Abstract
Objective
The aim of this study was to investigate the different ictal onset stereoelectroencephalography patterns (IOPs) in patients with drug-resistant temporal lobe epilepsy (TLE). We examined whether the IOPs relate to different TLE subtypes, MRI findings, and underlying pathologies, and we evaluated their prognostic value for predicting the surgical outcome.
Methods
We retrospectively analyzed data from patients with TLE who underwent stereoelectroencephalography (SEEG) monitoring followed by surgical resection between January 2018 and January 2020. The SEEG recordings were independently analyzed by two epileptologists.
Results
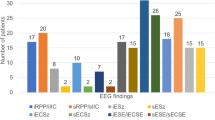
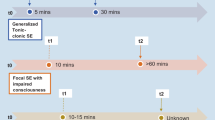
Forty-five patients were included in the study, and 61seizures were analyzed. Five IOPs were identified: low voltage fast activity (LVFA; 44.3%), spike-and-wave activity (16.4%), low frequency high-amplitude periodic spikes (LFPS; 18%), a burst of high-amplitude polyspikes (8.2%), and rhythmic sharp activity at ≤ 13 Hz (13.1%). Thirty-two patients were found to have a single IOP, while the other 13 patients had two or more IOPs. All five IOPs were found to occur in the medial temporal lobe epilepsy (MTLE), while four IOPs occurred in the lateral temporal lobe epilepsy (LTLE). The LFPS was a common IOP that could distinguish MTLE from LTLE (x2 = 7.046, p = 0.011). Among the MTLE patients, the LFPS was exclusively seen in cases of hippocampal sclerosis (x2 = 5.058, p = 0.038), while the LVFA was associated with nonspecific histology (x2 = 6.077, p = 0.023). The IOPs were not found to differ according to whether the MRI scans were positive or negative. After surgery, patients achieved the higher seizure-free rate at 81.8% and 77.8%, respectively, if the LFPS and LVFA were the predominant patterns. Multiple IOPs or a negative MRI did not indicate a poor prognosis.
Conclusions
Five distinct IOPs were identified in the patients with TLE. The differences found have important clinical implications and could provide complementary information for surgical decision-making, especially in MRI-negative patients.


Similar content being viewed by others
References
Alter AS, Dhamija R, McDonough TL, Shen S, McBrian DK, Mandel AM, McKhann GM, Feldstein NA, Akman CI (2019) Ictal onset patterns of subdural intracranial electroencephalogram in children: how helpful for predicting epilepsy surgery outcome? Epilepsy Res 149:44–52. https://doi.org/10.1016/j.eplepsyres.2018.10.008
Avoli M, de Curtis M, Gnatkovsky V, Gotman J, Kohling R, Levesque M, Manseau F, Shiri Z, Williams S (2016) Specific imbalance of excitatory/inhibitory signaling establishes seizure onset pattern in temporal lobe epilepsy. J Neurophysiol 115:3229–3237. https://doi.org/10.1152/jn.01128.2015
Blumcke I, Thom M, Aronica E, Armstrong DD, Vinters HV, Palmini A, Jacques TS, Avanzini G, Barkovich AJ, Battaglia G, Becker A, Cepeda C, Cendes F, Colombo N, Crino P, Cross JH, Delalande O, Dubeau F, Duncan J, Guerrini R, Kahane P, Mathern G, Najm I, Ozkara C, Raybaud C, Represa A, Roper SN, Salamon N, Schulze-Bonhage A, Tassi L, Vezzani A, Spreafico R (2011) The clinicopathologic spectrum of focal cortical dysplasias: a consensus classification proposed by an ad hoc Task Force of the ILAE Diagnostic Methods Commission. Epilepsia 52:158–174. https://doi.org/10.1111/j.1528-1167.2010.02777.x
