Abstract
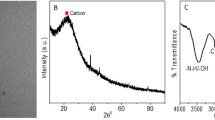
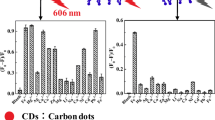
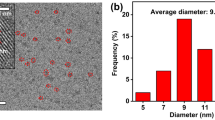
Carbon dots doped with N/Ce, N/Eu and N/Tb were prepared by a microwave based hydrothermal technique. The fluorescence of the N/Ce co-doped carbon dots (CDN/Ce) is strongest. They have excitation/emission maxima at 340/441 nm. CDN/Ce was characterized by scanning electron microscopy, infrared and fluorescence spectroscopy. On addition of the nucleic acid aptamer (Apt) against arsenic(III) in pH 7 solution, the blue fluorescence of the doped carbon dots is partially quenched due to electrostatic interaction. On addition of As(III), it will bind to the aptamer, and the carbon dots are released. Hence, fluorescence becomes gradually restored. In addition, the resonance Rayleigh scattering signal (measured at 340 nm) is reduced. This dual-mode assay works in the 0.5–5.8 μg·L−1 As(III) concentration range and has a 0.2 μg·L−1 detection limit.

Schematic representation of fluorometric and resonance Rayleigh scattering dual mode analysis of As3+ by using coupled Apt and CDN/Ce probes. Apt: Aptamer. CD: Carbon dot. Flu: Fluorescence. RRS: Resonance Rayleigh scattering.








Similar content being viewed by others
References
Ellington AD, Szostak JW (1992) Selection in vitro of single-stranded DNA molecules that fold into specific ligand-binding structures. Nature 355:850–852
Zhang B, Wei C (2017) Highly sensitive and selective fluorescence detection of Hg2+ based on turn-on aptamer DNA silver nanoclusters. RSC Adv 7:56289–56295
Lin Z, Li M, Lv S, Zhang K, Lu M (2017) In-situ synthesis of fluorescent polydopamine nanoparticles coupled with enzyme-controlled dissolution of MnO2 nanoflakes for sensitive immunoassay of cancer biomarkers. J Mater Chem B 5:8506–8513
Tang D, Lin Y, Zhou Q, Lin Y, Li P (2014) Low-cost and highly sensitive immunosensing platform for aflatoxins using one-step competitive displacement reaction mode and portable glucometer-based detection. Anal Chem 86:11451–11458
Li D, Song S, Fan C (2010) Target-responsive structural switching for nucleic acid-based sensors. Accounts Chem Res 43:631–641
Du J, Cui J, **g C (2014) Rapid in situ identification of arsenic species using a portable Fe3O4@Ag SERS sensor. Chem Commun 50:347–349
Wu Y, Liu L, Zhan S, Wang F, Zhou P (2012) Ultrasensitive aptamer biosensor for arsenic(III) detection in aqueous solution based on surfactant-induced aggregation of gold nanoparticles. Analyst 137:4171–4178
Divsar F, Habibzadeh K, Shariati S, Shahriarinour M (2015) Aptamer conjugated silver nanoparticles for the colorimetric detection of arsenic ions using response surface methodology. Anal Methods 7:4568–4576
Lv X, Zhang Y, Liu G, Du L, Wang S (2017) Aptamer-based fluorescent detection of ochratoxin a by quenching of gold nanoparticles. RSC Adv 7:16290–16294
Wang P, Wan Y, Ali A, Deng S, Su Y (2016) Aptamer-wrapped gold nanoparticles for the colorimetric detection of omethoate. Sci China Chem 59:237–242
Dehghani S, Danesh NM, Ramezani M, Alibolandi M, Lavaee P, Nejabat M, Abnous K, Taghdisi SM (2018) A label-free fluorescent aptasensor for detection of kanamycin based on dsDNA-capped mesoporous silica nanoparticles and rhodamine B. Anal Chim Acta 2670:142–147
Tang M, Wen G, Luo Y, Kang C, Liang A (2014) A label-free DNAzyme-cleaving fluorescence method for the determination of trace Pb2+ based on catalysis of AuPd nanoalloy on the reduction of rhodamine 6G: DNAzyme-cleaving fluorescence method for Pb. Luminescence 30:296–302
Ikanovic M, Rudzinski WE, Bruno JG, Allman A, Carrillo MP (2007) Fluorescence assay based on aptamer-quantum dot binding to bacillus thuringiensis spores. J Fluoresc 17:193–199
Taghdisi SM, Danesh NM, Beheshti HR, Ramezani M, Abnous K (2016) A novel fluorescent aptasensor based on gold and silica nanoparticles for the ultrasensitive detection of ochratoxin a. Nanoscale 8:3439–3446
Mishra M, Chakravarty A, Bhowmik K, Bhowmik K, De G (2015) Carbon nanodot- ORMOSIL fluorescent paint and films. J Mater Chem C 3:714–719
