Abstract
Purpose
Safety concerns regarding the application of bone morphogenetic proteins (BMPs) have been highlighted in recent years. It is noted that both BMP and their receptors being identified as a trigger for cancer growth. Here, we aimed to determine the safety and efficacy of BMP for spinal fusion surgery.
Methods
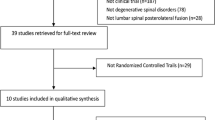
We conducted this systematic review on topics of spinal fusion surgery with rhBMP application from three database (PubMed, EuropePMC, and Clinicaltrials.gov) with MeSH phrases such as “rh-BMP,” ”rhBMP,” “spine surgery,” “spinal arthrodesis,” and “spinal fusion” were searched (using the Boolean operators “and” and “or”). Our research includes all articles, as long as published in English language. In the face of disagreement between the two reviewers, we discussed it together until all authors reached a consensus. The primary key outcome of our study is the incidence of cancer following rhBMP implantation.
Results
Our study included a total of 8 unique studies (n = 37,682). The mean follow-up varies among all studies, with the longest follow-up is 66 months. Our meta-analysis showed that exposure to rhBMP in spinal surgery did increase the risk of cancers (RR 1.85, 95%CI [1.05, 3.24], p = 0.03).
Conclusions
Our study found that rhBMP was not associated with the increased risk of cancer incidence within the rhBMP cohort. Still, we did face several limitations, in which further studies are needed to confirm the result of our meta-analysis.




Similar content being viewed by others
Data availability
All data will be given upon reasonable requests by contacting the corresponding author of the current study.
Abbreviations
- rhBMP:
-
Recombinant human bone morphogenetic protein
- OP-1:
-
Osteogenic protein 1
- FDA:
-
US food and drug administration
- PRISMA:
-
Preferred reporting items for systematic review and meta-analysis
- NOS:
-
Newcastle–Ottawa Scale
- RoB:
-
Risk of bias
- RR:
-
Risk ratio
- CI:
-
Confidence interval
References
Bannwarth M, Smith JS, Bess S et al (2021) Use of rhBMP-2 for adult spinal deformity surgery: patterns of usage and changes over the past decade. Neurosurg Focus 50:E4. https://doi.org/10.3171/2021.3.FOCUS2164
Jain A, Yeramaneni S, Kebaish KM et al (2020) Cost-utility analysis of rhBMP-2 use in adult spinal deformity surgery. Spine (Phila Pa 1976) 45:1009–1015. https://doi.org/10.1097/BRS.0000000000003442
Golubovsky JL, Ejikeme T, Winkelman R, Steinmetz MP (2021) Osteobiologics. Oper Neurosurg (Hagerstown, Md) 21:S2–S9. https://doi.org/10.1093/ons/opaa383
Ye F, Zeng Z, Wang J et al (2018) Comparison of the use of rhBMP-7 versus iliac crest autograft in single-level lumbar fusion: a meta-analysis of randomized controlled trials. J Bone Miner Metab 36:119–127. https://doi.org/10.1007/s00774-017-0821-z
Poynton AR, Lane JM (2002) Safety profile for the clinical use of bone morphogenetic proteins in the spine. Spine (Phila Pa 1976) 27:S40–S48. https://doi.org/10.1097/00007632-200208151-00010
Carragee EJ, Hurwitz EL, Weiner BK (2011) A critical review of recombinant human bone morphogenetic protein-2 trials in spinal surgery: emerging safety concerns and lessons learned. Spine J 11:471–491. https://doi.org/10.1016/j.spinee.2011.04.023
Glassman SD, Dimar JR, Burkus K et al (2007) The efficacy of rhBMP-2 for posterolateral lumbar fusion in smokers. Spine (Phila Pa 1976) 32:1693–1698. https://doi.org/10.1097/BRS.0b013e318074c366
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Int J Surg. https://doi.org/10.1136/bmj.n71
