Abstract
Key message
Rhizophora mangle plants tolerate high salinity under field conditions by increasing the transcription of genes involved in photosynthesis, antioxidant system, and salt transport.
Abstract
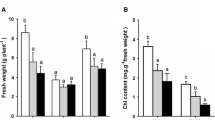
Rhizophora mangle is a mangrove species adapted to a wide range of salinity and has great ecological importance for mangrove ecosystems. Nevertheless, little is known about the genetic basis underlying the salt-tolerance mechanisms of this species. Here, we investigated the physiological and molecular basis of the salt tolerance in R. mangle under field conditions based on analyses of gas exchange, chlorophyll contents, chlorophyll a fluorescence, and gene expression by qRT-PCR. The gas exchange data showed that the plants growing on both mangrove sites, of low and high salinity, present comparable CO2 assimilation rates and stomatal conductance. Our data suggest that the photosynthetic maintenance under high-salinity conditions was supported by an improved PSII activity, as indicated by chlorophyll a fluorescence parameters and chlorophyll a/b ratio, including the increase of active reaction centers (RCs) and stable oxygen-evolving complexes (OECs). Additionally, the psbA (D1 protein of the RC) and PSBO2 (subunit of the OEC) genes were up-regulated under high salinity, which may be related to increased efficiency to repair injuries in the RC and OEC through the synthesis of new subunits, improving the PSII activity. Likewise, the expression of genes involved in ATP synthesis, RubisCO activation, ROS scavenging, GABA synthesis, and vacuolar Na+ sequestration was up-regulated under high salinity. Thereby, the energy balance and the avoidance of oxidative stress and ion toxicity are also important means by which R. mangle deals with increasing salt levels in natural environments. Taken together, our data shed light on the salt-tolerance mechanisms of R. mangle in its natural ecosystem, including the findings of some possible salt-regulated genes.






Similar content being viewed by others
References
Allen JF, Pfannschmidt T (2000) Balancing the two photosystems: photosynthetic electron transfer governs transcription of reaction centre genes in chloroplasts. Philos Trans R Soc Lond B Biol Sci 355:1351–1359. https://doi.org/10.1098/rstb.2000.0697
Al-Quraan NA, Sartawe FAB, Qaryouti MM (2013) Characterization of γ-aminobutyric acid metabolism and oxidative damage in wheat (Triticum aestivum L.) seedlings under salt and osmotic stress. J Plant Physiol 170:1003–1009. https://doi.org/10.1016/j.jplph.2013.02.010
Baker NR, Oxborough K (2004) Chlorophyll fluorescence as a probe of photosynthetic productivity. In: Papageorgiou GC, Govindjee (eds) Chlorophyll a fluorescence advances in photosynthesis and respiration. Springer, Dordrecht, pp 65–82. https://doi.org/10.1007/978-1-4020-3218-9_3
Ball MC (1988) Ecophysiology of mangroves. Trees 2:129–142. https://doi.org/10.1007/BF00196018
Banjara M, Zhu L, Shen G, Payton P, Zhang H (2012) Expression of an Arabidopsis sodium/proton antiporter gene (AtNHX1) in peanut to improve salt tolerance. Plant Biotechnol Rep 6:59–67. https://doi.org/10.1007/s11816-011-0200-5
Barbier EB (2016) The protective service of mangrove ecosystems: a review of valuation methods. Mar Pollut Bull 109:676–681. https://doi.org/10.1016/j.marpolbul.2016.01.033
Bernini E, Silva MA, Carmo TMS, Cuzzuol GRF (2006) Composição de folhas das espécies do manguezal do estuário do rio São Mateus, Espírito Santo. Brasil Rev Bras Bot 29(4):689–699. https://doi.org/10.1590/S0100-84042006000400018
Bhat JY, Thieulin-Pardo G, Hartl FU, Hayer-Hartl M (2017) Rubisco activases: AAA+ chaperones adapted to enzyme repair. Front Mol Biosci 4:20. https://doi.org/10.3389/fmolb.2017.00020
