Toll-like receptor 2/interleukin 10 double-knockout mice. J Endod 45:181–188. https://doi.org/10.1016/j.joen.2018.10.007
He Y, Gan Y, Lu J, Feng Q, Wang H, Guan H, Jiang Q (2017) Pulpal tissue inflammatory reactions after experimental pulpal exposure in mice. J Endod 43:90–95. https://doi.org/10.1016/j.joen.2016.09.003
Percie du Sert N, Ahluwalia A, Alam S, Avey MT, Baker M, Browne WJ, Clark A, Cuthill IC, Dirnagl U, Emerson M, Garner P, Holgate ST, Howells DW, Hurst V, Karp NA, Lazic SE, Lidster K, MacCallum CJ, Macleod M, Pearl EJ, Petersen OH, Rawle F, Reynolds P, Rooney K, Sena ES, Silberberg SD, Steckler T, Wurbel H (2020) Reporting animal research: explanation and elaboration for the ARRIVE guidelines 2.0. PLoS Biol 18:e3000411. https://doi.org/10.1371/journal.pbio.3000411
Erdo F, Torok K, Aranyi P, Szekely JI (1993) A new assay for antiphlogistic activity: zymosan-induced mouse ear inflammation. Agents Actions 39:137–142. https://doi.org/10.1007/BF01998966
Rider D, Furusho H, Xu S, Trachtenberg AJ, Kuo WP, Hirai K, Susa M, Bahammam L, Stashenko P, Fujimura A, Sasaki H (2016) Elevated CD14 (cluster of differentiation 14) and Toll-like receptor (TLR) 4 signaling deteriorate periapical inflammation in TLR2 deficient mice. Anat Rec (Hoboken) 299:1281–1292. https://doi.org/10.1002/ar.23383
Hebling J, Giro EM, Costa CA (1999) Biocompatibility of an adhesive system applied to exposed human dental pulp. J Endod 25:676–682. https://doi.org/10.1016/s0099-2399(99)80354-9
Leonardo MR, Barnett F, Debelian GJ, de Pontes Lima RK, Bezerra da Silva LA (2007) Root canal adhesive filling in dogs’ teeth with or without coronal restoration: a histopathological evaluation. J Endod 33:1299–1303. https://doi.org/10.1016/j.joen.2007.07.037
Myles MH, Livingston RS, Livingston BA, Criley JM, Franklin CL (2003) Analysis of gene expression in ceca of Helicobacter hepaticus-infected A/JCr mice before and after development of typhlitis. Infect Immun 71:3885–3893. https://doi.org/10.1128/IAI.71.7.3885-3893.2003
Ye J, Coulouris G, Zaretskaya I, Cutcutache I, Rozen S, Madden TL (2012) Primer-BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC Bioinformatics 13:134. https://doi.org/10.1186/1471-2105-13-134
Byers MR, Närhi MV (1999) Dental injury models: experimental tools for understanding neuroinflammatory interactions and polymodal nociceptor functions. Crit Rev Oral Biol Med 10:4–39. https://doi.org/10.1177/10454411990100010101
Bernardino ÍdM, Farias ÍdL, Cardoso A, Xavier A, Cavalcanti A (2014) Use of animal models in experimental research in dentistry in Brazil. Pesqui Bras Odontopediatria Clin Integr 14:17–21
Alzarea B (2014) Selection of animal models in dentistry: state of art, review article. J Anim Vet Adv 13:1080–1085
Renard E, Gaudin A, Bienvenu G, Amiaud J, Farges JC, Cuturi MC, Moreau A, Alliot-Licht B (2015) Immune cells and molecular networks in experimentally induced pulpitis. J Dent Res. https://doi.org/10.1177/0022034515612086
Chattipakorn SC, Sigurdsson A, Light AR, Narhi M, Maixner W (2002) Trigeminal c-Fos expression and behavioral responses to pulpal inflammation in ferrets. Pain 99:61–69. https://doi.org/10.1016/s0304-3959(02)00054-4
