Abstract
A straightforward method has been developed for selective separation of chloramphenicol (CAP) from marine sediment samples. Magnetic molecularly imprinted nanoparticles (NPs) of type Fe3O4@SiO2 were prepared via surface imprinting with CAP. The NPs were characterized by Fourier transform infrared spectroscopy, transmission electron microscopy and thermogravimetric analysis. They have perfect core-shell structure, excellent thermal stability, high affinity and selectivity to CAP. The imprinting factor and Scatchard analysis also reveal good specific recognition to the template. The imprinted NPs were applied as sorbents for fast and selective extraction of CAP from marine sediment samples. The experimental parameters affecting separation efficiency were optimized. Three marine sediment samples were analyzed. Following desorption with methanol/water (90/10,v/v), CAP was quantified by HPLC with DAD detection. The limit of detection is 0.1 μg L−1 with a good linear response between 0.1–20 mg L−1 of CAP concentration (R2 = 0.999, n = 3). The method exhibits satisfactory recoveries from spiked samples (77.9–102.5%) and has low relative standard deviations (<6.3%). The magnetic material can be used at least 5 times by the regeneration without any loss of selectivity and adsorption capability.

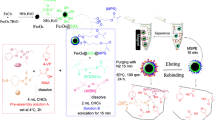
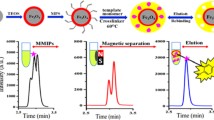
Schematic presentation of magnetic molecularly imprinted nanoparticles (MMIPs) as sorbent for fast extraction and chromatographic analysis of chloramphenicol (CAP) from marine sediments. CAP-MMIPs are synthesized by surface imprinting method using 3-methacryloxy propyl trimethoxy silane (MPS) as the silane coupling agent.





Similar content being viewed by others
References
Chu W, Krasner SW, Gao N, Templeton MR, Yin D (2016) Contribution of the antibiotic chloramphenicol and its analogues as precursors of dichloroacetamide and other disinfection by products in drinking water. Environ Sci Technol 50:388–396
Tomassetti M, Angeloni R, Martini E, Castrucci M, Campanella L (2018) Enzymatic DMFC device used for direct analysis of chloramphenicol and a comparison with the competitive immunosensor method. Sens Actuators B Chem 255:1545–1552
Meenakshi S, Sophia SJ, Pandian K (2018) High surface graphene nanoflakes as sensitive sensing platform for simultaneous electrochemical detection of metronidazole and chloramphenicol. Mater Sci Eng C 90:407–419
Pan XD, Wu PG, Jiang W, Ma B (2015) Determination of chloramphenicol, thiamphenicol, and florfenicol in fish muscle by matrix solid-phase dispersion extraction (MSPD) and ultra-high pressure liquid chromatography tandem mass spectrometry. Food Control 52:34–38
Di X, Wang X, Liu Y, Guo X, **n D (2019) Solid-phase extraction coupled with switchable hydrophilicity solvent-based homogeneous liquid-liquid microextraction for chloramphenicol enrichment in environmental water samples: a novel alternative to classical extraction techniques. Anal Bioanal Chem 411:803–812
Javidi M, Housaindokht MR, Verdian A, Razavizadeh BM (2018) Detection of chloramphenicol using a novel apta-sensing platform based on aptamer terminal-lock in milk samples. Anal Chim Acta 1039:116–123
Armenta S, de la Guardia M, Abad-Fuentes A, Abad-Somovilla A, Esteve-Turrillas FA (2016) Highly selective solid-phase extraction sorbents for chloramphenicol determination in food and urine by ion mobility spectrometry. Anal Bioanal Chem 408:8559–8567
Yu X, He Y, Jiang J, Cui H (2014) A competitive immunoassay for sensitive detection of small molecules chloramphenicol based on luminol functionalized silver nanoprobe. Anal Chim Acta 812:236–242
Mamani MCV, Reyes FGR, Rath S (2009) Multiresidue determination of tetracyclines, sulphonamides and chloramphenicol in bovine milk using HPLC-DAD. Food Chem 117:545–552
Kikuchi H, Sakai T, Teshima R, Nemoto S, Akiyama H (2017) Total determination of chloramphenicol residues in foods by liquid chromatography-tandem mass spectrometry. Food Chem 230:589–593
Liu T, **e J, Zhao J, Song G, Hu Y (2014) Magnetic chitosan nanocomposite used as cleanup material to detect chloramphenicol in milk by GC-MS. Food Anal Methods 7:814–819
Khoshbin Z, Verdian A, Housaindokht MR, Izadyar M, Rouhbakhsh Z (2018) Aptasensors as the future of antibiotics test kits-a case study of the aptamer application in the chloramphenicol detection. Biosens Bioelectron 122:263–283
Lian ZR, Li HB, Wang JT (2016) Experimental and computational studies on molecularly imprinted solid-phase extraction for gonyautoxins 2,3 from dinoflagellate Alexandrium minutum. Anal Bioanal Chem 408:5527–5535
