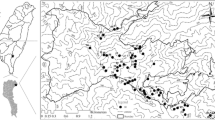
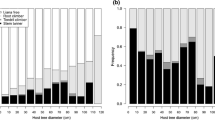
Abstract
Soil hydrology, nutrient availability, and forest disturbance determine the variation of tropical tree species composition locally. However, most habitat filtering is explained by tree species' hydraulic traits along the hydrological gradient. We asked whether these patterns apply to lianas. At the community level, we investigated whether hydrological gradient, soil fertility, and forest disturbance explain liana species composition and whether liana species-environment relationships are mediated by leaf and stem wood functional traits. We sampled liana species composition in 18 1-ha plots across a 64 km2 landscape in Central Amazonia and measured eleven leaf and stem wood traits across 115 liana species in 2000 individuals. We correlated liana species composition, summarized using PCoA with the functional composition summarized using principal coordinate analysis (PCA), employing species mean values of traits at the plot level. We tested the relationship between ordination axes and environmental gradients. Liana species composition was highly correlated with functional composition. Taxonomic (PCoA) and functional (PCA) compositions were strongly associated with the hydrological gradient, with a slight influence from forest disturbance on functional composition. Species in valley areas had larger stomata size and higher proportions of self-supporting xylem than in plateaus. Liana species on plateaus invest more in fast-growing leaves (higher SLA), although they show a higher wood density. Our study reveals that lianas use different functional solutions in dealing with each end of the hydrological gradient and that the relationships among habitat preferences and traits explain lianas species distributions less directly than previously found in trees.





Similar content being viewed by others
Availability of data and material
The datasets generated for this study are available on request to the corresponding author.
References
Aerts R (1995) The advantages of being evergreen. Trends in Ecology & 10:402–407. https://doi.org/10.1016/S0169-5347(00)89156-9
Aerts R, Chapin FS (2000) The mineral nutrition of wild plants revisited A re-evaluation of processes and patterns. Adv Ecol Res 30:1–67. https://doi.org/10.1016/S0065-2504(08)60016-1
Angyalossy V, Pace MR, Lima AC (2015) Liana anatomy: a broad perspective on structural evolution of the vascular system. In: Schnitzer SA, Bongers F, Burnham RJ, Putz FE (eds) The Ecology of Lianas. Wiley-Blackwell, Oxford, UK, pp 251–287
Apgaua DMG, Tng DYP, Laurance SGW (2022) Tropical wet and dry forest tree species exhibit contrasting hydraulic architecture. Flora. https://doi.org/10.1016/j.flora.2022.152072
Arnott HJ (1959) Leaf clearings. Turtox News 37:192–194
Balvanera P, Quijas S, Pérez-Jiménez, (2011) Distribution patterns of tropical dry forest trees along a mesoscale water availability gradient. Biotropica 43:414–422. https://doi.org/10.1111/j.1744-7429.2010.00712.x
Baraloto C, Paine TC, Poorter L, Beauchene J, Bonal D, Domenach AM et al (2010) Decoupled leaf and stem economics in rain forest trees. Ecol Lett 13(11):1338–1347. https://doi.org/10.1111/j.1461-0248.2010.01517.xÓ2010
Barbosa AC, Gerolamo CS, Lima AC, Angyalossy V, Pace MR (2021) Polishing entire stems and roots using sandpaper under water: An alternative method for macroscopic analyses. Appl Plant Sci 9:15–21. https://doi.org/10.1002/aps3.11421
Barton K (2014) Package ‘MuMIn’: multi-model inference. R package version 1.13.4. https://cran.r-project.org/web/packages/MuMIn/ index.html