Kawanishi HN, Kawashima N, Suzuki N, Suda H, Takagi M (2004) Effects of an inducible nitric oxide synthase inhibitor on experimentally induced rat pulpitis. Eur J Oral Sci 112:332–337. https://doi.org/10.1111/j.1600-0722.2004.00139.x
Kawashima N, Nakano-Kawanishi H, Suzuki N, Takagi M, Suda H (2005) Effect of NOS inhibitor on cytokine and COX2 expression in rat pulpitis. J Dent Res 84:762–767. https://doi.org/10.1177/154405910508400815
Li M, Tian J, Xu Z, Zeng Q, Chen W, Lei S, Wei X (2021) Histology-based profile of inflammatory mediators in experimentally induced pulpitis in a rat model: screening for possible biomarkers. Int Endod J 54:1328–1341. https://doi.org/10.1111/iej.13514
Rutherford RB, Gu K (2000) Treatment of inflamed ferret dental pulps with recombinant bone morphogenetic protein-7. Eur J Oral Sci 108:202–206. https://doi.org/10.1034/j.1600-0722.2000.108003202.x
Chung MK, Lee J, Duraes G, Ro JY (2011) Lipopolysaccharide-induced pulpitis up-regulates TRPV1 in trigeminal ganglia. J Dent Res 90:1103–1107. https://doi.org/10.1177/0022034511413284
Cleaton-Jones P, Duggal M, Parak R, Williams S, Setzer S (2004) Pulpitis induction in baboon primary teeth using carious dentine or Streptococcus mutans. Sadj 59:119–122
Eba H, Murasawa Y, Iohara K, Isogai Z, Nakamura H, Nakamura H, Nakashima M (2012) The anti-inflammatory effects of matrix metalloproteinase-3 on irreversible pulpitis of mature erupted teeth. PLoS ONE 7:e52523. https://doi.org/10.1371/journal.pone.0052523
Fuks AB, Jones PC, Michaeli Y, Bimstein E (1991) Pulp response to collagen and glutaraldehyde in pulpotomized primary teeth of baboons. Pediatr Dent 13:142–150
Oliveira LAd, Barbosa SV (2003) The reaction of dental pulp to Escherichia coli lipopolysaccharide and Enterococcus faecalis lipoteichoic acid. Braz J Microbiol 34:179–181
Ruiz-Herrera J, Elorza MV, Valentin E, Sentandreu R (2006) Molecular organization of the cell wall of Candida albicans and its relation to pathogenicity. FEMS Yeast Res 6:14–29. https://doi.org/10.1111/j.1567-1364.2005.00017.x
Abbas AK, Murphy KM, Sher A (1996) Functional diversity of helper T lymphocytes. Nature 383:787–793. https://doi.org/10.1038/383787a0
Romagnani S (1994) Lymphokine production by human T cells in disease states. Annu Rev Immunol 12:227–257. https://doi.org/10.1146/annurev.iy.12.040194.001303
Bradley JR (2008) TNF-mediated inflammatory disease. J Pathol 214:149–160. https://doi.org/10.1002/path.2287
Akdis M, Burgler S, Crameri R, Eiwegger T, Fujita H, Gomez E, Klunker S, Meyer N, O'Mahony L, Palomares O, Rhyner C, Ouaked N, Schaffartzik A, Van De Veen W, Zeller S, Zimmermann M and Akdis CA (2011) Interleukins, from 1 to 37, and interferon-γ: receptors, functions, and roles in diseases. J Allergy Clin Immunol 127:701–21.e1–70. https://doi.org/10.1016/j.jaci.2010.11.050
Nagabhushanam V, Solache A, Ting LM, Escaron CJ, Zhang JY, Ernst JD (2003) Innate inhibition of adaptive immunity: Mycobacterium tuberculosis-induced IL-6 inhibits macrophage responses to IFN-gamma. J Immunol 171:4750–4757. https://doi.org/10.4049/jimmunol.171.9.4750
Li JG, Lin JJ, Wang ZL, Cai WK, Wang PN, Jia Q, Zhang AS, Wu GY, Zhu GX, Ni LX (2015) Melatonin attenuates inflammation of acute pulpitis subjected to dental pulp injury. Am J Transl Res 7:66–78