Bompy F, Lequeue G, Imbert D, Dulormne M (2014) Increasing fluctuations of soil salinity affect seedling growth performances and physiology in three Neotropical mangrove species. Plant Soil 380:399–413. https://doi.org/10.1007/s11104-014-2100-2
Cascio C, Schaub M, Novak K, Desotgiu R, Bussotti F, Strasser RJ (2010) Foliar responses to ozone of Fagus sylvatica L. seedlings grown in shaded and in full sunlight conditions. Environ Exp Bot 68:188–197. https://doi.org/10.1016/j.envexpbot.2009.10.003
Chen S, Strasser RJ, Qiang S (2014) In vivo assessment of effect of phytotoxin tenuazonic acid on PSII reaction centers. Plant Physiol Biochem 84:10–21. https://doi.org/10.1016/j.plaphy.2014.09.004
Chen Y, Ye (2014) Effects of salinity and nutrient addition on mangrove Excoecaria agallocha. PLoS ONE 9(4):e93337. https://doi.org/10.1371/journal.pone.0093337
Cintrón G, Lugo AE, Pool DJ, Morris G (1978) Mangroves of arid environments in Puerto Rico and adjacent islands. Biotrop 10(2):110–121
Dangremond EM, Feller IC, Sousa WP (2015) Environmental tolerances of rare and common mangroves along light and salinity gradients. Oecologia 179:1187–1198. https://doi.org/10.1007/s00442-015-3408-1
Dassanayake M, Haas JS, Bohnert HJ, Cheeseman JM (2009) Shedding light on an extremophile lifestyle through transcriptomics. New Phytol 183:764–775. https://doi.org/10.1111/j.1469-8137.2009.02913.x
Desotgiu R, Pollastrini M, Cascio C, Gerosa G, Marzuoli R, Bussotti F (2013) Responses to ozone on Populus “Oxford” clone in an open top chamber experiment assessed before sunrise and in full sunlight. Photosynthetica 51:267–280. https://doi.org/10.1007/s11099-012-0074-y
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Duke NC, Allen JA (2006) Rhizophora mangle, R. samoensis, R. racemose, R. x harrisonii (Atlantic-East Pacific red mangrove). In: Elevitch CR (ed) Species profiles for Pacific Island agroforestry. Permanent Agriculture Resources, Hölualoa
Fan DY, Hope AB, Smith PJ, Jia H, Pace RJ, Anderson JM, Chow WS (2007) The stoichiometry of the two photosystems in higher plants revisited. Biochim Biophys Acta 1767:1064–1072. https://doi.org/10.1016/j.bbabio.2007.06.001
Farnworth EJ, Ellison AM, Gong WK (1996) Elevated CO2 alters anatomy, physiology, growth, and reproduction of red mangrove (Rhizophora mangle L.). Oecologia 108:599–609
Flowers TJ, Colmer TD (2008) Salinity tolerance in halophytes. New Phytol 179:945–963. https://doi.org/10.1111/j.1469-8137.2008.02531.x
Guo W, Wu H, Zhang Z, Yang C, Hu L, Shi X, Jian S, Shi S, Huang Y (2017) Comparative analysis of transcriptomes in Rhizophoraceae provides insights into the origin and adaptive evolution of mangrove plants in intertidal environments. Front Plant Sci 8:795. https://doi.org/10.3389/fpls.2017.00795
Hasegawa PM, Bressan RA, Zhu JK, Bohnert HJ (2000) Plant cellular and molecular responses to high salinity. Annu Rev Plant Physiol Plant Mol Biol 51:463–499
Heldt HW, Piechulla B (2011) Plant Biochemistry, 4rd. Academic Press, Elsevier
Järvi S, Suorsa M, Aro EM (2015) Photosystem II repair in plant chloroplasts–regulation, assisting proteins and shared components with photosystem II biogenesis. Biochim Biophys Acta 1847:900–909. https://doi.org/10.1016/j.bbabio.2015.01.006
**g X, Hou P, Lu Y, Deng S, Li N, Zhao R, Sun J, Wang Y, Han Y, Lang T, Ding M, Shen X, Chen S (2015) Overexpression of copper/zinc superoxide dismutase from mangrove Kandelia candel in tobacco enhances salinity tolerance by the reduction of reactive oxygen species in chloroplast. Front Plant Sci 6:1–13. https://doi.org/10.3389/fpls.2015.00023
Kalaji HM, Govindjee BK, Koscielniak J, Zuk-Gołaszewskae K (2011) Effects of salt stress on photosystem II efficiency and CO2 assimilation of two Syrian barley landraces. Environ Exp Bot 73:64–72. https://doi.org/10.1016/j.envexpbot.2010.10.009