Brodribb TJ, Holbrook NM (2006) Declining hydraulic efficiency as transpiring leaves desiccate: two types of response. Plant Cell Environ 29:2205–2215. https://doi.org/10.1111/j.1365-3040.2006.01594.x
Bukatsch F (1972) Bemerkungen zur doppelfärbung astrablau-safranin. Mikrocosmos 61:255
Campanello PI, Manzané E, Villagra M, Zhang YJ, Panizza AM, di Francescantonio D et al (2016) Carbon allocation and water relations of lianas versus trees. In: Goldstein G, Santiago L (eds) Tropical tree physiology, Springer, Cham, Germany, pp 103–124. https://doi.org/10.1007/978-3-319-27422-5_5
Carlquist S (1975) Anatomy and Habitat: Ecological Strategies of Xylem Evolution In: University of California Press, Berkeley, Doi: https://doi.org/10.1525/9780520320567
Castilho CV, Magunsson WE, Araújo RNO, Luizão RCC, Luizão JF, Lima AP et al (2006) Variation in aboveground tree live biomass in a central Amazonian Forest: Effects of soil and topography. For Ecol Manag 234:85–96. https://doi.org/10.1016/j.foreco.2006.06.024
Castilho CV, Magnusson WE, FJ, (2010) Short-term temporal changes in tree live biomass in a central Amazonian Forest, Brazil. Biotropica 42:95–103. https://doi.org/10.1111/j.1744-7429.2009.00543.x
Chauvel A, Lucas Y, Boulet R (1987) On the genesis of the soil mantle of the region of Manaus, Central Amazonia, Brazil. Experientia 43:234–241. https://doi.org/10.1007/BF01945546
Chave J, Coomes D, Jansen S, Lewis SL, Swenson NG, Zanne AE (2009) Towards a worldwide wood economics spectrum. Ecol Lett 12:351–366. https://doi.org/10.1111/j.1461-0248.2009.01285.x
Chen YJ, Cao KF, Schnitzer SA, Ze-**n V, Zhang JL et al (2015) Water-use advantage for lianas over trees in tropical seasonal forests. New Phytol 205:128–136. https://doi.org/10.1111/nph.13036
Condit R, Bettina MJ, Engelbrecht DP, Pérez R, Turner BL (2013) Species distributions in response to individual soil nutrients and seasonal drought across a community of tropical trees. – Proc. Natl Acad Sci USA 110:5064–5068. https://doi.org/10.1073/pnas.1218042110
Cornelissen JHC, Lavorel S, Garnier E, Díaz S, Buchmann N, Gurvich DE et al (2003) A handbook of protocols for standardised and easy measurement of plant functional traits worldwide. Aust J Bot 51:335–380. https://doi.org/10.1071/BT02124
Cosme LHM, Schietti J, Costa FRC, Oliveira RS (2017) The importance of hydraulic architecture to the distribution patterns of trees in a central Amazonian forest. New Phytol 215:113–125. https://doi.org/10.1111/nph.14508
Costa FRC, Magnusson WE (2010) The need for large-scale, integrated studies of biodiversity – The experience of the program for biodiversity research in Brazilian Amazonia. Nat Conserv 8:1. https://doi.org/10.4322/natcon.00801001
Costa FRC, Magnusson WE, Luizão RC (2005) Mesoscale distribution patterns of Amazonian understory herbs in relation to topography, soil and watersheds. J Ecol 93:863–878. https://doi.org/10.1111/j.1365-2745.2005.01020.x
Costa FRC, Guillaumet JL, Lima AP, Pereira OS (2009) Gradients within gradients: the mesoscale distribution patterns of palms in a central amazonian forest. J Veg Sci 20:69–78. https://doi.org/10.1111/j.1654-1103.2009.05314.x
Costa FRC, Schietti J, Stark SC, Smith MN (2022) The other side of tropical forest drought: do shallow water table regions of Amazonia act as large-scale hydrological refugia from drought? New Phytol. https://doi.org/10.1111/nph.17914
De Deurwaerder H, Hervé-Fernández P, Stahl C, Burban B, Petronelli P, Hoffman B et al (2018) Liana and tree below-ground water competition-evidence for water resource partitioning during the dry season. Tree Physiol 38:1071–1083. https://doi.org/10.1093/treephys/tpy002
DeWalt SJ, Schnitzer SA, Alves LF, Bongers F, Burnham RJ, Cai Z et al (2015) Biogeographical patterns of liana abundance and diversity. In: Schnitzer SA, Bongers F, Burnham RJ, Putz FE (eds) Ecology of Lianas. Wiley, Chichester, pp 131–146
