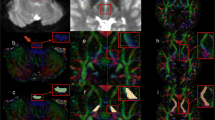
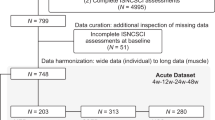
Abstract
Chronic and severe upper-limb motor deficits can result from damage to the corticospinal tract. However, it remains unclear what their characteristics are and whether only corticospinal tract damage determines their characteristics. This study aimed to investigate the clinical characteristics and neural bases of chronic and severe upper-limb motor deficits. Motor deficits, including spasticity, of 45 patients with brain lesions were assessed using clinical scales. Regarding their scores, we conducted a principal component analysis that statistically extracted the clinical characteristics as two principal components. Using these principal components, we investigated the neural bases underlying their characteristics through lesion analyses of lesion volume, lesion sites, corticospinal tract, or other regional white-matter integrity. Principal component analysis showed that the clinical characteristics of chronic and severe upper-limb motor deficits could be described as a comprehensive severity and a trade-off relationship between proximal motor functions and wrist/finger spasticity. Lesion analyses revealed that the comprehensive severity was correlated with corticospinal tract integrity, and the trade-off relationship was associated with the integrity of other regional white matter located anterior to the posterior internal capsule, such as the anterior internal capsule. This study indicates that the severity of chronic and severe upper-limb motor deficits can be determined according to the corticospinal tract integrity, and such motor deficits may be further characterized by the integrity of other white matter, where the corticoreticular pathway can pass through, by forming a trade-off relationship where patients have higher proximal motor functions but more severe wrist/finger spasticity, and vice versa.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- aCR-A:
-
Anterior corona radiata in the affected hemisphere
- aIC-A:
-
Anterior internal capsule in the affected hemisphere
- aIC-U:
-
Anterior internal capsule in the unaffected hemisphere
- ARAT:
-
Action research arm test
- CST:
-
Corticospinal tract
- FA:
-
Fractional anisotropy
- FMA-UE:
-
Fugl-Meyer assessment of upper extremity
- MAS:
-
Modified Ashworth scale
- MNI:
-
Montreal neurological institute
- PC:
-
Principal component
- PCA:
-
Principal component analysis
- pCR-A:
-
Posterior corona radiata in the affected hemisphere
- pIC-A:
-
Posterior internal capsule in the affected hemisphere
- pIC-U:
-
Posterior internal capsule in the unaffected hemisphere
- VLSM:
-
Voxel-based lesion-symptom map**
References
Alt Murphy M, Resteghini C, Feys P, Lamers I (2015) An overview of systematic reviews on upper extremity outcome measures after stroke. BMC Neurol 15:29. https://doi.org/10.1186/s12883-015-0292-6
Ansari NN, Naghdi S, Arab TK, Jalaie S (2008) The interrater and intrarater reliability of the Modified Ashworth Scale in the assessment of muscle spasticity: limb and muscle group effect. NeuroRehabilitation 23:231–237. https://doi.org/10.3233/NRE-2008-23304
Assaf Y, Pasternak O (2008) Diffusion tensor imaging (DTI)-based white matter map** in brain research: a review. J Mol Neurosci 34:51–61. https://doi.org/10.1007/s12031-007-0029-0
Barlow SJ (2016) Identifying the brain regions associated with acute spasticity in patients diagnosed with an ischemic stroke. Somatosens Mot Res 33:104–111. https://doi.org/10.1080/08990220.2016.1197114
