Abstract
Background
The tumor suppressor gene AT-rich interactive domain 1A (ARID1A) and systemic inflammatory response (SIR) have been reported to be related to the sensitivity to immunotherapy. This study intended to explore the relationship between ARID1A expression and SIR, and to further elucidate the prognostic value of ARID1A expression in gastric cancer (GC).
Methods
The mRNA and protein expression of ARID1A were detected in 272 formalin-fixed paraffin-embedded (FFPE) tumor tissues. The data of nine systemic inflammation markers were collected 1 week before gastrectomy. Univariate and multivariate COX analysis were used to screen out independent predictors of GC.
Results
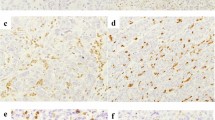
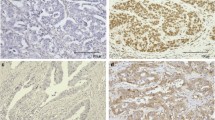
Negative expression of ARID1A protein was related to GC with deficient mismatch repair (dMMR) (p = 0.033), positive programmed cell death-ligand 1 (PD-L1) (p = 0.005) and lower albumin level (p = 0.0064). Low expression of ARID1A mRNA was common in GC with abnormal E-cadherin (p = 0.020) and a higher platelet/lymphocyte ratio (PLR) (p = 0.0391). Multivariate COX analysis showed that the expression of ARID1A protein (p = 0.023), age (p = 0.004), T stage (p = 0.009) and N stage (p = 0.009) were independent predictors of GC. The nomogram established by independent predictors can accurately evaluate the survival risk of patients with GC.
Conclusions
The loss of ARID1A protein expression was associated with the dMMR subtype and high expression of PD-L1 in GC. Negative ARID1A protein and low expression of mRNA were associated with aberrant systemic inflammatory markers. The expression of ARID1A protein had important prognostic significance in GC.





Similar content being viewed by others
Data availability
The data described in this manuscript are contained in published articles or available from the corresponding author upon reasonable request.
References
Ahn S, Lee S, Kim Y et al (2017) High-throughput protein and mRNA Expression-based classification of gastric cancers can identify clinically distinct subtypes, concordant with recent molecular classifications. Am J Surg Pathol 5:5. https://doi.org/10.1097/pas.0000000000000756
Chao J, Fuchs C, Shitara K et al (2021) Assessment of pembrolizumab therapy for the treatment of microsatellite instability-high gastric or gastroesophageal junction cancer among patients in the KEYNOTE-059, KEYNOTE-061, and KEYNOTE-062 clinical trials. JAMA Oncol. https://doi.org/10.1001/jamaoncol.2021.0275
Chen P, Roh W, Reuben A et al (2016) Analysis of immune signatures in longitudinal tumor samples yields insight into biomarkers of response and mechanisms of resistance to immune checkpoint blockade. Cancer Discov. https://doi.org/10.1158/2159-8290.Cd-15-1545
Cho J, Ahn S, Son DS et al (2019) Bridging genomics and phenomics of gastric carcinoma. Int J Cancer. https://doi.org/10.1002/ijc.32228
Cancer Genome Atlas Research Network (2014) Comprehensive molecular characterization of gastric adenocarcinoma. Nature. https://doi.org/10.1038/nature13480
Dharmapuri S, Özbek U, Lin J et al (2020) Predictive value of neutrophil to lymphocyte ratio and platelet to lymphocyte ratio in advanced hepatocellular carcinoma patients treated with anti-PD-1 therapy. Cancer Med. https://doi.org/10.1002/cam4.3135
Formica V, Morelli C, Patrikidou A et al (2020) Gastric Inflammatory Prognostic Index (GIPI) in patients with metastatic gastro-esophageal junction/gastric cancer treated with PD-1/PD-L1 immune checkpoint inhibitors. Target Oncol. https://doi.org/10.1007/s11523-020-00723-z
Fuchs C, Doi T, Jang R et al (2018a) Safety and efficacy of pembrolizumab monotherapy in patients with previously treated advanced gastric and gastroesophageal junction cancer: phase 2 Clinical KEYNOTE-059 trial. JAMA Oncol. https://doi.org/10.1001/jamaoncol.2018.0013
Fujimoto A, Totoki Y, Abe T et al (2012) Whole-genome sequencing of liver cancers identifies etiological influences on mutation patterns and recurrent mutations in chromatin regulators. Nat Genet. https://doi.org/10.1038/ng.2291