Lin JJ, Du Y, Cai WK, Kuang R, Chang T, Zhang Z, Yang YX, Sun C, Li ZY, Kuang F (2015) Toll-like receptor 4 signaling in neurons of trigeminal ganglion contributes to nociception induced by acute pulpitis in rats. Sci Rep 5:12549. https://doi.org/10.1038/srep12549
Renard E, Gaudin A, Bienvenu G, Amiaud J, Farges JC, Cuturi MC, Moreau A, Alliot-Licht B (2016) Immune cells and molecular networks in experimentally induced pulpitis. J Dent Res 95:196–205. https://doi.org/10.1177/0022034515612086
Márton IJ, Kiss C (2014) Overlap** protective and destructive regulatory pathways in apical periodontitis. J Endod 40:155–163. https://doi.org/10.1016/j.joen.2013.10.036
Staquet MJ, Carrouel F, Keller JF, Baudouin C, Msika P, Bleicher F, Kufer TA, Farges JC (2011) Pattern-recognition receptors in pulp defense. Adv Dent Res 23:296–301. https://doi.org/10.1177/0022034511405390
Brown GD (2006) Dectin-1: a signalling non-TLR pattern-recognition receptor. Nat Rev Immunol 6:33–43. https://doi.org/10.1038/nri1745
Ifrim DC, Joosten LA, Kullberg BJ, Jacobs L, Jansen T, Williams DL, Gow NA, van der Meer JW, Netea MG, Quintin J (2013) Candida albicans primes TLR cytokine responses through a Dectin-1/Raf-1-mediated pathway. J Immunol 190:4129–4135. https://doi.org/10.4049/jimmunol.1202611
Liu J, Geng F, Sun H, Wang X, Zhang H, Yang Q, Zhang J (2018) Candida albicans induces TLR2/MyD88/NF-κB signaling and inflammation in oral lichen planus-derived keratinocytes. J Infect Dev Ctries 12:780–786. https://doi.org/10.3855/jidc.8062
Viriyakosol S, Fierer J, Brown GD, Kirkland TN (2005) Innate immunity to the pathogenic fungus Coccidioides posadasii is dependent on Toll-like receptor 2 and Dectin-1. Infect Immun 73:1553–1560. https://doi.org/10.1128/iai.73.3.1553-1560.2005
Rogers NC, Slack EC, Edwards AD, Nolte MA, Schulz O, Schweighoffer E, Williams DL, Gordon S, Tybulewicz VL, Brown GD, Reis e Sousa C, (2005) Syk-dependent cytokine induction by Dectin-1 reveals a novel pattern recognition pathway for C type lectins. Immunity 22:507–517. https://doi.org/10.1016/j.immuni.2005.03.004
Steele C, Rapaka RR, Metz A, Pop SM, Williams DL, Gordon S, Kolls JK, Brown GD (2005) The beta-glucan receptor dectin-1 recognizes specific morphologies of Aspergillus fumigatus. PLoS Pathog 1:e42. https://doi.org/10.1371/journal.ppat.0010042
Jouault T, Ibata-Ombetta S, Takeuchi O, Trinel PA, Sacchetti P, Lefebvre P, Akira S, Poulain D (2003) Candida albicans phospholipomannan is sensed through toll-like receptors. J Infect Dis 188:165–172. https://doi.org/10.1086/375784
Li M, Chen Q, Shen Y, Liu W (2009) Candida albicans phospholipomannan triggers inflammatory responses of human keratinocytes through Toll-like receptor 2. Exp Dermatol 18:603–610. https://doi.org/10.1111/j.1600-0625.2008.00832.x
Netea MG, Gow NA, Munro CA, Bates S, Collins C, Ferwerda G, Hobson RP, Bertram G, Hughes HB, Jansen T, Jacobs L, Buurman ET, Gijzen K, Williams DL, Torensma R, McKinnon A, MacCallum DM, Odds FC, Van der Meer JW, Brown AJ, Kullberg BJ (2006) Immune sensing of Candida albicans requires cooperative recognition of mannans and glucans by lectin and Toll-like receptors. J Clin Invest 116:1642–1650. https://doi.org/10.1172/jci27114