Bates E, Wilson SM, Saygin AP, Dick F, Sereno MI, Knight RT, Dronkers NF (2003) Voxel-based lesion–symptom map**. Nat Neurosci 6:448–450. https://doi.org/10.1038/nn1050
Biering-Sørensen F, Nielsen JB, Klinge K (2006) Spasticity-assessment: a review. Spinal Cord 44:708–722. https://doi.org/10.1038/sj.sc.3101928
Binkofski F, Seitz RJ, Hackländer T, Pawelec D, Mau J, Freund HJ (2001) Recovery of motor functions following hemiparetic stroke: a clinical and magnetic resonance-morphometric study. Cerebrovasc Dis 11:273–281. https://doi.org/10.1159/000047650
Boyne P, DiFrancesco M, Awosika OO, Williamson B, Vannest J (2022) Map** the human corticoreticular pathway with multimodal delineation of the gigantocellular reticular nucleus and high-resolution diffusion tractography. J Neurol Sci 434:120091. https://doi.org/10.1016/j.jns.2021.120091
Carod-Artal J, Egido JA, González JL, Varela de Seijas E (2000) Quality of life among stroke survivors evaluated 1 year after stroke. Stroke 31:2995–3000. https://doi.org/10.1161/01.STR.31.12.2995
Cheng B, Forkert ND, Zavaglia M et al (2014) Influence of stroke infarct location on functional outcome measured by the modified rankin scale. Stroke 45:1695–1702. https://doi.org/10.1161/STROKEAHA.114.005152
DeMarco AT, Turkeltaub PE (2018) A multivariate lesion symptom map** toolbox and examination of lesion-volume biases and correction methods in lesion-symptom map**. Hum Brain Mapp 39:4169–4182. https://doi.org/10.1002/hbm.24289
Do KH, Yeo SS, Lee J, Jang SH (2013) Injury of the corticoreticular pathway in patients with proximal weakness following cerebral infarct: diffusion tensor tractography study. Neurosci Lett 546:21–25. https://doi.org/10.1016/j.neulet.2013.04.040
Frenkel-Toledo S, Fridberg G, Ofir S et al (2019) Lesion location impact on functional recovery of the hemiparetic upper limb. Plos one 14:e0219738. https://doi.org/10.1371/journal.pone.0219738
Fries W, Danek A, Scheidtmann K, Hamburger C (1993) Motor recovery following capsular stroke: role of descending pathways from multiple motor areas. Brain 116:369–382. https://doi.org/10.1093/brain/116.2.369
Guilford JP (1956) Fundamental statistics in psychology and education. McGraw Hill, New York
Hedges LV (1981) Distribution theory for Glass’s estimator of effect size and related estimators. J Educ Stat 6:107–128. https://doi.org/10.3102/10769986006002107
Hendricks HT, van Limbeek J, Geurts AC, Zwarts MJ (2002) Motor recovery after stroke: a systematic review of the literature. Arch Phys Med Rehabil 83:1629–1637. https://doi.org/10.1053/apmr.2002.35473
Higano S, Zhong J, Shrier DA, Shibata DK, Takase Y, Wang H, Numaguchi Y (2001) Diffusion anisotropy of the internal capsule and the corona radiata in association with stroke and tumors as measured by diffusion-weighted MR imaging. AJNR Am J Neuroradiol 22:456–463
Jang SH (2009) The role of the corticospinal tract in motor recovery in patients with a stroke: a review. NeuroRehabilitation 24:285–290. https://doi.org/10.3233/NRE-2009-0480
Jang SH (2014) The corticospinal tract from the viewpoint of brain rehabilitation. J Rehabil Med 46:193–199. https://doi.org/10.2340/16501977-1782
Jang SH, Choi EB (2022) Relation between the corticospinal tract state and activities of daily living in patients with intracerebral hemorrhage. Stroke 53:544–551. https://doi.org/10.1161/STROKEAHA.121.034939
Jang SH, Lee SJ (2019) Corticoreticular tract in the human brain: a mini review. Front Neurol. https://doi.org/10.3389/fneur.2019.01188