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:1–9. https://doi.org/10.1136/BMJ.N71
Wells GA, Shea B, O’Connell D et al (2021) The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed 6 Jan 2022
Sterne JAC, Savović J, Page MJ et al (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. https://doi.org/10.1136/bmj.l4898
Vaccaro AR, Anderson DG, Patel T et al (2005) Comparison of OP-1 Putty (rhBMP-7) to iliac crest autograft for posterolateral lumbar arthrodesis: a minimum 2-year follow-up pilot study. Spine (Phila Pa 1976) 30:2709–2716. https://doi.org/10.1097/01.brs.0000190812.08447.ba
Delawi D, Dhert WJA, Rillardon L et al (2010) A prospective, randomized, controlled, multicenter study of osteogenic protein-1 in instrumented posterolateral fusions: report on safety and feasibility. Spine (Phila Pa 1976) 35:1185–1191. https://doi.org/10.1097/BRS.0b013e3181d3cf28
Epstein NE (2014) Basic science and spine literature document bone morphogenetic protein increases cancer risk. Surg Neurol Int 5:S552–S560. https://doi.org/10.4103/2152-7806.148039
Latzman JM, Kong L, Liu C, Samadani U (2010) Administration of human recombinant bone morphogenetic protein-2 for spine fusion may be associated with transient postoperative renal insufficiency. Spine (Phila Pa 1976) 35:231–237. https://doi.org/10.1097/BRS.0b013e3181c71447
Baldus C, Kelly MP, Yanik EL et al (2017) Incidence of cancer in spinal deformity patients receiving high-dose (≥40 mg) bone morphogenetic protein (rhBMP-2). SpineSpine (Phila Pa 1976) 42:1785–1791. https://doi.org/10.1097/BRS.0000000000002232
Carragee EJ, Chu G, Rohatgi R et al (2013) Cancer risk after use of recombinant bone morphogenetic protein-2 for spinal arthrodesis. J Bone Joint Surg Am 95:1537–1545. https://doi.org/10.2106/JBJS.L.01483
Lad SP, Bagley JH, Karikari IO et al (2013) Cancer after spinal fusion: the role of bone morphogenetic protein. Neurosurgery 73:440–449. https://doi.org/10.1227/NEU.0000000000000018
Cooper GS, Kou TD (2018) Risk of cancer following lumbar fusion surgery with recombinant human bone morphogenic protein-2 (rhBMP-2): an analysis using a commercially insured patient population. Int J spine Surg 12:260–268. https://doi.org/10.14444/50323
Khan TR, Pearce KR, McAnany SJ et al (2018) Comparison of transforaminal lumbar interbody fusion outcomes in patients receiving rhBMP-2 versus autograft. Spine J 18:439–446. https://doi.org/10.1016/j.spinee.2017.08.230
Dettori JR, Chapman JR, DeVine JG et al (2019) Longer follow-up continues to reveal no increased risk of cancer with the use of recombinant human bone morphogenetic protein in spine fusion. Spine J 19:1640–1647. https://doi.org/10.1016/j.spinee.2019.05.005
Avila MJ, Walter CM, Skoch J et al (2015) Fusion after intradural spine tumor resection in adults: a review of evidence and practices. Clin Neurol Neurosurg 138:169–173. https://doi.org/10.1016/j.clineuro.2015.08.020
Cai Z, Zhao Y, Tang X et al (2022) Factors associated with spinal fixation mechanical failure after tumor resection: a systematic review and meta-analysis. J Orthop Surg Res 17:110. https://doi.org/10.1186/s13018-022-03007-6
Noh SH, Takahashi T, Inoue T et al (2022) Postoperative spinal deformity and instability after cervical spinal cord tumor resection in adults: a systematic review and meta-analysis. J Clin Neurosci 100:148–154. https://doi.org/10.1016/j.jocn.2022.04.005
Yee TJ, Saadeh YS, Strong MJ et al (2021) Survival, fusion, and hardware failure after surgery for spinal metastatic disease. J Neurosurg Spine 34:665–672. https://doi.org/10.3171/2020.8.SPINE201166