Brown GD, Herre J, Williams DL, Willment JA, Marshall AS, Gordon S (2003) Dectin-1 mediates the biological effects of beta-glucans. J Exp Med 197:1119–1124. https://doi.org/10.1084/jem.20021890
Inoue M, Shinohara ML (2011) Intracellular osteopontin (iOPN) and immunity. Immunol Res 49:160–172. https://doi.org/10.1007/s12026-010-8179-5
Zohar R, Suzuki N, Suzuki K, Arora P, Glogauer M, McCulloch CA, Sodek J (2000) Intracellular osteopontin is an integral component of the CD44-ERM complex involved in cell migration. J Cell Physiol 184:118–130. https://doi.org/10.1002/(sici)1097-4652(200007)184:1%3c118::Aid-jcp13%3e3.0.Co;2-y
Lund SA, Giachelli CM, Scatena M (2009) The role of osteopontin in inflammatory processes. J Cell Commun Signal 3:311–322. https://doi.org/10.1007/s12079-009-0068-0
Renkl AC, Wussler J, Ahrens T, Thoma K, Kon S, Uede T, Martin SF, Simon JC, Weiss JM (2005) Osteopontin functionally activates dendritic cells and induces their differentiation toward a Th1-polarizing phenotype. Blood 106:946–955. https://doi.org/10.1182/blood-2004-08-3228
Saito K, Nakatomi M, Ida-Yonemochi H, Kenmotsu S, Ohshima H (2011) The expression of GM-CSF and osteopontin in immunocompetent cells precedes the odontoblast differentiation following allogenic tooth transplantation in mice. J Histochem Cytochem 59:518–529. https://doi.org/10.1369/0022155411403314
Inoue M, Moriwaki Y, Arikawa T, Chen YH, Oh YJ, Oliver T, Shinohara ML (2011) Cutting edge: critical role of intracellular osteopontin in antifungal innate immune responses. J Immunol 186:19–23. https://doi.org/10.4049/jimmunol.1002735
Acknowledgements
This study was part of a doctoral research. The authors acknowledge the help of Ms. Shazmeen Aslam and Ms. Anam Razzak of Dow Institute for Advanced Biological and Animal Research for arranging logistics, Mr Mohammad Adeel of Department of Histopathology Dow University of Health Sciences for histology and Dr Abdul Samad Khan of Imam Abdulrahman Bin Faisal University for his valuable suggestions.
Author information
Authors and Affiliations
Contributions
Arshad Hasan, Talat Roome, Mohsin Wahid and Shazia Akbar Ansari were involved in conceptualizing the study. Methodology was developed and performed by Arshad Hasan, Javeria Ali khan, Syeda Neha Ahmed Jilani, Abira Jawed and Amber Kiyani. Supervision of the project was performed by Talat Roome and Mohsin Wahid. Formal analysis was performed by Arshad Hasan, Abira Jawed and Amber Kiyani. The initial writing-review and editing was done by Arshad Hasan, Talat Roome, Mohsin Wahid, Shazia Akbar ANsari, Abira Jawed and Amber Kiyani. Investigation was performed by Arshad Hasan and Syeda Neha Ahmed Jilani. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The study was approved by the Institutional Review Board for Animal Research and Ethics of Dow University of Health Sciences (Letter no. AR.IRB-06/DUHS/Approval/2016/05). This study was in accordance with the ARRIVE guidelines 2.0 for animal research.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hasan, A., Roome, T., Wahid, M. et al. Expression of Toll-like receptor 2, Dectin-1, and Osteopontin in murine model of pulpitis. Clin Oral Invest 27, 1177–1192 (2023). https://doi.org/10.1007/s00784-022-04732-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-022-04732-2