Díaz S, Kattge J, Cornelissen JH, Wright IJ, Lavorel S, Dray S et al (2016) The global spectrum of plant form and function. Nature 529(7585):167–171. https://doi.org/10.1038/nature16489
Dormann CF, Elith J, Bacher S, Buchmann C, Carl G, Carré G et al (2013) Collinearity: A review of methods to deal with it and a simulation study evaluating their performance. Ecography (cop) 36:27–46. https://doi.org/10.1111/j.1600-0587.2012.07348.x
Drucker DP, Costa FRC, Magnusson WE (2008) How wide is the riparian zone of small streams in tropical forests? A test with terrestrial herbs. J Trop Ecol 24:65–74. https://doi.org/10.1017/S0266467407004701
Ewers FW, Fisher JB, Fichtner K (1991) Water flux and xylem structure in vines. In: Putz FE (ed) The biology of vines. Cambridge University Press, Cambridge, pp 127–160
Fan Y, Miguez-Macho G, Jobbágy EG, Jackson RB, Casal CO (2017) Hydrologic regulation of plant rooting depth. Proc Natl Acad Sci USA 40:10572–10577. https://doi.org/10.1073/pnas.1712381114
Garnier E, Navas ML (2012) A trait-based approach to comparative functional plant ecology: concepts, methods and applications for agroecology. A Review Agron Sustain Dev 32:365–399. https://doi.org/10.1007/s13593-011-0036-y
Garnier E, Shipley B, Roumet C, Laurent G (2001a) A standardized protocol for the determination of specific leaf area and leaf dry matter content. Funct Ecol 15:688–695. https://doi.org/10.1046/j.0269-8463.2001.00563.x
Garnier E, Laurent G, Debain S, Berthelier P, Ducout B, Roumet C et al (2001b) Consistency of species ranking based on functional leaf traits. New Phytol 152:69–83. https://doi.org/10.1046/j.0028-646x.2001.00239.x
Gentry AH (1991) The distribution and evolution of climbing plants. In: Putz FE, Mooney HA (eds) The biology of vines. Cambridge University Press, Cambridge, UK, pp 618–629
Gerolamo CS, Angyalossy V (2017) Wood anatomy and conductivity in lianas, shrubs and trees of Bignoniaceae. IAWA J 38:412–432. https://doi.org/10.1163/22941932-20170177
Gerolamo CS, Nogueira A, Costa FRC, Castilho VC, Angyalossy V (2018) Local dynamic variation of lianas along topography maintains unchanging abundance at the landscape scale in central Amazonia. J Veg Sci 29:651–661. https://doi.org/10.1111/jvs.12644
Gerolamo CS, Nogueira A, Pace MR, Angyalossy V (2020) Interspecific anatomical differences result in similar highly flexible stems in Bignoniaceae lianas. Am J Bot 107:1622–1634. https://doi.org/10.1002/ajb2.1577
Gerwing JJ, Schnitzer SA, Burnham RJ, Bongers F, Chave J, DeWalt SJ et al (2006) A standard protocol for liana censuses 1. Biotropica 38:256–261. https://doi.org/10.1111/j.1744-7429.2006.00134.x
Grime JP, Thompson K, Hunt R, Hodgson JG, JCornelissen HC, Rorison IH, et al (1997) Integrated screening validates primary axes of specialization in plants. Oikos 79:259–281. https://doi.org/10.2307/3546011
Hobbie SE (1992) Effects of plant species on nutrient cycling. Trends Ecol Evol 7:336–339. https://doi.org/10.1016/0169-5347(92)90126-V
Hodnett MG, Vendrame I, AO, Marques Filho MD, Oyama MD, Tomasella J, (1997) Soil and groundwater behaviour in a catenary sequence beneath forest in Central Amazonia: I. Comparisons between plateau, slope and valley floor. Hydrol Earth Sys Sci 1:265–277. https://doi.org/10.5194/hess-1-265-1997
Hofhansl F, Chacón-Madrigal E, Brännström Å, Dieckmann U, Franklin O (2021) Mechanisms driving plant functional trait variation in a tropical forest. Ecol Evol 13:3856–3870. https://doi.org/10.1002/ece3.7256
Hopkins MJG (2005) Flora da Reserva Ducke, Amazonas, Brasil. Rodriguesia 56:9–25. https://doi.org/10.1590/2175-78602005568602
