Abstract
The authors describe an ultrasensitive amperometric enzymatic assay for uranyl ion. It is based on the use of mesoporous silica nanoparticles (mesoSiNPs) loaded with Methylene Blue (MB) and functionalized with an UO2(II)-dependent DNAzyme. The electroactive label MB was sealed in the inner pores of the mesoSiNPs along with double stranded DNA (containing the DNAzyme and the substrate strand). In the presence of UO2(II), the DNAzyme is actived to cleave the substrate strands. This leads to the cleavage of the caps and the release of MB from the mesoSiNPs. The amount of released MB depends on the concentration of UO2(II) and can be determined amperometrically, best at a working voltage of −0.25 V (vs SCE), by using a chitosan coated carbon paste electrode. Response is linear in the 20 pM to 0.1 nM UO2(II) concentration range, and the detection limit is as low as 0.15 pM. Recoveries from spiked samples varied from 91.3 to 99.4%. The assay is highly specific, selective, and not interfered by other metal ions.

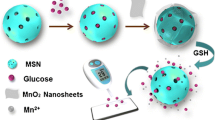
(a) Schematic representation of the synthesis, MB loading, and dsDNA Binding of the mesoSiNPs, as well as the release of MB from mesoSiNPs in the presence of UO2 2+. (b) Schematic illustration of the stepwise DNAzyme-based electrochemical sensor for uranium detection.








Similar content being viewed by others
References
Liu JW, Brown AK, Meng X, Cropek DM, Istok JD, Watson DB, Lu Y (2007) A catalytic beacon sensor for uranium with parts-per-trillion sensitivity and millionfold selectivity. P Natl Acad Sci 104:2056–2061. doi:10.1073/pnas.0607875104
Gongalsky KB (2003) Impact of pollution caused by uranium production on soil macrofauna. Environ Monit Asses 89:197–219. doi:10.1023/A:1026031224658
Villa M, Manjon G, Hurtado S, García-Tenorio R (2011) Uranium pollution inan estuary affected by pyrite acid mine drainage and releases of naturally occurring radioactive materials. Mar Pollut Bull 62:1521–1529. doi:10.1016/j.marpolbul.2011.04.003
Alam MN, Rahman N, Azmi SNH (2008) Optimized and validated spectrophotometric method for the determination of uranium(VI) via complexation with meloxicam. J Hazard Mater 155:261–268. doi:10.1016/j.jhazmat.2007.11.055
**ao G, Jones RL, Saunders D, Caldwell KL (2014) Determination of 234U/238U, 235U/238U and 236U/238U isotope ratios in urine using sector field inductively coupled plasma mass spectrometry. Radiat Prot Dosim 1:1–7. doi:10.1093/rpd/ncu023
Santos JS, Teixeira Leonardo SG, Dos Santos Walter NL, Lemos VA, Godoy JM, Ferreira Sérgio LC (2010) Uranium determination using atomic spectrometric techniques:an overview. Anal Chim Acta 674:143–156. doi:10.1016/j.aca.2010.06.010
Michon J, Frelon S, Garnier C, Coppin F (2010) Determinations of uranium(VI) binding properties with some metalloproteins (transferrin, albumin, metallothionein and ferritin) by flluorescence quenching. J Fluoresc 20:581–590. doi:10.1007/s10895-009-0587-3
Dutta RK, Kumar A (2016) Highly sensitive and selective method for detecting ultratrace levels of aqueous uranyl ions by strongly photoluminescent-responsive amine-modified cadmium sulfide quantum dots. Anal Chem 88:9071–9078. doi:10.1021/acs.analchem. 6b01943
Zhang D, Chen Z, Omar H, Deng L, Khashab NM (2015) Colorimetric peroxidase mimetic assay for uranyl detection in sea water. ACS Appl Mater Interfaces 7:4589–4594. doi:10.1021/am507361x
Breaker RR, Joyce GF (1994) A DNA enzyme that cleaves RNA. Chem Biol 1:223–229. doi:10.1016/1074-5521(94)90014-0
Hu KC, Lan DX, Li XM, Zhang SS (2008) Electrochemical DNA biosensor based on nanoporous gold electrode and multifunctional encoded DNA-Au bio bar codes. Anal Chem 80:9124–9130. doi:10.1021/ac8017197
Viswanathan S, Radecka H, Radecki J (2009) Electrochemical biosensor for pesticides based on acetylcholinesterase immobilized on polyaniline deposited on vertically assembled carbon nanotubes wrapped with ssDNA. Biosens Bioelectron 24:2772–2777. doi:10.1016/j.bios.2009.01.044
Zhang J, Shi PW, Yan PP, Wang MB, Tang QH, Cai FD, Deng AP, Li JG (2015) Quantum dots based electrochemiluminescent immunosensor for ultrasensitive and specific determination of mercury (II) ions using gold nanoparticles and a monoclonal antibody. J Electrochem Soc 162:B22–B26. doi:10.1149/2.0631501jes