Jang SH, Seo JP (2015) The anatomical location of the corticobulbar tract at the corona radiata in the human brain: diffusion tensor tractography study. Neurosci Lett 590:80–83. https://doi.org/10.1016/j.neulet.2015.01.072
Jang SH, Kim K, Kim SH, Son SM, Jang WH, Kwon HG (2014) The relation between motor function of stroke patients and diffusion tensor imaging findings for the corticospinal tract. Neurosci Lett 572:1–6. https://doi.org/10.1016/j.neulet.2014.04.044
Jang SH, Choi BY, Kim SH, Chang CH, Jung YJ, Yeo SS (2015) Injury of the corticoreticular pathway in subarachnoid haemorrhage after rupture of a cerebral artery aneurysm. J Rehabil Med 47:133–137. https://doi.org/10.2340/16501977-1896
Jolliffe IT, Cadima J (2016) Principal component analysis: a review and recent developments. Philos Trans A Math Phys Eng Sci 374:20150202. https://doi.org/10.1098/rsta.2015.0202
Kably B, Drew T (1998) Corticoreticular pathways in the cat. I. Projection patterns and collaterization. J Neurophysiol 80:389–405. https://doi.org/10.1152/jn.1998.80.1.389
Kaneko F, Shindo K, Yoneta M, Okawada M, Akaboshi K, Liu M (2019) A case series clinical trial of a novel approach using augmented reality that inspires self-body cognition in patients with stroke: effects on motor function and resting-state brain functional connectivity. Front Syst Neurosci. https://doi.org/10.3389/fnsys.2019.00076
Krzanowski WJ, Lai YT (1988) A criterion for determining the number of groups in a data set using sum-of-squares clustering. Biometrics 44:23–34. https://doi.org/10.2307/2531893
Lawrence DG, Kuypers HGJM (1968) The functional organization of the motor system in the monkey: I. The effects of bilateral pyramidal lesions. Brain 91:1–14. https://doi.org/10.1093/brain/91.1.1
Lee KB, Hong BY, Kim JS et al (2019) Which brain lesions produce spasticity? An observational study on 45 stroke patients. Plos one 14:e0210038. https://doi.org/10.1371/journal.pone.0210038
Lee AY, Choi KT, Chang MC (2020) Prediction of muscle loss after stroke by analysis of corticospinal tract. Transl Neurosci 11:328–333. https://doi.org/10.1515/tnsci-2020-0114
Lemon SC, Roy J, Clark MA, Friedmann PD, Rakowski W (2003) Classification and regression tree analysis in public health: methodological review and comparison with logistic regression. Ann Behav Med 26:172–181. https://doi.org/10.1207/S15324796ABM2603_02
Lewis RJ (2000) An Introduction to Classification and Regression Tree (CART) Analysis. http://www.saem.org/download/lewis1.pdf. Accessed 17 Feb 2022
Li S, Francisco GE (2015) New insights into the pathophysiology of post-stroke spasticity. Front Hum Neurosci. https://doi.org/10.3389/fnhum.2015.00192
Li S, Chen YT, Francisco GE, Zhou P, Rymer WZ (2019) A unifying pathophysiological account for post-stroke spasticity and disordered motor control. Front Neurol. https://doi.org/10.3389/fneur.2019.00468
Lindenberg R, Renga V, Zhu LL, Betzler F, Alsop D, Schlaug G (2010) Structural integrity of corticospinal motor fibers predicts motor impairment in chronic stroke. Neurology 74:280–287. https://doi.org/10.1212/WNL.0b013e3181ccc6d9
Löuvbld KO, Baird AE, Schlaug G et al (1997) Ischemic lesion volumes in acute stroke by diffusion-weighted magnetic resonance imaging correlate with clinical outcome. Ann Neurol 42:164–170. https://doi.org/10.1002/ana.410420206