Pisarev OA, Polyakova IV (2018) Molecularly imprinted polymers based on methacrylic acid and ethyleneglycol dimethacrylate for L-lysine recognition. React Funct Polym 130:98–110
Ostovan A, Ghaedi M, Arabi M, Yang Q, Li J, Chen L (2018) Hydrophilic multitemplate molecularly imprinted biopolymers based on a green synthesis strategy for determination of B-family vitamins. ACS Appl Mater Inter 10:4140–4150
Chen D, Han X, Wang Q, **e L, Ai Y, Dang X, Huang J, Chen H (2017) Magnetic molecularly imprinted polymers for selective extraction and determination of naphthols. Microchim Acta 184:3373–3379
Zhang Z, Li J, Fu L, Liu D, Chen L (2015) Magnetic molecularly imprinted microsensor for selective recognition and transport of fluorescent phycocyanin in seawater. J Mater Chem A 3:7437–7444
Bagheri AR, Arabi M, Ghaedi M, Ostovan A, Wang X, Li J, Chen L (2019) Dummy molecularly imprinted polymers based on a green synthesis strategy for magnetic solid-phase extraction of acrylamide in food samples. Talanta 195:390–400
Chen L, Wang X, Lu W, Wu X, Li J (2016) Molecular imprinting: perspectives and applications. Chem Soc Rev 45:2137–2211
Azodi-Deilami S, Najafabadi AH, Asadi E, Abdouss M, Kordestani D (2014) Magnetic molecularly imprinted polymer nanoparticles for the solid-phase extraction of paracetamol from plasma samples, followed its determination by HPLC. Microchim Acta 181:1823–1832
Dai J, He J, **e A, Gao L, Pan J, Chen X, Zhou Z, Wei X, Yan Y (2016) Novel pitaya-inspired well-defined core-shell nanospheres with ultrathin surface imprinted nanofilm from magnetic mesoporous nanosilica for highly efficient chloramphenicol removal. Chem Eng J 284:812–822
Chen L, Li B (2013) Magnetic molecularly imprinted polymer extraction of chloramphenicol from honey. Food Chem 141:23–28
Wei S, Li J, Liu Y, Ma J (2016) Development of magnetic molecularly imprinted polymers with double templates for the rapid and selective determination of amphenicol antibiotics in water, blood, and egg samples. J Chromatogr A 1473:19–27
Lian Z, Wang J (2018) Selective detection of chloramphenicol based on molecularly imprinted solid-phase extraction in seawater from Jiaozhou Bay, China. Mar Pollut Bull 133:750–755
Fan L, Luo C, Lv F, Lu F, Qiu H (2011) Removal of ag+ from water environment using a novel magnetic thiourea-chitosan imprinted ag+. J Hazard Mater 194:193–201
Sorribes-Soriano A, Esteve-Turrillas FA, Armenta S, de la Guardia M, Herrero-Martínez JM (2017) Cocaine abuse determination by ion mobility spectrometry using molecular imprinting. J Chromatogr A 1481:23–30
Chen FF, **e XY, Shi YP (2013) Magnetic molecularly imprinted polymer for the selective extraction of sildenafil, vardenafil and their analogs from herbal medicines. Talanta 115:482–489
Zhou Y, Zhou T, ** H, **g T, Song B, Zhou Y, Mei S, Lee YI (2015) Rapid and selective extraction of multiple macrolide antibiotics in foodstuff samples based on magnetic molecularly imprinted polymers. Talanta 137:1–10
Attallah OA, Al-Ghobashy MA, Ayoub AT, Nebsen M (2018) Magnetic molecularly imprinted polymer nanoparticles for simultaneous extraction and determination of 6-mercaptopurine and its active metabolite thioguanine in human plasma. J Chromatogr A 1561:28–38
Tang W, Li G, Row KH, Zhu T (2016) Preparation of hybrid molecularly imprinted polymer with double- templates for rapid simultaneous purification of theophylline and chlorogenic acid in green tea. Talanta 152:1–8
Samanidou V, Kehagia M, Kabir A, Furton GK (2016) Matrix molecularly imprinted mesoporous solegel sorbent for efficient solid-phase extraction of chloramphenicol from milk. Anal Chim Acta 914:62–74
Rejtharová M, Rejthar L (2009) Determination of chloramphenicol in urine, feed water, milk and honey samples using molecular imprinted polymer clean-up. J Chromatogr A 1216:8246–8253
Shi X, Wu A, Zheng S, Li R, Zhang D (2007) Molecularly imprinted polymer microspheres for solid-phase extraction of chloramphenicol residues in foods. J Chromatogr B 850:24–30
Acknowledgements
This project was supported by the National Key Research and Development Program of China (Grant No. 2018YFD0900704), the National Natural Science Foundation of China (Grant No.41506092) and the Joint Funds of the National Natural Science Foundation of China (Grant No. U1806202).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOC 1595 kb)
Rights and permissions
About this article
Cite this article
Qin, D., Wang, J., Ge, C. et al. Fast extraction of chloramphenicol from marine sediments by using magnetic molecularly imprinted nanoparticles. Microchim Acta 186, 428 (2019). https://doi.org/10.1007/s00604-019-3548-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-019-3548-9