Abstract
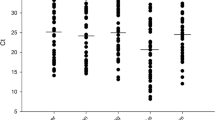
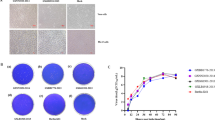
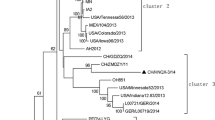
Getah virus (GETV) is an emerging zoonotic virus that can infect humans and many mammals through mosquitoes. In this study, a novel pathogenic GETV strain, GDQY2022, was isolated from a pig farm in Guangdong Province, China. Sequence comparisons and phylogenetic analysis showed that GDQY2022 belongs to group III (GIII) and was most closely related to strain HeN202009-2, with 99.78% nucleotide sequence identity. Histopathological examination revealed significant pathological changes, such as widened alveolar septum in the lungs with mild congestion and hemorrhage. Differences in viral load between tissues were assessed by real-time RT-PCR, and significantly higher levels of GETV were found in abdominal lymph nodes and lungs of subclinically and clinically affected pigs (P < 0.01). This study provides valuable data for understanding the risk of GETV infection in the pig industry and a reliable basis for studying the pathogenic mechanisms and diagnostic surveillance of GETV.




Similar content being viewed by others
References
Li XD, Qiu FX, Yang H, Rao YN, Calisher CH (1992) Isolation of Getah virus from mosquitos collected on Hainan Island, China, and results of a serosurvey. Southeast Asian J Trop Med Public Health 23:730–734
Li Y, Fu S, Guo X, Li X, Li M, Wang L, Gao X, Lei W, Cao L, Lu Z et al (2019) Serological Survey of Getah Virus in Domestic Animals in Yunnan Province, China. Vector Borne Zoonotic Dis 19:59–61. https://doi.org/10.1089/vbz.2018.2273
Karabatsos N (1985) International catalogue of arthropod-borne viruses, vol 3. American Society for Tropical Medicine and Hygiene, San Antonio (TX)
King AM, Lefkowitz E, Adams MJ, Carstens EB (2011) Virus taxonomy: ninth report of the International Committee on Taxonomy of Viruses. Elsevier
Morita K, Igarashi A (1984) Oligonucleotide Fingerprint Analysis of Strains of Getah Virus Isolated in Japan and Malaysia. J Gen Virol 65:1899–1908. https://doi.org/10.1099/0022-1317-65-11-1899
Khwankamon Rattanatumhi N, Prasertsincharoen N, Naimon R, Kuwata H, Shimoda K, Ishijima K, Yonemitsu et al (2022) Shohei Minami, Supriyono, Ngo Thuy Bao Tran, A serological survey and characterization of Getah virus in domestic pigs in Thailand, 2017–2018. Transbound Emerg Dis 69:913–918. https://doi.org/10.1111/tbed.14042
Lu G, Ou J, Ji J, Ren Z, Hu X, Wang C, Li S (2019) Emergence of Getah Virus Infection in Horse With Fever in China, 2018. Frontiers in Microbiology 10: https://www.frontiersin.org/articles/10.3389/fmicb.2019.01416 [Accessed November 5, 2022]
Li Y-Y, Liu H, Fu S-H, Li X-L, Guo X-F, Li M-H, Feng Y, Chen W-X, Wang L-H, Lei W-W et al (2017) From discovery to spread: The evolution and phylogeny of Getah virus. Infect Genet Evol 55:48–55. https://doi.org/10.1016/j.meegid.2017.08.016
Turell MJ, O’guinn ML, Wasieloski LP, Dohm DJ, Lee W, Cho H, Kim H, Burkett DA, Mores CN, Coleman RE et al (2003) Isolation of Japanese Encephalitis and Getah Viruses from Mosquitoes (Diptera: Culicidae) Collected near Camp Greaves, Gyonggi Province, Republic of Korea, 2000. J Med Entomol 40:580–584. https://doi.org/10.1603/0022-2585-40.4.580
Ksiazek TG, Trosper JH, Cross JH, Basaca-Sevilla V (1981) Isolation of Getah virus from Nueva Ecija Province, Republic of the Philippines. Trans R Soc Trop Med Hyg 75:312–313. https://doi.org/10.1016/0035-9203(81)90346-1
l’vov SD, Gromashevski’i VL, Aristova VA, Morozova TN, Skvortsova TM, Gushchina EA, Petrova ES (2000) L’vov DK. [Isolation of Getah virus (Togaviridae, Alfavirus) strains in North- Eastern Asia]. Vopr Virusol 45:14–18
