Abstract
Background
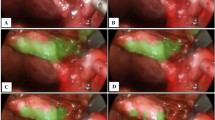
Operative courses of laparoscopic cholecystectomies vary widely due to differing pathologies. Efforts to assess intra-operative difficulty include the Parkland grading scale (PGS), which scores inflammation from the initial view of the gallbladder on a 1–5 scale. We investigated the impact of PGS on intra-operative outcomes, including laparoscopic duration, attainment of the critical view of safety (CVS), and gallbladder injury. We additionally trained an artificial intelligence (AI) model to identify PGS.
Methods
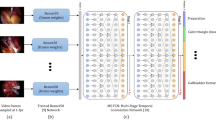
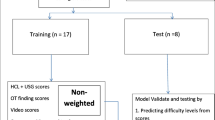
One surgeon labeled surgical phases, PGS, CVS attainment, and gallbladder injury in 200 cholecystectomy videos. We used multilevel Bayesian regression models to analyze the PGS’s effect on intra-operative outcomes. We trained AI models to identify PGS from an initial view of the gallbladder and compared model performance to annotations by a second surgeon.
Results
Slightly inflamed gallbladders (PGS-2) minimally increased duration, adding 2.7 [95% compatibility interval (CI) 0.3–7.0] minutes to an operation. This contrasted with maximally inflamed gallbladders (PGS-5), where on average 16.9 (95% CI 4.4–33.9) minutes were added, with 31.3 (95% CI 8.0–67.5) minutes added for the most affected surgeon. Inadvertent gallbladder injury occurred in 25% of cases, with a minimal increase in gallbladder injury observed with added inflammation. However, up to a 28% (95% CI − 2, 63) increase in probability of a gallbladder hole during PGS-5 cases was observed for some surgeons. Inflammation had no substantial effect on whether or not a surgeon attained the CVS. An AI model could reliably (Krippendorff’s α = 0.71, 95% CI 0.65–0.77) quantify inflammation when compared to a second surgeon (α = 0.82, 95% CI 0.75–0.87).
Conclusions
An AI model can identify the degree of gallbladder inflammation, which is predictive of cholecystectomy intra-operative course. This automated assessment could be useful for operating room workflow optimization and for targeted per-surgeon and per-resident feedback to accelerate acquisition of operative skills.
Graphical abstract





Similar content being viewed by others
References
Sugrue M, Sahebally SM, Ansaloni L, Zielinski MD (2015) Grading operative findings at laparoscopic cholecystectomy—a new scoring system. World J Emerg Surg 10:14. https://doi.org/10.1186/s13017-015-0005-x
Nassar AHM, Ashkar KA, Mohamed AY, Hafiz AA (1995) Is laparoscopic cholecystectomy possible without video technology? Minim Invasive Ther 4:63–65. https://doi.org/10.3109/13645709509152757
Madni TD, Leshikar DE, Minshall CT, Nakonezny PA, Cornelius CC, Imran JB, Clark AT, Williams BH, Eastman AL, Minei JP, Phelan HA, Cripps MW (2018) The Parkland grading scale for Cholecystitis. Am J Surg 215:625–630. https://doi.org/10.1016/j.amjsurg.2017.05.017
Sugrue M, Coccolini F, Bucholc M, Johnston A, Manatakis D, Ioannidis O, Bonilauri S, Gachabayov M, Isik A, Ghnnam W, Shelat V, Aremu M, Mohan R, Montori G, Walędziak M, Pisarska M, Kong V, Strzałka M, Fugazzola P, Nita GE, Nardi M, Major P, Negoi I, Allegri A, Konstantoudakis G, Di Carlo I, Massalou D, D’Amico G, Solaini L, Ceresoli M, Bini R, Zielinski M, Tomasoni M, Litvin A, De Simone B, Lostoridis E, Hernandez F, Panyor G, Machain VGM, Pentara I, Baiocchi L, Ng KC, Ansaloni L, Sartelli M, Arellano ML, Savala N, Couse N, McBride S, Contributors from WSES (2019) Intra-operative gallbladder scoring predicts conversion of laparoscopic to open cholecystectomy: a WSES prospective collaborative study. World J Emerg Surg 14:12. https://doi.org/10.1186/s13017-019-0230-9