Kathiresan K, Moorthy P (1993) Influence of different irradiance on growth and photosynthetic characteristics in seedlings of Rhizophora species. Photosynthetica 29:143–146
Kitao M, Utsugi H, Kuramoto S, Tabuchi R, Fujimoto K, Lihpai S (2003) Light-dependent photosynthetic characteristics indicated by chlorophyll fluorescence in five mangrove species native to Pohnpei Island, Micronesia. Physiol Plant 117:376–382. https://doi.org/10.1034/j.1399-3054.2003.00042.x
Krauss KW, Twilley RR, Thomas W, Doyle TW, Gardiner ES (2006) Leaf gas exchange characteristics of three neotropical mangrove species in response to varying hydroperiod. Tree Physiol 26:959–968. https://doi.org/10.1093/treephys/26.7.959
Krishnamurthy P, Vishal B, Khoo K, Rajappa S, Loh CS, Kumar PP (2019) Expression of AoNHX1 increases salt tolerance of rice and Arabidopsis, and bHLH transcription factors regulate AtNHX1 and AtNHX6 in Arabidopsis. Plant Cell Rep 38(10):1299–1315. https://doi.org/10.1007/s00299-019-02450-w
Lara RJ, Cohen MCL (2006) Sediment porewater salinity and mangrove vegetation height in Bragança, North Brazil: an ecohydrology-based empirical model. Wetl Ecol Manag 14:349–358. https://doi.org/10.1007/s11273-005-4991-4
Liang W, Ma X, Wan P, Liu L (2018) Plant salt-tolerance mechanism: a review. Biochem Biophys Res Commun 495:286–291. https://doi.org/10.1016/j.bbrc.2017.11.043
Lima KOO, Tognella MMP, Cunha SR, De Andrade HA (2018) Growth models of Rhizophora mangle L. seedlings in tropical southwestern Atlantic. Estuar Coast Shelf Sci 207:154–163. https://doi.org/10.1016/j.ecss.2018.03.021
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−ΔΔC(T)) method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lopes DMS, Tognella MMPT, Falqueto AR, Soares MLG (2019) Salinity variation effects on photosynthetic responses of the mangrove species Rhizophora mangle L. growing in natural habitats. Photosynthetica 57(4):1142–1155. https://doi.org/10.32615/ps.2019.121
López-Hoffman L, DeNoyer JL, Monroe IE, Shaftel R, Anten NPR, Martínez-Ramos M, David D, Ackerly DD (2006) Mangrove seedling net photosynthesis, growth, and survivorship are interactively affected by salinity and light. Biotropica 38(5):606–616. https://doi.org/10.1111/j.1744-7429.2006.00189.x
Lovelock CE, Feller IC, Mckee KL, Engelbrecht BMJ, Ball MC (2004) The effect of nutrient enrichment on growth, photosynthesis and hydraulic conductance of dwarf mangroves in Panamá. Funct Ecol 18:25–33. https://doi.org/10.1046/j.0269-8463.2004.00805.x
Lovelock CE, Reef R, Ball MC (2017) Isotopic signatures of stem water reveal differences in water sources accessed by mangrove tree species. Hydrobiologia 803:133–145. https://doi.org/10.1007/s10750-017-3149-8
Luzhen C, Wenqing W, Peng L (2005) Photosynthetic and physiological responses of Kandelia candel I. Druce seedlings to duration of tidal immersion in artificial seawater. Environ Exp Bot 54:256–266. https://doi.org/10.1016/j.envexpbot.2004.09.004
Mehta P, Jajoo A, Mathur S, Bharti S (2010) Chlorophyll a fluorescence study revealing effects of high salt stress on Photosystem II in wheat leaves. Plant Physiol Biochem 48:16–20. https://doi.org/10.1016/j.plaphy.2009.10.006
Mirzaei S, Mansouri M, Mohammadi-Nejad G, Sablok G (2018) Comparative assessment of chloroplast transcriptional responses highlights conserved and unique patterns across Triticeae members under salt stress. Photosynth Res 136:357–369. https://doi.org/10.1007/s11120-017-0469-5
Naidoo G, Tuffers AV, von Willert DJ (2002) Changes in gas exchange and chlorophyll fluorescence characteristics of two mangroves and a mangrove associate in response to salinity in the natural environment. Trees 16:140–146. https://doi.org/10.1007/s00468-001-0134-6
Naidoo G, Hiralal O, Naidoo Y (2011) Hypersalinity effects on leaf ultrastructure and physiology in the mangrove Avicennia marina. Flora - Morphology Distribution Functional Ecology of Plants 206(9):814-820. https://doi.org/10.1016/j.flora.2011.04.009