Bryant JE, Crabtree MB, Nam VS, Yen NT, Duc HM, Miller BR (2005) Isolation of arboviruses from mosquitoes collected in northern Vietnam. Am J Trop Med Hyg 73:470–473
Brown CM, Timoney PJ (1998) Getah virus infection of Indian horses. Trop Anim Health Prod 30:241–252. https://doi.org/10.1023/A:1005079229232
Nemoto M, Bannai H, Tsujimura K, Kobayashi M, Kikuchi T, Yamanaka T, Kondo T Getah Virus Infection among Racehorses, Japan, 2014 - Volume 21, Number 5—May 2015 - Emerging Infectious Diseases journal - CDC. https://doi.org/10.3201/eid2105.141975
Fukunaga Y, Kumanomido T, Kamada M (2000) Getah Virus as an Equine Pathogen. Veterinary Clin North America: Equine Pract 16:605–617. https://doi.org/10.1016/S0749-0739(17)30099-8
Kanamitsu M, Taniguchi K, Urasawa S, Ogata T, Wada Y, Wada Y, Saroso JS (1979) Geographic Distribution of Arbovirus Antibodies in Indigenous Human Populations in the Indo-Australian Archipelago. Am J Trop Med Hyg 28:351–363. https://doi.org/10.4269/ajtmh.1979.28.351
Li XD, Qiu FX, Yang H, Rao YN, Calisher CH (1992) Isolation of Getah virus from mosquitos collected on Hainan Island, China, and results of a serosurvey. Southeast Asian J Trop Med Public Health 23:730–734
Nemoto M, Bannai H, Tsujimura K, Kobayashi M, Kikuchi T, Yamanaka T, Kondo T (2015) Getah Virus Infection among Racehorses, Japan, 2014. Emerg Infect Dis 21:883–885. https://doi.org/10.3201/eid2105.141975
Bannai H, Ochi A, Nemoto M, Tsujimura K, Yamanaka T, Kondo T (2016) A 2015 outbreak of Getah virus infection occurring among Japanese racehorses sequentially to an outbreak in 2014 at the same site. BMC Vet Res 12:98. https://doi.org/10.1186/s12917-016-0741-5
Lu G, Ou J, Ji J, Ren Z, Hu X, Wang C, Li S (2019) Emergence of Getah Virus Infection in Horse With Fever in China, 2018. Front Microbiol 10:1416. https://doi.org/10.3389/fmicb.2019.01416
Gang Lu J, Ou J, Ji Z, Ren X, Wang HC, Li S (2019) Emergence of Getah Virus Infection in Horse With Fever in China, 2018. Front Microbiol 10:1416. https://doi.org/10.3389/fmicb.2019.01416
Liu H, Zhang X, Li L-X, Shi N, Sun X-T, Liu Q, ** N-Y, Si X-K (2019) First isolation and characterization of Getah virus from cattle in northeastern China. BMC Vet Res 15:320. https://doi.org/10.1186/s12917-019-2061-z
l’vov SD, Gromashevski’i VL, Aristova VA, Morozova TN, Skvortsova TM, Gushchina EA, Petrova ES (2000) L’vov DK. [Isolation of Getah virus (Togaviridae, Alfavirus) strains in North- Eastern Asia]. Vopr Virusol 45:14–18
Shi N, Zhu X, Qiu X, Cao X, Jiang Z, Lu H, ** N (2022) Origin, genetic diversity, adaptive evolution and transmission dynamics of Getah virus. Transbound Emerg Dis 69:e1037–e1050. https://doi.org/10.1111/tbed.14395
Li B, Wang H, Liang G (2022) Getah Virus (Alphavirus): An Emerging, Spreading Zoonotic Virus. Pathogens 11:945. https://doi.org/10.3390/pathogens11080945
Kuwata R, Shimoda H, Phichitraslip T, Prasertsincharoen N, Noguchi K, Yonemitsu K, Minami S, Supriyono, Tran NTB, Takano A et al (2018) Getah virus epizootic among wild boars in Japan around 2012. Arch Virol 163:2817–2821. https://doi.org/10.1007/s00705-018-3897-4
Rattanatumhi K, Prasertsincharoen N, Naimon N, Kuwata R, Shimoda H, Ishijima K, Yonemitsu K, Minami S, Supriyono, Tran NTB et al (2022) A serological survey and characterization of Getah virus in domestic pigs in Thailand, 2017–2018. Transbounding Emerg Dis 69:913–918. https://doi.org/10.1111/tbed.14042
Yang T, Li R, Hu Y, Yang L, Zhao D, Du L, Li J, Ge M (2018) Yu. An outbreak of Getah virus infection among pigs in China, 2017. Transbound Emerg Dis 65:632–637. https://doi.org/10.1111/tbed.12867
Yang T, Li R, Hu Y, Yang L, Zhao D, Du L, Li J, Ge M, Yu X (2018) An outbreak of Getah virus infection among pigs in China, 2017. Transbound Emerg Dis 65:632–637. https://doi.org/10.1111/tbed.12867