Bulacio JC, Chauvel P, McGonigal A (2016) Stereoelectroencephalography: interpretation. J Clin Neurophysiol 33:503–510. https://doi.org/10.1097/WNP.0000000000000305
Cardinale F, Rizzi M, Vignati E, Cossu M, Castana L, d’Orio P, Revay M, Costanza MD, Tassi L, Mai R, Sartori I, Nobili L, Gozzo F, Pelliccia V, Mariani V, Lo Russo G, Francione S (2019) Stereoelectroencephalography: retrospective analysis of 742 procedures in a single centre. Brain 142:2688–2704. https://doi.org/10.1093/brain/awz196
Chassoux F, Navarro V, Catenoix H, Valton L, Vignal JP (2018) Planning and management of SEEG. Neurophysiol Clin 48:25–37. https://doi.org/10.1016/j.neucli.2017.11.007
Conrad EC, Tomlinson SB, Wong JN, Oechsel KF, Shinohara RT, Litt B, Davis KA, Marsh ED (2020) Spatial distribution of interictal spikes fluctuates over time and localizes seizure onset. Brain 143:554–569. https://doi.org/10.1093/brain/awz386
Dolezalova I, Brazdil M, Hermanova M, Horakova I, Rektor I, Kuba R (2013) Intracranial EEG seizure onset patterns in unilateral temporal lobe epilepsy and their relationship to other variables. Clin Neurophysiol 124:1079–1088. https://doi.org/10.1016/j.clinph.2012.12.046
Elwan S, Alexopoulos A, Silveira DC, Kotagal P (2018) Lateralizing and localizing value of seizure semiology: comparison with scalp EEG, MRI and PET in patients successfully treated with resective epilepsy surgery. Seizure 61:203–208. https://doi.org/10.1016/j.seizure.2018.08.026
Engel J Jr, McDermott MP, Wiebe S, Langfitt JT, Stern JM, Dewar S, Sperling MR, Gardiner I, Erba G, Fried I, Jacobs M, Vinters HV, Mintzer S, Kieburtz K, EarlyRandomizedSurgicalEpilepsy Trial Study G (2012) Early surgical therapy for drug-resistant temporal lobe epilepsy: a randomized trial. JAMA 307:922–930. https://doi.org/10.1001/jama.2012.220
Faught E, Kuzniecky RI, Hurst DC (1992) Ictal EEG wave forms from epidural electrodes predictive of seizure control after temporal lobectomy. Electroencephalogr Clin Neurophysiol 83:229–235. https://doi.org/10.1016/0013-4694(92)90116-y
Feng R, Farrukh Hameed NU, Hu J, Lang L, He J, Wu D, Fan Z, Jiang S, Guo Q, Liu F, Mao Y, Li C, Sun B, Chen L, Pan L (2020) Ictal stereo-electroencephalography onset patterns of mesial temporal lobe epilepsy and their clinical implications. Clin Neurophysiol 131:2079–2085. https://doi.org/10.1016/j.clinph.2020.05.033
Gao R, Yu T, Xu C, Zhang X, Yan X, Ni D, Zhang X, Ma K, Qiao L, Zhu J, Wang X, Ren Z, Zhang X, Zhang G, Li Y (2020) The value of magnetoencephalography for stereo-EEG-guided radiofrequency thermocoagulation in MRI-negative epilepsy. Epilepsy Res 163:106322. https://doi.org/10.1016/j.eplepsyres.2020.106322
Goldstein HE, Youngerman BE, Shao B, Akman CI, Mandel AM, McBrian DK, Riviello JJ, Sheth SA, McKhann GM, Feldstein NA (2018) Safety and efficacy of stereoelectroencephalography in pediatric focal epilepsy: a single-center experience. J Neurosurg-Pediatr 22:444–452. https://doi.org/10.3171/2018.5.Peds1856
Gonzalez Otarula KA, von Ellenrieder N, Cuello-Oderiz C, Dubeau F, Gotman J (2019) High-frequency oscillation networks and surgical outcome in adult focal epilepsy. Ann Neurol 85:485–494. https://doi.org/10.1002/ana.25442
Jimenez-Jimenez D, Martin-Lopez D, Masood MA, Selway RP, Valentin A, Alarcon G (2016) Prognostic value of the second ictal intracranial pattern for the outcome of epilepsy surgery. Clin Neurophysiol 127:230–237. https://doi.org/10.1016/j.clinph.2015.07.001