Matsuyama K, Drew T (1997) Organization of the projections from the pericruciate cortex to the pontomedullary brainstem of the cat: a study using the anterograde tracer Phaseolus vulgaris-leucoagglutinin. J Comp Neurol 389:617–641. https://doi.org/10.1002/(SICI)1096-9861(19971229)389:4%3c617::AID-CNE6%3e3.0.CO;2-3
Meyer S, Kessner SS, Cheng B et al (2016) Voxel-based lesion-symptom map** of stroke lesions underlying somatosensory deficits. NeuroImage Clin 10:257–266. https://doi.org/10.1016/j.nicl.2015.12.005
Miyawaki Y, Yoneta M, Okawada M, Kawakami M, Liu M, Kaneko F (2022) Model-based analyses for the causal relationship between post-stroke impairments and functional brain connectivity regarding the effects of kinesthetic illusion therapy combined with conventional exercise. Front Syst Neurosci. https://doi.org/10.3389/fnsys.2021.804263
Møller M, Frandsen J, Andersen G, Gjedde A, Vestergaard-Poulsen P, Østergaard L (2007) Dynamic changes in corticospinal tracts after stroke detected by fibretracking. J Neurol Neurosurg Psychiatry 78:587–592. https://doi.org/10.1136/jnnp.2006.100248
Mori S, Oishi K, Jiang H et al (2008) Stereotaxic white matter atlas based on diffusion tensor imaging in an ICBM template. Neuroimage 40:570–582. https://doi.org/10.1016/j.neuroimage.2007.12.035
Muellbacher W, Richards C, Ziemann U et al (2002) Improving hand function in chronic stroke. Arch Neurol 59:1278. https://doi.org/10.1001/archneur.59.8.1278
Owen M, Ingo C, Dewald JPA (2017) Upper extremity motor impairments and microstructural changes in bulbospinal pathways in chronic hemiparetic stroke. Front Neurol. https://doi.org/10.3389/fneur.2017.00257
Picelli A, Tamburin S, Gajofatto F et al (2014) Association between severe upper limb spasticity and brain lesion location in stroke patients. BioMed Res Int 2014:e162754. https://doi.org/10.1155/2014/162754
Puig J, Pedraza S, Blasco G et al (2010) Wallerian degeneration in the corticospinal tract evaluated by diffusion tensor imaging correlates with motor deficit 30 days after middle cerebral artery ischemic stroke. Am J Neuroradiol 31:1324–1330. https://doi.org/10.3174/ajnr.a2038
Radlinska B, Ghinani S, Leppert IR, Minuk J, Pike GB, Thiel A (2010) Diffusion tensor imaging, permanent pyramidal tract damage, and outcome in subcortical stroke. Neurology 75:1048–1054. https://doi.org/10.1212/WNL.0b013e3181f39aa0
Rorden C, Karnath HO, Bonilha L (2007) Improving lesion-symptom map**. J Cogn Neurosci 19:1081–1088. https://doi.org/10.1162/jocn.2007.19.7.1081
Rorden C, Bonilha L, Fridriksson J, Bender B, Karnath HO (2012) Age-specific CT and MRI templates for spatial normalization. Neuroimage 61:957–965. https://doi.org/10.1016/j.neuroimage.2012.03.020
Schaechter JD, Fricker ZP, Perdue KL et al (2009) Microstructural status of ipsilesional and contralesional corticospinal tract correlates with motor skill in chronic stroke patients. Hum Brain Mapp 30:3461–3474. https://doi.org/10.1002/hbm.20770
Schulz R, Park CH, Boudrias MH, Gerloff C, Hummel FC, Ward NS (2012) Assessing the Integrity of corticospinal pathways from primary and secondary cortical motor areas after stroke. Stroke 43:2248–2251. https://doi.org/10.1161/STROKEAHA.112.662619
Seitz RJ, Höflich P, Binkofski F, Tellmann L, Herzog H, Freund HJ (1998) Role of the premotor cortex in recovery from middle cerebral artery infarction. Arch Neurol 55:1081–1088. https://doi.org/10.1001/archneur.55.8.1081
Sheean G, McGuire JR (2009) Spastic hypertonia and movement disorders: pathophysiology, clinical presentation, and quantification. PM R 1:827–833. https://doi.org/10.1016/j.pmrj.2009.08.002