West Midlands Research Collaborative, Griffiths EA, Hodson J, Vohra RS, Marriott P, Katbeh T, Zino S, Nassar AHM (2019) Utilisation of an operative difficulty grading scale for laparoscopic cholecystectomy. Surg Endosc 33:110–121. https://doi.org/10.1007/s00464-018-6281-2
Madni TD, Nakonezny PA, Barrios E, Imran JB, Clark AT, Taveras L, Cunningham HB, Christie A, Eastman AL, Minshall CT, Luk S, Minei JP, Phelan HA, Cripps MW (2019) Prospective validation of the Parkland grading scale for Cholecystitis. Am J Surg 217:90–97. https://doi.org/10.1016/j.amjsurg.2018.08.005
Stepaniak PS, Heij C, Mannaerts GHH, de Quelerij M, de Vries G (2009) Modeling procedure and surgical times for current procedural terminology-anesthesia-surgeon combinations and evaluation in terms of case-duration prediction and operating room efficiency: a multicenter study. Anesth Analg 109:1232–1245. https://doi.org/10.1213/ANE.0b013e3181b5de07
Bellard F (2021) FFmpeg. https://ffmpeg.org/about.html. Accessed 21 Jun 2021
Ban Y, Rosman G, Ward T, Hashimoto D, Kondo T, Iwaki H, Meireles O, Rus D (2021) Aggregating long-term context for learning laparoscopic and robot-assisted surgical workflows. Accessed https://arxiv.org/abs/2009.00681
Strasberg SM, Hertl M, Soper NJ (1995) An analysis of the problem of biliary injury during laparoscopic cholecystectomy. J Am Coll Surg 180:101–125
He K, Zhang X, Ren S, Sun J (2016) Deep residual learning for image recognition. In: Proceedings of the IEEE conference on computer vision and pattern recognition (CVPR)
Howard J, Gugger S (2020) Fastai: a layered API for deep learning. Information 11:108. https://doi.org/10.3390/info11020108
Smith LN (2017) Cyclical learning rates for training neural networks. In: 2017 IEEE winter conference on applications of computer vision (WACV). pp 464–472
Strum DP, May JH, Vargas LG (2000) Modeling the uncertainty of surgical procedure times. Anesthesiology 92:1160–1167. https://doi.org/10.1097/00000542-200004000-00035
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
McElreath R (2020) Rethinking: statistical rethinking book package. CRC Press, Boca Raton
Gabry J, Češnovar R (2020) cmdstanr: R interface to “CmdStan”
Vehtari A, Gelman A, Simpson D, Carpenter B, Bürkner P-C (2021) Rank-normalization, folding, and localization: an improved Rˆ for assessing convergence of MCMC. Bayesian Anal 1:1–38. https://doi.org/10.1214/20-BA1221
Krippendorff K (2004) Content analysis: an introduction to its methodology, 2nd edn. Sage, Thousand Oaks
Gamer M, Lemon J, Singh IFP (2019) irr: various coefficients of interrater reliability and agreement. CRAN
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York
Kay M (2021) ggdist: visualizations of distributions and uncertainty
Ward TM (2021) tmward/pgs: artificial intelligence prediction of cholecystectomy operative course from automated identification of gallbladder inflammation code. Accessed https://doi.org/10.5281/zenodo.5328655
Levine WC, Dunn PF (2015) Optimizing operating room scheduling. Anesthesiol Clin 33:697–711. https://doi.org/10.1016/j.anclin.2015.07.006