Jirka S, McDonald AJ, Johnson MS, Feldpausch TR, Couto EG, Susan RJ (2007) Relationships between soil hydrology and forest structure and composition in the southern Brazilian Amazon. J Veg Sci 18:183. https://doi.org/10.1658/1100-9233
Johansen DA (1940) Plant Mycrotechnique. Mc Graw Hill Books, New York, p 523
Johnson DM, McCulloh KA, Meinzer FC, Woodruff DR, Eissenstat DM, Phillips N (2011) Hydraulic patterns and safety margins, from stem to stomata, in three eastern US tree species. Tree Physiol 6:659–668. https://doi.org/10.1093/treephys/tpr050
Kahn F, Castro A (1985) The Palm community in a forest of Central Amazonia, Brazil. Biotropica 17:210–216. https://doi.org/10.2307/2388221
Kalliola R, Puhakka M (1988) River dynamics and vegetation mosaicism: a case study of the River Kamajohka. North Finl J Biogeogr 15:703–719. https://doi.org/10.2307/2845334
Kraus J, Arduim M (1997) Manual básico de métodos em morfologia vegetal. EDUR, Seropédica, Brazil, pp 1–98
Larcher W (2000) Temperature stress and survival ability of Mediterranean sclerophyllous plants. Plant Biosyst 134:279–295. https://doi.org/10.1080/11263500012331350455
Ledo A, Illian JB, Schnitzer SA, Wright SJ, Dalling WJ, Burslem DF (2016) Lianas and soil nutrients predict fine-scale distribution of above-ground biomass in a tropical moist forest. J Ecol 104:1819–1828. https://doi.org/10.1111/1365-2745.12635
Liu C, He N, Zhang J, Li Y, Wang Q, Sack L et al (2017) Variation of stomatal traits from cold temperate to tropical forests and association with water use efficiency. Funct Ecol 32:20–28. https://doi.org/10.1111/fec.2018.32.issue-110.1111/1365-2435.12973
Liu Q, Sterck FJ, Zhang JL, Scheire A, Konings E, Cao M et al (2021) Traits, strategies, and niches of liana species in a tropical seasonal rainforest. Oecologia 196:499–514. https://doi.org/10.1007/s00442-021-04937-4
Luizão RCC, Luizão FJ, Paiva RQ, Monteiro TF, Sousa LS, Kruij B (2004) Variation of carbon and nitrogen cycling processes along a topographic gradient in a central Amazonian forest. Glob Change Biol 10:592–600. https://doi.org/10.1111/j.1529-8817.2003.00757.x
Magnusson WE, Lima AP, Luizão R, Luizão F, Costa FRC, Castilho CV et al (2005) RAPELD: A modification of the Gentry method for biodiversity surveys in long-term ecological research sites. Biota Neotrop 5:2. https://doi.org/10.1590/S1676-06032005000300002
Malizia A, Grau H, Lichstein J (2010) Soil phosphorus and disturbance influence liana communities in a subtropical montane forest. J Veg Sci 21:551–560. https://doi.org/10.1111/j.1654-1103.2009.01162.x
Manzané-Pinzón E, Goldstein G, Schnitzer SA (2018) Does soil moisture availability explain liana seedling distribution across a tropical rainfall gradient? Biotropica 50(2):215–224
Marques-Filho AO, Ribeiro MNG, Santos HM, Santos JM (1981) Estudos climatológicos da Reserva Florestal Ducke – Manaus-AM IV. Precipitação. Acta Amazôn 11:759–768. https://doi.org/10.1590/1809-43921981114759
Medina-Vega JA, Bongers F, Poorter SSA, Sterck FJ (2021) Lianas have more acquisitive traits than trees in a dry but not in a wet forest. J Ecol. https://doi.org/10.1111/1365-2745.13644
Muscarella R, Bacon CD, Faurby S, Antonelli A, Kristiansen SM, Svenning JC et al (2019) Soil fertility and flood regime are correlated with phylogenetic structure of Amazonian palm communities. Ann Bot 123:641–655. https://doi.org/10.1093/aob/mcy196
Nogueira A, Costa FRC, Castilho CV (2011) Liana abundance patterns: The role of ecological filters during development. Biotropica (lawrence, KS) 43:442–449. https://doi.org/10.1111/j.1744-7429.2010.00722.x
Oksanen J, Blanchet FG, Friendly M, Hara B, Simpson GL, O’ Hara RB, et al (2015) Vegan: community ecology package. https://cran.r-project.org/web/packages/vegan/index.html.