Jimenez-Jimenez D, Nekkare R, Flores L, Chatzidimou K, Bodi I, Honavar M, Mullatti N, Elwes RD, Selway RP, Valentin A, Alarcon G (2015) Prognostic value of intracranial seizure onset patterns for surgical outcome of the treatment of epilepsy. Clin Neurophysiol 126:257–267. https://doi.org/10.1016/j.clinph.2014.06.005
Jobst BC, Cascino GD (2015) Resective epilepsy surgery for drug-resistant focal epilepsy: a review. JAMA 313:285–293. https://doi.org/10.1001/jama.2014.17426
Jones AL, Cascino GD (2016) Evidence on use of neuroimaging for surgical treatment of temporal lobe epilepsy: a systematic review. Jama Neurol 73:464–470. https://doi.org/10.1001/jamaneurol.2015.4996
Josephson CB, Dykeman J, Fiest KM, Liu X, Sadler RM, Jette N, Wiebe S (2013) Systematic review and meta-analysis of standard vs selective temporal lobe epilepsy surgery. Neurology 80:1669–1676. https://doi.org/10.1212/WNL.0b013e3182904f82
Kahane P, Bartolomei F (2010) Temporal lobe epilepsy and hippocampal sclerosis: lessons from depth EEG recordings. Epilepsia 51(Suppl 1):59–62. https://doi.org/10.1111/j.1528-1167.2009.02448.x
Lagarde S, Bonini F, McGonigal A, Chauvel P, Gavaret M, Scavarda D, Carron R, Regis J, Aubert S, Villeneuve N, Giusiano B, Figarella-Branger D, Trebuchon A, Bartolomei F (2016) Seizure-onset patterns in focal cortical dysplasia and neurodevelopmental tumors: relationship with surgical prognosis and neuropathologic subtypes. Epilepsia 57:1426–1435. https://doi.org/10.1111/epi.13464
Lagarde S, Buzori S, Trebuchon A, Carron R, Scavarda D, Milh M, McGonigal A, Bartolomei F (2019) The repertoire of seizure onset patterns in human focal epilepsies: determinants and prognostic values. Epilepsia 60:85–95. https://doi.org/10.1111/epi.14604
Lee SA, Spencer DD, Spencer SS (2000) Intracranial EEG seizure-onset patterns in neocortical epilepsy. Epilepsia 41:297–307. https://doi.org/10.1111/j.1528-1157.2000.tb00159.x
Minotti L, Montavont A, Scholly J, Tyvaert L, Taussig D (2018) Indications and limits of stereoelectroencephalography (SEEG). Neurophysiol Clin 48:15–24. https://doi.org/10.1016/j.neucli.2017.11.006
Mullin JP, Shriver M, Alomar S, Najm I, Bulacio J, Chauvel P, Gonzalez-Martinez J (2016) Is SEEG safe? A systematic review and meta-analysis of stereo-electroencephalography-related complications. Epilepsia 57:386–401. https://doi.org/10.1111/epi.13298
Park YD, Murro AM, King DW, Gallagher BB, Smith JR, Yaghmai F (1996) The significance of ictal depth EEG patterns in patients with temporal lobe epilepsy. Electroencephalogr Clin Neurophysiol 99:412–415. https://doi.org/10.1016/s0013-4694(96)95120-2
Peltola ME, Trebuchon A, Lagarde S, Scavarda D, Carron R, Metsahonkala L, Lauronen L, Lepine A, Villeneuve N, Bartolomei F (2020) Anatomoelectroclinical features of SEEG-confirmed pure insular-onset epilepsy. Epilepsy Behav 105:106964. https://doi.org/10.1016/j.yebeh.2020.106964
Perucca P, Dubeau F, Gotman J (2014) Intracranial electroencephalographic seizure-onset patterns: effect of underlying pathology. Brain 137:183–196. https://doi.org/10.1093/brain/awt299
Schuh LA, Henry TR, Ross DA, Smith BJ, Elisevich K, Drury I (2000) Ictal spiking patterns recorded from temporal depth electrodes predict good outcome after anterior temporal lobectomy. Epilepsia 41:316–319. https://doi.org/10.1111/j.1528-1157.2000.tb00161.x