Stinear CM, Barber PA, Smale PR, Coxon JP, Fleming MK, Byblow WD (2007) Functional potential in chronic stroke patients depends on corticospinal tract integrity. Brain 130:170–180. https://doi.org/10.1093/brain/awl333
Sullivan EV, Zahr NM, Rohlfing T, Pfefferbaum A (2010) Fiber tracking functionally distinct components of the internal capsule. Neuropsychologia 48:4155–4163. https://doi.org/10.1016/j.neuropsychologia.2010.10.023
Trompetto C, Marinelli L, Mori L et al (2014) Pathophysiology of spasticity: implications for neurorehabilitation. BioMed Res Int 2014:e354906. https://doi.org/10.1155/2014/354906
van der Vliet R, Selles RW, Andrinopoulou ER et al (2020) Predicting upper limb motor impairment recovery after stroke: a mixture model. Ann Neurol 87:383–393. https://doi.org/10.1002/ana.25679
Wang X, Casadio M, Weber KA, Mussa-Ivaldi FA, Parrish TB (2014) White matter microstructure changes induced by motor skill learning utilizing a body machine interface. Neuroimage 88:32–40. https://doi.org/10.1016/j.neuroimage.2013.10.066
Ward NS, Cohen LG (2004) Mechanisms underlying recovery of motor function after stroke. Arch Neurol 61:1844–1848. https://doi.org/10.1001/archneur.61.12.1844
Woytowicz EJ, Rietschel JC, Goodman RN et al (2017) Determining levels of upper extremity movement impairment by applying a cluster analysis to the Fugl-Meyer assessment of the upper extremity in chronic stroke. Arch Phys Med Rehabil 98:456–462. https://doi.org/10.1016/j.apmr.2016.06.023
Wu O, Cloonan L, Mocking SJT et al (2015) Role of acute lesion topography in initial ischemic stroke severity and long-term functional outcomes. Stroke 46:2438–2444. https://doi.org/10.1161/STROKEAHA.115.009643
Yeh FC (2020) Shape analysis of the human association pathways. Neuroimage 223:117329. https://doi.org/10.1016/j.neuroimage.2020.117329
Yeh FC, Tseng WYI (2011) NTU-90: A high angular resolution brain atlas constructed by q-space diffeomorphic reconstruction. Neuroimage 58:91–99. https://doi.org/10.1016/j.neuroimage.2011.06.021
Yeh FC, Panesar S, Barrios J et al (2019) Automatic removal of false connections in diffusion MRI tractography using topology-informed pruning (TIP). Neurotherapeutics 16:52–58. https://doi.org/10.1007/s13311-018-0663-y
Yeo SS, Chang MC, Kwon YH, Jung YJ, Jang SH (2012) Corticoreticular pathway in the human brain: diffusion tensor tractography study. Neurosci Lett 508:9–12. https://doi.org/10.1016/j.neulet.2011.11.030
Yuan KH, Bentler PM (2000) Three likelihood-based methods for mean and covariance structure analysis with nonnormal missing data. Sociol Methodol 30:165–200. https://doi.org/10.1111/0081-1750.00078
Zhu LL, Lindenberg R, Alexander MP, Schlaug G (2010) Lesion load of the corticospinal tract predicts motor impairment in chronic stroke. Stroke 41:910–915. https://doi.org/10.1161/STROKEAHA.109.577023
Acknowledgements
This study was supported by the Japan Agency for Medical Research and Development (AMED) (Grant Number JP18he0402255) and the Japan Society for the Promotion of Science (JSPS) KAKENHI (Grant Number JP19H01088). We appreciate Dr. Norihiro Suzuki and the Radiology Department of the Shonan Keiiku Hospital for their academic support and encouragement.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Miyawaki, Y., Yoneta, M., Okawada, M. et al. Neural bases characterizing chronic and severe upper-limb motor deficits after brain lesion. J Neural Transm 130, 663–677 (2023). https://doi.org/10.1007/s00702-023-02622-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-023-02622-9