Oliveira RS, Costa FRC, van Baalen E, Jonge A, Bittencourt PR, Almanza Y et al (2019) Embolism resistance drives the distribution of Amazonian rainforest tree species along hydro-topographic gradients. New Phytol 221:1457–1465. https://doi.org/10.1111/nph.15463
Ordoñez JC, Van Bodegom PM, Witte JP, Wright IJ, Reich PB, Aerts R (2009) A global study of relationships between leaf traits, climate and soil measures of nutrient fertility. Glob Ecol Biogeogr 18:137–149. https://doi.org/10.1111/j.1466-8238.2008.00441.x
Parolin P, Ferreira LV, Junk WJ (2003) Germination characteristics and establishment of trees from central Amazonian flood plains. Trop Ecol 44:155–167
Phillips OL, Gentry AH (1994) Increasing turnover through time in tropical forests. Science 263:954–958
Putz FE (1980) Lianas vs. Trees. Biotropica 12(3):224–225
Putz FE (1984) The natural history of lianas on Barro Colorado Island, Panama. Ecology 68:1713–1724
Putz FE, Mooney HA (1991) The biology of vines. Cambridge University Press, Cambridge
Reich PB (2014) The world-wide ‘fast–slow’ plant economics spectrum: a traits manifesto. J Ecol 102:275–301. https://doi.org/10.1111/1365-2745.12211
Reich PB, Walters MB, Ellsworth DS (1997) From tropics to tundra: global convergence in plant functioning. Proc Natl Acad Sci USA 94:13730–13734. https://doi.org/10.1073/pnas.94.25.13730
Reich PB, Ellsworth DS, Walters MB, Vose JM, Gresham C, Volin JC et al (1999) Generality of leaf traits relationships: a test across six biomes. Ecology 80:1955–1969. https://doi.org/10.1890/0012-9658
Reich PB, Wright IJ, Cavender-Bares J, Craine J, Oleksyn J (2003) The evolution of plant functional variation: traits, spectra, and strategies. Int J Plant Sci 16:143–164. https://doi.org/10.1086/374368
Rennó CD, Nobre AD, Cuartas LA, Soares JV, Hodnett MG, Tomasella J et al (2008) HAND, a new terrain descriptor using SRTM-DEM; map** terra-firme rainforest environments in Amazonia. Remote Sens Environ 112:3469–3481. https://doi.org/10.1016/j.rse.2008.03.018
Ribeiro AJ, Hopkins MJG, Vicentini A, Sothers CA, Costa MAS, Brito JM, et al (1999) Flora da Reserva Ducke. Guia de Identificação das plantas vasculares de uma floresta de terra-firme na Amazônia Central. Manaus, Amazonas, Brasil Great Britain
Rocha EX, Schietti J, Gerolamo CS, Burnham RJ, Nogueira A (2020) Higher rates of liana regeneration after canopy fall drives species abundance patterns in central Amazonia. J Ecol 108:1311–1321. https://doi.org/10.1111/1365-2745.13345
Rowe N, Isnard S, Speck T (2004) Diversity of mechanical architectures in climbing plants: an evolutionary perspective. J Plant Growth Regul 23:108–128. https://doi.org/10.1007/s00344-004-0044-0
Sack L, Scoffoni C (2013) Leaf venation: structure, function, development, evolution, ecology and applications in the past, present and future. New Phytol 198:983–1000. https://doi.org/10.1111/nph.12253
Sandel B, Goldstein LB, Kraft NJB, Okie GJ, Shuldman MI, Bongers F et al (2010) Contrasting trait responses in plant communities to experimental and geographic variation in precipitation. New Phytol 188:565–575. https://doi.org/10.1111/j.1469-8137.2010.03382.x
Schietti J, Emilio T, Rennó CD, Drucker DP, Costa FRC, Nogueira A et al (2014) Vertical distance from drainage drives floristic composition changes in an Amazonian rainforest. Plant Ecol Divers 7:241–253. https://doi.org/10.1080/17550874.2013.783642