Singh S, Sandy S, Wiebe S (2015) Ictal onset on intracranial EEG: do we know it when we see it? State of the evidence. Epilepsia 56:1629–1638. https://doi.org/10.1111/epi.13120
Spanedda F, Cendes F, Gotman J (1997) Relations between EEG seizure morphology, interhemispheric spread, and mesial temporal atrophy in bitemporal epilepsy. Epilepsia 38:1300–1314. https://doi.org/10.1111/j.1528-1157.1997.tb00068.x
Velasco AL, Wilson CL, Babb TL, Engel J Jr (2000) Functional and anatomic correlates of two frequently observed temporal lobe seizure-onset patterns. Neural Plast 7:49–63. https://doi.org/10.1155/NP.2000.49
Wennberg R, Arruda F, Quesney LF, Olivier A (2002) Preeminence of extrahippocampal structures in the generation of mesial temporal seizures: evidence from human depth electrode recordings. Epilepsia 43:716–726. https://doi.org/10.1046/j.1528-1157.2002.31101.x
Wetjen NM, Marsh WR, Meyer FB, Cascino GD, So E, Britton JW, Stead SM, Worrell GA (2009) Intracranial electroencephalography seizure onset patterns and surgical outcomes in nonlesional extratemporal epilepsy. J Neurosurg 110:1147–1152. https://doi.org/10.3171/2008.8.JNS17643
Wiebe S, Blume WT, Girvin JP, Eliasziw M, Effectiveness, Efficiency of Surgery for Temporal Lobe EpilepsyStudy G (2001) A randomized, controlled trial of surgery for temporal-lobe epilepsy. N Engl J Med 345:311–318. https://doi.org/10.1056/NEJM200108023450501
Wieser HG, Blume WT, Fish D, Goldensohn E, Hufnagel A, King D, Sperling MR, Luders H, Pedley TA, Commission on Neurosurgery of the International LeagueAgainst E (2001) ILAE Commission Report. Proposal for a new classification of outcome with respect to epileptic seizures following epilepsy surgery. Epilepsia 42:282–286
Yu KJ, Yu T, Qiao L, Liu C, Wang XY, Zhou XX, Ni DY, Zhang GJ, Li YJ (2018) Electrical stimulation of the insulo-opercular region: visual phenomena and altered body-ownership symptoms. Epilepsy Res 148:96–106. https://doi.org/10.1016/j.eplepsyres.2018.09.014
Acknowledgements
We thank Jessica Foxton, PhD, from Liwen Bianji (Edanz) (www.liwenbianji.cn/) for editing the English text of a draft of this manuscript.
Funding
The study was supported by grants from Bei**g Natural Science Foundation (L182015), Bei**g Hospitals Authority Ascent Plan (DFL20190801), Capital Health Research and Development of Special Project (2020–1-2013).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Xuanwu Hospital, Capital Medical University, China, according to the Declaration of Helsinki. Informed consent was obtained from all patients or their guardians.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Comments
Nice retrospective study from a large volume epilepsy surgical center demonstrating how stereoencephalograpy recording of ictal onset patterns in temporal lope epilepsy may distinguish between MTLE and LTLE and support surgical success prognostication.
Carsten Reidies Bjarkam
Aalborg, Denmark
This article is part of the Topical Collection on Functional Neurosurgery — Epilepsy
Rights and permissions
About this article
Cite this article
Cui, D., Gao, R., Xu, C. et al. Ictal onset stereoelectroencephalography patterns in temporal lobe epilepsy: type, distribution, and prognostic value. Acta Neurochir 164, 555–563 (2022). https://doi.org/10.1007/s00701-022-05122-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-022-05122-z