Graesslin O, Abdulkarim B, Coutant C et al (2010) Nomogram to predict subsequent brain metastasis in patients with metastatic breast cancer. J Clin Oncol. https://doi.org/10.1200/jco.2009.24.6314
Guichard C, Amaddeo G, Imbeaud S et al (2012) Integrated analysis of somatic mutations and focal copy-number changes identifies key genes and pathways in hepatocellular carcinoma. Nat Genet. https://doi.org/10.1038/ng.2256
Han D, Suh Y, Kong S et al (2012) Nomogram predicting long-term survival after d2 gastrectomy for gastric cancer. J Clin Oncol. https://doi.org/10.1200/jco.2012.41.8343
Hanahan D, Weinberg R (2011) Hallmarks of cancer: the next generation. Cell. https://doi.org/10.1016/j.cell.2011.02.013
Hanley J, McNeil B (1983) A method of comparing the areas under receiver operating characteristic curves derived from the same cases. Radiology. https://doi.org/10.1148/radiology.148.3.6878708
Janjigian Y, Shitara K, Moehler M et al (2021) First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet (london, England). https://doi.org/10.1016/s0140-6736(21)00797-2
Jiang T, Qiao M, Zhao C et al (2018) Pretreatment neutrophil-to-lymphocyte ratio is associated with outcome of advanced-stage cancer patients treated with immunotherapy: a meta-analysis. Cancer Immunol Immunother. https://doi.org/10.1007/s00262-018-2126-z
** S, Xu B, Yu L et al (2017) The PD-1, PD-L1 expression and CD3+ T cell infiltration in relation to outcome in advanced gastric signet-ring cell carcinoma, representing a potential biomarker for immunotherapy. Oncotarget. https://doi.org/10.18632/oncotarget.16407
Jones S, Wang TL, Shih Ie M et al (2010) Frequent mutations of chromatin remodeling gene ARID1A in ovarian clear cell carcinoma. Science. https://doi.org/10.1126/science.1196333
Jones S, Li M, Parsons D et al (2012) Somatic mutations in the chromatin remodeling gene ARID1A occur in several tumor types. Hum Mutat. https://doi.org/10.1002/humu.21633
Kang Y, Boku N, Satoh T et al (2017) Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet (london, England). https://doi.org/10.1016/s0140-6736(17)31827-5
Kim S, Ahn S, Lee J et al (2016) Value of FGFR2 expression for advanced gastric cancer patients receiving pazopanib plus CapeOX (capecitabine and oxaliplatin). J Cancer Res Clin Oncol. https://doi.org/10.1007/s00432-016-2143-2
Kim ST, Cristescu R, Bass AJ et al (2018) Comprehensive molecular characterization of clinical responses to PD-1 inhibition in metastatic gastric cancer. Nat Med. https://doi.org/10.1038/s41591-018-0101-z
Kim YB, Ahn JM, Bae WJ et al (2019) Functional loss of ARID1A is tightly associated with high PD-L1 expression in gastric cancer. Int J Cancer. https://doi.org/10.1002/ijc.32140
Kinjo T, Taniguchi H, Kushima R et al (2012) Histologic and immunohistochemical analyses of α-fetoprotein–producing cancer of the stomach. Am J Surg Pathol. https://doi.org/10.1097/PAS.0b013e31823aafec
Liang X, Vacher S, Boulai A et al (2018) Targeted next-generation sequencing identifies clinically relevant somatic mutations in a large cohort of inflammatory breast cancer. Breast Cancer Res. https://doi.org/10.1186/s13058-018-1007-x
Mamo A, Cavallone L, Tuzmen S et al (2012) An integrated genomic approach identifies ARID1A as a candidate tumor-suppressor gene in breast cancer. Oncogene. https://doi.org/10.1038/onc.2011.386
Mehrvarz Sarshekeh A, Alshenaifi J, Roszik J et al (2021) ARID1A mutation may define an immunologically active subgroup in patients with microsatellite stable colorectal cancer. Clin Cancer Res. https://doi.org/10.1158/1078-0432.Ccr-20-2404
Okamura R, Kato S, Lee S et al (2020) ARID1A alterations function as a biomarker for longer progression-free survival after anti-PD-1/PD-L1 immunotherapy. J Immunother Cancer. https://doi.org/10.1136/jitc-2019-000438
Rossi S, Toschi L, Finocchiaro G et al (2020) Neutrophil and lymphocyte blood count as potential predictive indicators of nivolumab efficacy in metastatic non-small-cell lung cancer. Immunotherapy. https://doi.org/10.2217/imt-2019-0154
Setia N, Agoston A, Han H et al (2016) A protein and mRNA expression-based classification of gastric cancer. Mod Pathol. https://doi.org/10.1038/modpathol.2016.55