Schmitt S, Hérault B, Ducouret E, Baranger A, Tysklind N, Heuertz M, et al (2020) Topography consistently drives intra- and inter-specific leaf trait variation within tree species complexes in a Neotropical forest. Oikos 129:1521–1530. https://doi.org/10.1111/oik.07488
Schnitzer AS (2005) A mechanistic explanation for global patterns of liana abundance and distribution. Am Nat 166:262–276. https://doi.org/10.1086/431250
Schnitzer SA (2018) Testing ecological theory with lianas. New Phytol 220:366–380. https://doi.org/10.1111/nph.15431
Schnitzer SA, Bongers F (2002) The ecology of lianas and their role in forests. Trends Ecol Evol 17:223–230. https://doi.org/10.1016/S0169-5347(02)02491-6
Schnitzer SA, Bongers F (2011) Large-scale changes in tropical forests: emerging patterns and putative mechanisms. Ecol Lett 14:397–406. https://doi.org/10.1111/j.1461-0248.2011.01590.x
Schnitzer SA, Carson WP (2010) Lianas suppress tree regeneration and diversity in treefall gaps. Ecol Lett 13:849–857. https://doi.org/10.1111/j.1461-0248.2010.01480.x
Schnitzer AS, Filippis DM, Visser M, Villegas SE, Camaña RR, Boris B et al (2021) Local canopy disturbance as an explanation for long-term increases in liana abundance. Ecol Lett 00:1–13. https://doi.org/10.1111/ele.13881
Silvertown J, Dodd M, Gowing DJ, Mountford JO (1999) Hydrologically defined niches reveal a basis for species richness in plant communities. Nature 400:61–63
Silvertown J, Araya Y, Gowing D (2015) Hydrological niches in terrestrial plant communities: a review. J Ecol 103:93–108. https://doi.org/10.1111/1365-2745.12332
Smith-Martin CM, Bastos CL, Lopez OR, Power J, Schnitzer AS (2019) Effects of dry-season irrigation on leaf physiology and biomass allocation in tropical lianas and trees. Ecology 100:e02827
Sombroek W (2000) Amazon landforms and soils in relation to biological diversity. Acta Amaz 30:81–81. https://doi.org/10.1590/1809-43922000301100
Spence RD, Wu H, Sharpe PJH, Clark KG (1986) Water stress effects on guard cell anatomy and the mechanical advantage of the epidermal cells. Plant Cell Environ 9:197–202. https://doi.org/10.1111/1365-3040.ep11611639
Suding KN, Lavorel S, Chapin FS, Cornelissen JHC, Diaz S, Garnier E et al (2008) Scaling environmental change through the community-level: a trait-based response-and-effect framework for plants. Glob Chang Biol 14:1125–1140. https://doi.org/10.1111/j.1365-2486.2008.01557.x
Toledo JJ, Magnusson WE, Castilho CV, Nascimento HEM (2011) How much variation in tree mortality is predicted by soil and topography in Central Amazonia? For Ecol Manage 262:331–338. https://doi.org/10.1016/j.foreco.2011.03.039
Toledo JJ, Castilho CV, Magnusson WE, Nascimento HEM (2017) Soil controls biomass and dynamics of an Amazonian forest through the shifting of species and traits. Braz J Bot 40:451–461. https://doi.org/10.1007/s40415-016-0351-2
Tomasella J, Hodnett MG, Cuartas LA, Nobre AD, Waterloo MJ, Oliveira SM (2008) The water balance of an Amazonia micro-catchment: the effect of interannual variability of rainfall on hydrological behaviour. Hydrol Process 13:2133–2147. https://doi.org/10.1002/hyp.6813