Oukarroum A, Bussotti F, Goltsev V, Kalaji HM (2015) Correlation between reactive oxygen species production and photochemistry of photosystems I and II in Lemna gibba L. plants under salt stress. Environ Exp Bot 109:80–88. https://doi.org/10.1016/j.envexpbot.2014.08.005
Palliyath S, Puthur JT (2018) The modulation of various physiochemical changes in Bruguiera cylindrica (L.) Blume affected by high concentrations of NaCl. Acta Physiol Plant 40:160. https://doi.org/10.1007/s11738-018-2735-3
Parida AK, Jha B (2010) Salt tolerance mechanisms in mangroves: a review. Trees 24:199–217. https://doi.org/10.1007/s00468-010-0417-x
Parida AK, Das AB, Mohanty P (2004) Investigations on the antioxidative defence responses to NaCl stress in a mangrove, Bruguiera parviflora: differential regulations of isoforms of some antioxidative enzymes. Plant Growth Regul 42:213–226. https://doi.org/10.1023/B:GROW.0000026508.63288.39
Parry MA, Keys AJ, Madgwick PJ, Carmo-Silva AE, Andralojc PJ (2008) Rubisco regulation: a role for inhibitors. J Exp Bot 59:1569–1580. https://doi.org/10.1093/jxb/ern084
Peng YL, Wang YS, Gu JD (2015) Identification of suitable reference genes in mangrove Aegiceras corniculatum under abiotic stresses. Ecotoxicology 24(7–8):1714–1721. https://doi.org/10.1007/s10646-015-1487-8
Scholander PF (1968) How mangrove desalinated water. Physiol Plant 21:251–261. https://doi.org/10.1111/j.1399-3054.1968.tb07248.x
Soares MLG, Tognella MMP, Cuevas E, Medina E (2015) Photosynthetic capacity and intrinsic water-use efficiency of Rhizophora mangle at its southernmost western Atlantic range. Photosynthetica 53:464–470. https://doi.org/10.1007/s11099-015-0119-0
Sobrado MA (2005) Leaf characteristics and gas exchange of the mangrove Laguncularia racemosa as affected by salinity. Photosynthetica 43:217–221. https://doi.org/10.1007/s11099-005-0036-8
Strasser RJ, Tsimilli-Michael M, Srivastava A (2004) Analysis of the chlorophyll a fluorescence transient. In: Papageorgiou GC, Govindjee (eds) Chlorophyll a fluorescence. Advances in photosynthesis and respiration. Springer, Dordrecht, pp 321–362. https://doi.org/10.1007/978-1-4020-3218-9_12
Su W, Ye C, Zhang Y, Hao S, Li QQ (2019) Identification of putative key genes for coastal environments and cold adaptation in mangrove Kandelia obovata through transcriptome analysis. Sci Total Environ 681:191–201. https://doi.org/10.1016/j.scitotenv.2019.05.127
Sugihara K, Hanagata N, Dubinsky Z, Baba S, Karube I (2000) Molecular characterization of cDNA encoding oxygen evolving enhancer protein 1 increased by salt treatment in the mangrove Bruguiera gymnorrhiza. Plant and Cell Physiology 41(11):1279–1285. https://doi.org/10.1093/pcp/pcd061
Suo J, Zhao Q, David L, Chen S, Dai S (2017) Salinity response in chloroplasts: insights from gene characterization. Int J Mol Sci 18(5):1–17. https://doi.org/10.3390/ijms18051011
Tikhonov AN (2013) pH-dependent regulation of electron transport and ATP synthesis in chloroplasts. Photosynth Res 116:511–534. https://doi.org/10.1007/s11120-013-9845-y
Twilley RR, Rivera-Monroy VH, Chen R, Botero L (1999) Adapting an ecological mangrove model to simulate trajectories in restoration ecology. Mar Pollut Bull 37:404–419. https://doi.org/10.1016/S0025-326X(99)00137-X
Wang L, Liu X, Liang M, Tan F, Liang W, Chen Y, Lin Y, Huang L, **ng J, Chen W (2014) Proteomic analysis of salt-responsive proteins in the leaves of mangrove Kandelia candel during short-term stress. PLoS ONE 9(1):e83141. https://doi.org/10.1371/journal.pone.0083141
Wang L, Pan D, Li F, Hoffmann-Benning S, Liang W, Chen W (2015) Proteomic analysis of changes in the Kandelia candel chloroplast proteins reveals pathways associated with salt tolerance. Plant Sci 231:159–172. https://doi.org/10.1016/j.plantsci.2014.11.013