REED LJ, MUENCH H. A SIMPLE METHOD OF ESTIMATING FIFTY, PER CENT ENDPOINTS12 (1938) Am J Epidemiol 27:493–497. https://doi.org/10.1093/oxfordjournals.aje.a118408
Thawng CN, Smith GB (2022) A transcriptome software comparison for the analyses of treatments expected to give subtle gene expression responses. BMC Genomics 23:452. https://doi.org/10.1186/s12864-022-08673-8
Katoh K, Rozewicki J, Yamada KD (2019) MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief Bioinform 20:1160–1166. https://doi.org/10.1093/bib/bbx108
Zhou Z-J, Qiu Y, Pu Y, Huang X, Ge X-Y, BioAider (2020) An efficient tool for viral genome analysis and its application in tracing SARS-CoV-2 transmission. Sustain Cities Soc 63:102466. https://doi.org/10.1016/j.scs.2020.102466
Nguyen L-T, Schmidt HA, von Haeseler A, Minh BQ (2015) IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol 32:268–274. https://doi.org/10.1093/molbev/msu300
Eisenberg E, Levanon EY (2013) Human housekee** genes, revisited. Trends Genet 29:569–574. https://doi.org/10.1016/j.tig.2013.05.010
Zhang Y, Li Y, Guan Z, Yang Y, Zhang J, Sun Q, Li B, Qiu Y, Liu K, Shao D et al (2022) Rapid Differential Detection of Japanese Encephalitis Virus and Getah Virus in Pigs or Mosquitos by a Duplex TaqMan Real-Time RT-PCR Assay. Front Vet Sci 9:839443. https://doi.org/10.3389/fvets.2022.839443
Livak KJ, Schmittgen TD (2001) Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2 – ∆∆CT Method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Scherer WF, Funkenbusch M, Buescher EL, Izumit (1962) null. Sagiyama virus, a new group A arthropod-borne virus from Japan. I. Isolation, immunologic classification, and ecologic observations. Am J Trop Med Hyg 11:255–268. https://doi.org/10.4269/ajtmh.1962.11.255
**ng C, Jiang J, Lu Z, Mi S, He B, Tu C, Liu X, Gong W (2020) Isolation and characterization of Getah virus from pigs in Guangdong province of China. Transbound Emerg Dis tbed13567. https://doi.org/10.1111/tbed.13567
Ren T, Mo Q, Wang Y, Wang H, Nong Z, Wang J, Niu C, Liu C, Chen Y, Ouyang K et al (2020) Emergence and Phylogenetic Analysis of a Getah Virus Isolated in Southern China. Front Vet Sci 7:552517. https://doi.org/10.3389/fvets.2020.552517
de Jong MD, Simmons CP, Thanh TT, Hien VM, Smith GJD, Chau TNB, Hoang DM, Chau NVV, Khanh TH, Dong VC et al (2006) Fatal outcome of human influenza A (H5N1) is associated with high viral load and hypercytokinemia. Nat Med 12:1203–1207. https://doi.org/10.1038/nm1477
Funding
This work was supported by grants from the Guangdong Provincial Natural Science Fund (2017A030310612).
Author information
Authors and Affiliations
Contributions
LZH designed and monitored the project. LZH, JHP, and HQZ wrote and revised the paper, performed all analyses, and interpreted the data. JHP, HQZ, GJY, SNC, XQC, MYZ, HH, YZG, JML, SCL, MJL, and MLM performed the experiments. LZH, JHP, and HQZ collected the samples. All authors revised and approved the paper for publication.
Corresponding author
Ethics declarations
Conflict of interest
GZY, SNC, MLM, and MJL were employed by Guangdong Findergene Biotechnology Co., Ltd., Foshan, Guangdong Province, China. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics statement
The archived porcine samples analyzed in this study were collected from clinically diseased pigs, and animal experiments were not conducted. Thus, no ethical approval was required.
Additional information
Communicated by Patricia Aguilar
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pan, J., Zhang, H., Chen, X. et al. Evolutionary characterization and pathogenicity of Getah virus from pigs in Guangdong Province of China. Arch Virol 168, 258 (2023). https://doi.org/10.1007/s00705-023-05886-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00705-023-05886-4