Shen J, Ju Z, Zhao W et al (2018) ARID1A deficiency promotes mutability and potentiates therapeutic antitumor immunity unleashed by immune checkpoint blockade. Nat Med. https://doi.org/10.1038/s41591-018-0012-z
Shitara K, Ozguroglu M, Bang YJ et al (2018) Pembrolizumab versus paclitaxel for previously treated, advanced gastric or gastro-oesophageal junction cancer (KEYNOTE-061): a randomised, open-label, controlled, phase 3 trial. Lancet. https://doi.org/10.1016/S0140-6736(18)31257-1
Sung H, Ferlay J, Siegel R et al (2021) Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer J Clin. https://doi.org/10.3322/caac.21660
Tumeh P, Harview C, Yearley J et al (2014) PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature. https://doi.org/10.1038/nature13954
Wang F, Wei XL, Wang FH et al (2019) Safety, efficacy and tumor mutational burden as a biomarker of overall survival benefit in chemo-refractory gastric cancer treated with toripalimab, a PD-1 antibody in phase Ib/II clinical trial NCT02915432. Ann Oncol. https://doi.org/10.1093/annonc/mdz197
Wiegand K, Shah S, Al-Agha O et al (2010) ARID1A mutations in endometriosis-associated ovarian carcinomas. N Engl J Med. https://doi.org/10.1056/NEJMoa1008433
Wu RC, Wang TL, Shih Ie M (2014) The emerging roles of ARID1A in tumor suppression. Cancer Biol Ther. https://doi.org/10.4161/cbt.28411
Yan H, Wang X, Zhang Q et al (2014) Reduced expression of the chromatin remodeling gene ARID1A enhances gastric cancer cell migration and invasion via downregulation of E-cadherin transcription. Carcinogenesis. https://doi.org/10.1093/carcin/bgt398
Yang L, Wei S, Zhao R et al (2016) Loss of ARID1A expression predicts poor survival prognosis in gastric cancer: a systematic meta-analysis from 14 studies. Sci Rep. https://doi.org/10.1038/srep28919
Zhao R, Liu Y, Dai W et al (2021) A substage increase in the AJCC classification system improves prognostic prediction in stage III gastric cancer with insufficient lymph nodes removed. Front Oncol. https://doi.org/10.3389/fonc.2021.624413
Zhu Y, Sheng L, Wu J et al (2018) Loss of ARID1A expression is associated with poor prognosis in patients with gastric cancer. Hum Pathol. https://doi.org/10.1016/j.humpath.2018.04.003
Zou J, Qin W, Yang L et al (2020) Genetic alterations and expression characteristics of ARID1A impact tumor immune contexture and survival in early-onset gastric cancer. Am J Cancer Res 10:3947–3972
Acknowledgements
The authors would like to thank all patients who participated in this study.
Funding
This work was funded by grants from National Natural Science Foundation of China (82073382) and the Distinguished Young Scholars of Jiangsu Province (BK20190001). The funding sources had no role in the study design, data collection, data analysis, data interpretation, or writing of this review. The funding sources have no involvement in study design; the collection, analysis, and interpretation of data; in the writing of the report; or in the decision to submit the paper for publication.
Author information
Authors and Affiliations
Contributions
XW: drafting the article; KC: revising it critically for important intellectual content; TS: the acquisition of data; QL: Visualization; XX: analysis and interpretation of data; HW: analysis and interpretation of data; LY: the acquisition of data; BL: supervision and final approval of the version to be submitted; JW: conceptualization and funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical approval
This study was conducted in accordance with the code of ethics of the World Medical Association (Declaration of Helsinki) and approved by the Ethics Committee of Nan**g Drum Tower Hospital (No. 2016-196-01).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, X., Che, K., Shi, T. et al. Loss of ARID1A expression is associated with systemic inflammation markers and has important prognostic significance in gastric cancer. J Cancer Res Clin Oncol 148, 1583–1595 (2022). https://doi.org/10.1007/s00432-022-03971-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-03971-w