Wang Y, Gu W, Meng Y, **e T, Li L, Li J, Wei S (2017) γ-Aminobutyric acid imparts partial protection from salt stress injury to maize seedlings by improving photosynthesis and upregulating osmoprotectants and antioxidants. Sci Rep 7:43609. https://doi.org/10.1038/srep43609
Werner A, Stelzer R (1990) Physiological responses of the mangrove Rhizophora mangle grown in the absence and presence of NaCl. Plant Cell Environ 13:243–255. https://doi.org/10.1111/j.1365-3040.1990.tb01309.x
Woodroffe C (1992) Mangrove sediments and geomorphology. In: Robertson AI, Alongi DM (eds) Tropical mangrove ecosystem. American Geophysical Union, Washington D.C., pp 7–41
Xue ZY, Zhi DY, Xue G, Zhang H, Zhao Y, **a G (2004) Enhanced salt tolerance of transgenic wheat (Tritivum aestivum L.) expressing a vacuolar Na+/H+ antiporter gene with improved grain yields in saline soils in the field and a reduced level of leaf Na+. Plant Sci 167:849–859. https://doi.org/10.1016/j.plantsci.2004.05.034
Yang Y, Guo Y (2018) Elucidating the molecular mechanisms mediating plant salt-stress responses. New Phytol 217:523–539. https://doi.org/10.1111/nph.14920
Yusuf MA, Kumar D, Rajwanshi R, Strasser RJ, Tsimilli-Michae LM, Govindjee SNB (2010) Overexpression of γ-tocopherol methyl transferase gene in transgenic Brassica juncea plants alleviates abiotic stress: Physiological and chlorophyll a fluorescence measurements. Biochi Biophys Acta 1797:1428–1438. https://doi.org/10.1016/j.bbabio.2010.02.002
Zamprogno GC, Tognella MMP, Quaresma VS, Costa MB, Pascoalini SS, Couto GF (2016) The structural heterogeneity of an urbanised mangrove forest area in southeastern Brazil: Influence of environmental factors and anthropogenic stressors. Braz j Oceanogr 64(2):157–172. https://doi.org/10.1590/S1679-87592016111706402
Zhang RH, Zhang XR, Camberato JJ, Xue JQ (2015) Photosynthetic performance of maize hybrids to drought stress. Russ J Plant Physiol 62:788–796. https://doi.org/10.1134/S1021443715060187
Zhu Z, Chen J, Zheng HL (2012) Physiological and proteomic characterization of salt tolerance in a mangrove plant, Bruguiera gymnorrhiza (L.) Lam. Tree Physiol 32:1378–1388. https://doi.org/10.1093/treephys/tps097
Acknowledgements
The authors thank Dr. Mário Luiz Gomes Soares from Rio de Janeiro State University, for lending us the IRGA, and Dr. Gilberto Fillmann, coordinator of the national project, financed by the FINEP, which supported us in the acquisition of the Ultrafreezer.
Funding
This study was supported by Fundo Brasileiro para Biodiversidade (FUNBIO), TFCA Agreement No. 04/2012, and FINEP (CT-Hidro No. 01/2013). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brazil (CAPES) - Finance Code 001 - during the first author's PhD. The second author was supported by the National Council for Scientific and Technological Development, Brazil (CNPq) during her Phd studies. The last author was supported by Fundação de Amparo à Pesquisa e Inovação do Espírito Santo (FAPES) research budget (Grants No. 60127627/2012 and No. 263/2016).
Author information
Authors and Affiliations
Contributions
DMSL, MMPT, ARF, MR, and ASL conceived and designed the research. DMSL, ASL, ARF, MR, and MMPT conducted experiments and analyzed the data. ABPLG, ARF, MR, and MMPT contributed with reagents and materials. DMSL, MMPT, ASL, and MR wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Communicated by B. Wang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lopes, D., Lopes, A., Falqueto, A. et al. Photosynthetic and gene expression analyses in Rhizophora mangle L. plants growing in field conditions provide insights into adaptation to high-salinity environments. Trees 37, 733–747 (2023). https://doi.org/10.1007/s00468-022-02380-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-022-02380-3