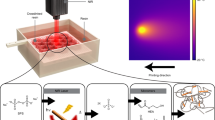
Abstract
The present study highlights the synthesis of a photosensitive norbornene monomer containing an o-nitrobenzyl ester (o-NBE) group, which undergoes controlled bond cleavage upon UV exposure. As the o-NBE chromophores are transparent in the visible light region, photopolymers are readily formed by visible light-induced thiol-ene chemistry using a long-wavelength absorbing photoinitiator. Selective photolysis of the o-NBE moieties is then obtained by irradiation with light in the UV-A spectral region. The sequence-dependent orthogonality of the curing and cleavage reaction are confirmed by FT-IR spectroscopy. Owing to their constrained ring system, norbornene moieties are highly reactive in radical-induced thiol-ene reactions, which enables the vat photopolymerization 3D printing of functional structures with reasonable build speed. Once printed, the wettability of the 3D objects’ surface was conveniently tuned by UV-induced cleavage of the o-NBE groups (release of free carboxylic acids) and related photo-oxidation of the non-reacted thiol moieties (formation of sulfonic acids). With this approach, we were able to introduce wettability gradients onto 3D-printed objects, which is interesting for microfluidic devices and lab-on-chip technologies.
Graphical abstract






Similar content being viewed by others
Data Availability
All data generated or analysed during this study are included in this published article (and its supplementary information files).
References
Crivello JV, Reichmanis E (2014) Chem Mater 26:533
Sangermano M, Razza N, Crivello JV (2014) Macromol Mater Eng 299:775
Schlögl S, Reischl M, Ribitsch V, Kern W (2012) Prog Org Coat 73:54
Bandl C, Kern W, Schlögl S (2020) Int J Adhes Adhes 99:102585
Rueggeberg FA (2011) Dent Mater 27:39
Roy D, Basu PK, Eswaran SV (2002) Reson 7:44
Ligon SC, Liska R, Stampfl J, Gurr M, Mülhaupt R (2017) Chem Rev 117:10212
Hoyle CE, Kinstle JF (1990) Radiation curing of polymeric materials, vol 417. American Chemical Society, Washington, DC
Hoyle CE, Bowman CN (2010) Angew Chem Int Ed 49:1540
Bertrand O, Gohy J-F (2017) Polym Chem 8:52
Cazin I, Rossegger E, de La Guedes Cruz G, Griesser T, Schlögl S (2021) Polymers 13:56–57
Gernhardt M, Blasco E, Hippler M, Blinco J, Bastmeyer M, Wegener M, Frisch H, Barner-Kowollik C (2019) Adv Mater 31:e1901269
Radl S, Roppolo I, Pölzl K, Ast M, Spreitz J, Griesser T, Kern W, Schlögl S, Sangermano M (2017) Polymer 109:349
Radl S, Kreimer M, Manhart J, Griesser T, Moser A, Pinter G, Kalinka G, Kern W, Schlögl S (2015) Polymer 69:159
Song G-S, Heo Y-J, Baek JJ, Lee H, Bae GY, Choi KH, Koh W-G, Shin G (2021) J Polym Sci 59:340
Romano A, Roppolo I, Rossegger E, Schlögl S, Sangermano M (2020) Materials 13:2777
Wang X, Yang Y, Liu C, Guo H, Chen Z, **a J, Liao Y, Tang C-Y, Law W-C (2021) Polymer 229:123961
Sana B, Finne-Wistrand A, Pappalardo D (2022) Mater Today Chem 25:100963
Radl SV, Schipfer C, Kaiser S, Moser A, Kaynak B, Kern W, Schlögl S (2017) Polym Chem 8:1562
Lowe AB (2010) Polym Chem 1:17
Rossegger E, Hennen D, Griesser T, Roppolo I, Schlögl S (2019) Polym Chem 10:1882
Rossegger E, Nees D, Turisser S, Radl S, Griesser T, Schlögl S (2020) Polym Chem 11:3125
Decker C, Bianchi C, Decker D, Morel F (2001) Prog Org Coat 42:253
Hinczewski C, Corbel S, Chartier T (1998) J Eur Ceram Soc 18:583
Acknowledgements
Part of the research work was performed with the “SMART” project. This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Sklodowska-Curie grant agreement No 860108. Part of the research work was performed also within the COMET-Module “Chemitecture“ (project-no.: 21647048) at the Polymer Competence Center Leoben GmbH (PCCL, Austria) within the framework of the COMET-program of the Federal Ministry for Transport, Innovation and Technology and the Federal Ministry for Digital and Economic Affairs with contributions by the Institute of Chemistry of Polymeric Materials (Montanuniversitaet Leoben, Austria). The PCCL is funded by the Austrian Government and the State Governments of Styria, Upper and Lower Austria.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (MOV 21446 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rossegger, E., Li, Y., Frommwald, H. et al. Vat photopolymerization 3D printing with light-responsive thiol-norbornene photopolymers. Monatsh Chem 154, 473–480 (2023). https://doi.org/10.1007/s00706-022-03016-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-022-03016-5