Thiels CA, Yu D, Abdelrahman AM, Habermann EB, Hallbeck S, Pasupathy KS, Bingener J (2017) The use of patient factors to improve the prediction of operative duration using laparoscopic cholecystectomy. Surg Endosc 31:333–340. https://doi.org/10.1007/s00464-016-4976-9
Ban Y, Rosman G, Ward T, Hashimoto D, Kondo T, Iwaki H, Meireles O, Rus D (2021) SUrgical PRediction GAN for events anticipation. Accessed https://arxiv.org/abs/2105.04642
Twinanda AP, Yengera G, Mutter D, Marescaux J, Padoy N (2019) RSDNet: learning to predict remaining surgery duration from laparoscopic videos without manual annotations. IEEE Trans Med Imaging 38:1069–1078. https://doi.org/10.1109/TMI.2018.2878055
The Prevention of Bile Duct Injury Consensus Work Group, Michael Brunt L, Deziel DJ, Telem DA, Strasberg SM, Aggarwal R, Asbun H, Bonjer J, McDonald M, Alseidi A, Ujiki M, Riall TS, Hammill C, Moulton C-A, Pucher PH, Parks RW, Ansari MT, Connor S, Dirks RC, Anderson B, Altieri MS, Tsamalaidze L, Stefanidis D (2020) Safe cholecystectomy multi-society practice guideline and state-of-the-art consensus conference on prevention of bile duct injury during cholecystectomy. Surg Endosc 34:2827–2855. https://doi.org/10.1007/s00464-020-07568-7
Ward TM, Mascagni P, Madani A, Padoy N, Perretta S, Hashimoto DA (2021) Surgical data science and artificial intelligence for surgical education. J Surg Oncol 124:221–230. https://doi.org/10.1002/jso.26496
Twinanda AP, Shehata S, Mutter D, Marescaux J, de Mathelin M, Padoy N (2017) EndoNet: a deep architecture for recognition tasks on laparoscopic videos. IEEE Trans Med Imaging 36:86–97. https://doi.org/10.1109/TMI.2016.2593957
Mascagni P, Vardazaryan A, Alapatt D, Urade T, Emre T, Fiorillo C, Pessaux P, Mutter D, Marescaux J, Costamagna G, Dallemagne B, Padoy N (2021) Artificial Intelligence for Surgical Safety: Automatic Assessment of the Critical View of Safety in Laparoscopic Cholecystectomy Using Deep Learning. Ann Surg. https://doi.org/10.1097/SLA.0000000000004351
Acknowledgements
This work was supported by a 2018 research award from the Risk Management Foundation of the Harvard Medical Institutions Incorporated (CRICO/RMF), grant number 233456. The authors thank Caitlin E. Stafford, CCRP, for her assistance and support in research management, and Allison J. Navarrete-Welton, for her assistance in data collection.
Funding
This study was funded by the Risk Management Foundation of the Harvard Medical Institutions Incorporated (CRICO/RMF), Grant Number 233456.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Drs. Ban, Hashimoto, Meireles, Rosman, and Ward receive research support from Olympus Corporation. Drs. Ban, Hashimoto, Meireles, Rosman, and Ward have received research support from the Risk Management Foundation of the Harvard Medical Institutions Incorporated (CRICO/RMF). Dr. Hashimoto is a consultant for Johnson & Johnson, Activ Surgical, and Verily Life Sciences. Dr. Hashimoto has received research support from the Intuitive Foundation and the Society of American Gastrointestinal and Endoscopic Surgeons. Dr. Rosman receives research support from Toyota Research Institute (TRI). Dr. Meireles is a consultant for Medtronic and Olympus Corporation.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ward, T.M., Hashimoto, D.A., Ban, Y. et al. Artificial intelligence prediction of cholecystectomy operative course from automated identification of gallbladder inflammation. Surg Endosc 36, 6832–6840 (2022). https://doi.org/10.1007/s00464-022-09009-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-022-09009-z