Guo Y, Jia X, Cui Y et al (2021) Sirt3-mediated mitophagy regulates AGEs-induced BMSCs senescence and senile osteoporosis. Redox Biol 41:101915. https://doi.org/10.1016/j.redox.2021.101915
Abe T, Miyazaki M, Ishihara T et al (2021) Optimal intermittent administration interval of parathyroid hormone 1–34 for bone morphogenetic protein-induced bone formation in a rat spinal fusion model. JOR spine 4:e1168. https://doi.org/10.1002/jsp2.1168
Fu R, Selph S, McDonagh M et al (2013) Effectiveness and harms of recombinant human bone morphogenetic protein-2 in spine fusion: a systematic review and meta-analysis. Ann Intern Med 158:890–902. https://doi.org/10.7326/0003-4819-158-12-201306180-00006
Cicciù M, Fiorillo L, Cervino G, Habal MB (2021) Bone morphogenetic protein application as grafting materials for bone regeneration in craniofacial surgery: current application and future directions. J Craniofac Surg 32:787–793. https://doi.org/10.1097/SCS.0000000000006937
Leal CR, de Carvalho RM, Ozawa TO et al (2019) Outcomes of alveolar graft with Rhbmp-2 in CLP: influence of cleft type and width, canine eruption, and surgeon. Cleft Palate Craniofac J 56:383–389. https://doi.org/10.1177/1055665618780981
Ramly EP, Alfonso AR, Kantar RS et al (2019) Safety and efficacy of recombinant human bone morphogenetic protein-2 (rhBMP-2) in craniofacial surgery. Plast Reconstr surgery Glob open 7:e2347. https://doi.org/10.1097/GOX.0000000000002347
Cho JH, Lee JH, Lee KM et al (2022) BMP-2 induced signaling pathways and phenotypes: comparisons between senescent and non-senescent bone marrow mesenchymal stem cells. Calcif Tissue Int 110:489–503. https://doi.org/10.1007/s00223-021-00923-3
Halloran D, Durbano HW, Nohe A (2020) Bone morphogenetic protein-2 in development and bone homeostasis. J Dev Biol. https://doi.org/10.3390/jdb8030019
Vavken J, Mameghani A, Vavken P, Schaeren S (2016) Complications and cancer rates in spine fusion with recombinant human bone morphogenetic protein-2 (rhBMP-2). Eur Spine J 25:3979–3989. https://doi.org/10.1007/s00586-015-3870-9
Malham GM, Louie PK, Brazenor GA et al (2022) Recombinant human bone morphogenetic protein-2 in spine surgery: recommendations for use and alternative bone substitutes-a narrative review. J spine Surg (Hong Kong) 8:477–490. https://doi.org/10.21037/jss-22-23
Hajibeigi A, Nasr K, Udayakumar D et al (2018) Breast tumor microcalcification induced by bone morphogenetic protein-2: a new murine model for human breast tumor diagnosis. Contrast Media Mol Imaging 2018:2082154. https://doi.org/10.1155/2018/2082154
Huang P, Chen A, He W et al (2017) BMP-2 induces EMT and breast cancer stemness through Rb and CD44. Cell death Discov 3:17039. https://doi.org/10.1038/cddiscovery.2017.39
Aluganti Narasimhulu C, Singla DK (2020) The role of bone morphogenetic protein 7 (BMP-7) in inflammation in heart diseases. Cells. https://doi.org/10.3390/cells9020280
Grey ZJ, Howie RN, Durham EL et al (2019) Sub-clinical dose of bone morphogenetic protein-2 does not precipitate rampant, sustained inflammatory response in bone wound healing. Wound Repair Regen 27:335–344. https://doi.org/10.1111/wrr.12710
Acknowledgement
None.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors report no conflicting interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wijaya, J.H., Tjahyanto, T., Alexi, R. et al. Application of rhBMP in spinal fusion surgery: any correlation of cancer incidence? A systematic review and meta-analysis. Eur Spine J 32, 2020–2028 (2023). https://doi.org/10.1007/s00586-023-07730-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-023-07730-4