van der Sande MT, Poorter L, Schnitzer SA, Engelbrecht BMJ, Markesteijn L (2019) The hydraulic efficiency–safety trade-off differs between lianas and trees. Ecology 100(5):e02666. https://doi.org/10.1002/ecy.2666
Vivek P, Parthasarathy N (2014) Liana community and functional trait analysis in tropical dry evergreen forest of India. J Plant Ecol 8:501–512. https://doi.org/10.1093/jpe/rtu031
Vleut I, Pérez-Salicrup DR (2005) Lianas and their supporting plants in the understorey at Los Tuxtlas Mexico. J Trop Ecol 21(5):577–580. https://doi.org/10.1017/S0266467405002531
Wright IJ, Reich PB, Westoby M, Ackerly DD, Baruch Z, Bongers F et al (2004) The worldwide leaf economics spectrum. Nature 428:821–827. https://doi.org/10.1038/nature02403
Wright SJ, Kitajima K, Kraft NJB, Reich PB, Wright IJ, Bunker DE et al (2010) Functional traits and the growth-mortality tradeoff in tropical trees. Ecology 91:3664–3674. https://doi.org/10.1890/09-2335.1
Xu Z, Zhou G (2008) Responses of leaf stomatal density to water status and its relationship with photosynthesis in a grass. J Exp Bot 59:3317–3325. https://doi.org/10.1093/jxb/ern185
Yong T, Kitching R, Cao M (2012) Lianas as structural parasites: A re-evaluation. Chin Sci Bull 57:307–312. https://doi.org/10.1007/s11434-011-4690-x
Zhang J, Nieminen K, Serra JAA, Helariutta Y (2014) The formation of wood and its control. Plant Biol 17:56–63
Acknowledgements
We are grateful to José Francisco for lab support and guidance on anatomical measurements. Erick Lavado and Lorena Rincón provided statistical support and valuable suggestions on the discussion. ER thanks José Raimundo for hel** in the field. INPA provided transport, and the logistical structure at the Ducke Reserve was provided by PELD and the Brazilian Biodiversity Research Program (PPBio).
Funding
This contribution is part of the doctoral thesis of E.R undertaken at the Instituto Nacional de Pesquisas da Amazônia (INPA), with a fellowship from Coordination of Superior Level Staff Improvement (CAPES). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001. Financial support was received from the Brazilian Long Term Ecological Research Program (CNPq/PELD# 441282/0.2016–4) and Project CENBAM- INCT Centro de Biodiversidade Amazônica (FAPEAM/FDB/INPA#003/2012). Additional funds were provided to A.N. by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) through a PDE scholarship (234000/2014–7) and Research Grant (434692/2018–2), and by the São Paulo Research Foundation (FAPESP) through a Young Investigators Grant (2019/19544–7).
Author information
Authors and Affiliations
Contributions
EXR, AN, JS, RJB and FRCC conceived the ideas and methodological design. EXR, SCG and CFH collected the data. AN, EXR and RJB identified the plant species. EXR and CSG analyzed the data. All authors contributed to the writing and gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Not applicable for that section.
Consent for publication
Not applicable for that section.
Consent to participate
Not applicable for that section.
Additional information
Communicated by Jennifer Funk.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rocha, E.X., Nogueira, A., Costa, F.R.C. et al. Liana functional assembly along the hydrological gradient in Central Amazonia. Oecologia 200, 183–197 (2022). https://doi.org/10.1007/s00442-022-05258-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-022-05258-w