Lee JH, Wang Z, Liu J, Lu Y (2008) Highly sensitive and selective colorimetric sensors for uranyl (UO2 2+):development and comparison of labeled and label-free DNAzyme -gold nanoparticle systems. J Am Chem Soc 130:14217–14226. doi:10.1021/ja803607z
Zhang HY, Ruan YJ, Lin L, Lin M, Zeng XX, Zeng X, Fu FF (2015) A turn-off fluorescent biosensor for the rapid and sensitive detection of uranyl ion based on molybdenum disulfide nanosheets and specific DNAzyme. Spectrochim Acta A 146:1–6. doi:10.1016/j.saa.2015.02.113
Baker SE, Cai W, Lasseter TL, Weidkamp KP, Hamers RJ (2002) Covalently bonded adducts of deoxyribonucleic acid (DNA) oligonucleotides with single-wall carbon nanotubes:synthesis and hybridization. Nano Lett 2:1413–1417. doi:10.1021/nl025729f
Zhang GY, Deng SY, Cai WR, Cosnier S, Zhang XJ, Shan D (2015) Magnetic zirconium hexacyanoferrate (II) nanoparticle as tracing tag for electrochemical DNA assay. Anal Chem 87:9093–9100. doi:10.1021/acs.analchem.5b02395
Ma DD, Yuan YL, **ao XL, Gao YY, Li YH, Xu WH, Long W (2014) A label-free electrochemical biosensor for trace uranium based on DNAzymes and gold nanoparticles. J Radioanal Nucl Chem 299:1911–1919. doi:10.1007/s10967-013-2897-9
Tang Q, Yuan YL, **ao XL, Guo P, Hu JB, Ma DD, Gao YY (2013) DNAzyme based electrochemical sensors for trace uranium. Microchim Acta 180:1059–1064. doi:10.1007/s00604-013-1021-8
Ren K, Wu J, Zhang Y, Yan F, Ju HX (2014) Proximity hybridization regulated DNA biogate for sensitive electrochemical immunoassay. Anal Chem 86:7494–7499. doi:10.1021/ac5012377
Qin QD, Ma J, Liu K (2009) Adsorption of anionic dyes on ammonium-functionalized MCM-41. J Hazard Mater 162:133–139. doi:10.1016/j.jhazmat.2008.05.016
Shao Y, Wang X, Kang Y, Shu Y, Sun Q, Li L (2014) Application of Mn/MCM-41 as an adsorbent to remove methyl blue from aqueous solution. J Colloid Interface Sci 429:25–33. doi:10.1016/j.jcis.2014.05.004
**ao N, Deng J, Cheng JL, Ju SQ, Zhao HQ, **e J, Qian D (2016) Carbon paste electrode modified with duplex molecularly imprinted polymer hybrid film for metronidazole detection. Biosens Bioelectron 81:54–60. doi:10.1016/j.bios.2016.02.041
van der Maaden K, Sliedregt K, Kros A, Jiskoot W, Bouwstra J (2012) Fluorescent nanoparticle adhesion assay:a novel method for surface pKa determination of self-assembled monolayers on silicon surfaces. Langmuir 28:3403–3411. doi:10.1021/la203560k
Vashist SK, Lam E, Hrapovic S, Male KB, Luong John HT (2014) Immobilization of antibodies and enzymes on 3-aminopropyltriethoxysilane-functionalized bioanalytical platforms for biosensors and diagnostics. Chem Rev 114:11083–11130. doi:10.1021/cr5000943
Gonza’lez AG, Herrador MA (2007) A practical guide to analytical method validation, including measurement uncertainty and accuracy profiles. Trac-Trend Anal Chem 26:227–238. doi:10.1016/j.trac.2007.01.009
Yun W, Cai D, Jiang J, Wang X, Liao JS, Zhang PC, Sang G (2016) An ultrasensitive electrochemical biosensor for uranyl detection based on DNAzyme and target-catalyzed hairpin assembly. Microchim Acta 183(4):1425–1432. doi:10.1007/s00604-016-1778-7
Zhou B, Wang YS, Yang HX, Xue JH, Wang JC, Liu SD, Zhao H (2014) A sensitive resonance light scattering assay for uranyl ion based on the conformational change of a nuclease-resistant aptamer and gold nanoparticles acting as signal reporters. Microchim Acta 181(11–12):1353–1360. doi:10.1007/s00604-014-1267-9
Acknowledgements
This research was supported by the National Natural Science Foundation of China (11405081), the Hunan Provincial Natural Science Foundation of China (2017JJ3276) and the Department of Education of Hunan Province (17B226).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOC 151 kb)
Rights and permissions
About this article
Cite this article
Wen, Y., Yuan, Y., Li, L. et al. Ultrasensitive DNAzyme based amperometric determination of uranyl ion using mesoporous silica nanoparticles loaded with Methylene Blue. Microchim Acta 184, 3909–3917 (2017). https://doi.org/10.1007/s00604-017-2397-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-017-2397-7