Du Y, Guo S (2016) Chemically doped fluorescent carbon and graphene quantum dots for bioimaging, sensor, catalytic and photoelectronic applications. Nanoscale 8:2532–2543
Ding H, Wei J, **ong H (2014) Nitrogen and sulfur co-doped carbon dots with strong blue luminescence. Nanoscale 6:13817–13823
Barati A, Shamsipur M, Abdollahi H (2015) Hemoglobin detection using carbon dots as a fluorescence probe. Biosens Bioelectron 71:470–475
Shaswat B, Satyabrat G, Raju K (2018) Fluorescence biosensor based on gold-carbon dot probe for efficient detection of cholesterol. Synth Met 244:92–98
Qiao C, Yu H, Yi G, Zhou J, Song G (2018) Ultrasensitive detection of heparin by exploiting the silver nanoparticle-enhanced fluorescence of graphitic carbon nitride (g-C3N4) quantum dots. Microchim Acta 185:332
Han S, Chang T, Zhao H, Du H, Liu S, Wu B, Qin S (2017) Cultivating fluorescent flowers with highly luminescent carbon dots fabricated by a double passivation method. Nanomaterials 7:176
Wang W, Zhao Y, ** Y (2013) Gold-nanorod-based colorimetric and fluorescent approach for sensitive and specific assay of disease-related gene and mutation. ACS Appl Mater Inter 5:11741–11746
Wang Z, Zong S, Wang L, Wang C, Xu S, Chen H, Cui Y (2012) SERS-fluorescence joint spectral encoding using organic-metal-QD hybrid nanoparticles with a huge encoding capacity for high-throughput biodetection: putting theory into practice. J Am Chem Soc 134:2993–3000
Liang W, Liu Z, Liu S, Yang J, He Y (2014) A novel surface modification strategy of CdTe/CdS QDs and its application for sensitive detection of ct-DNA. Sensor Actuat B 196:336–344
Wang J, Liu Z, Liu J, Liu S, Shen W (2008) Study on the interaction between fluoroquinolones and erythrosine by absorption, fluorescence and resonance Rayleigh scattering spectra and their application. Spectrochim Acta A 69:956–963
Moriarty MM, Koch I, Gordon RA, Reimer KJ (2009) Arsenic speciation of terrestrial invertebrates. Environ Sci Technol 43:4818–4823
Fendorf S, Michael HA, Van GA (2010) Spatial and temporal variations of groundwater arsenic in South and Southeast Asia. Science 328:1123–1127
Gutu CM, Preoteasa V, Olaru OT, Purdel CN, Ilie M (2014) Atomic absorption spectrometry and ICP-MS methods used to evaluate the arsenic intake in a toxicity assay on daphnia magna. Toxicol Lett 229:S110
Chen G, Chen T (2014) SPE speciation of inorganic arsenic in rice followed by hydride-generation atomic fluorescence spectrometric quantification. Talanta 119:202–206
Komorowicz I, Barałkiewicz D (2013) Arsenic speciation in water by high-performance liquid chromatography/inductively coupled plasma mass spectrometry-method validation and uncertainty estimation. Rapid Commun Mass Spectrom 28:159–168
Campillo N, Peñalver R, Viñas P (2009) Speciation of arsenic using capillary gas chromatography with atomic emission detection. Talanta 77:793–79914
Tang M, Wen G, Liang A, Jiang Z (2015) A simple and sensitive resonance Rayleigh scattering method for determination of As(III) using aptamer-modified nanogold as a probe. Luminescence 29:603–608
Tian H, Zhuang G, Ma A, **g C (2012) Arsenic interception by cell wall of bacteria observed with surface-enhanced Raman scattering. J Microbiol Methods 89:153–158
Pan J, Zhou D, Chen J (2018) Ultrasensitive aptamer biosensor for arsenic (III) detection based on label-free triple-helix molecular switch and fluorescence sensing platform. Talanta 189:370–376
Li G, Liu Y, Wang Y, Liao L (2009) The molecular resonance fluorescence method for determination of arsenic in hair samples. Spectrochim Acta A 72:811–815
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 21667006, 21767004).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The author(s) declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOC 46.8 mb)
Rights and permissions
About this article
Cite this article
Zhang, Z., Li, J., Wang, X. et al. Aptamer-mediated N/Ce-doped carbon dots as a fluorescent and resonance Rayleigh scattering dual mode probe for arsenic(III). Microchim Acta 186, 638 (2019). https://doi.org/10.1007/s00604-019-3764-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-019-3764-3