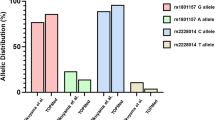
Abstract
Purpose
FOXP3 is a marker of the T regulatory (Treg) cell subset and drives its function and homeostasis. Its expression maintains the host immunosuppressive state that favors persistence of human papillomavirus (HPV) infection and squamous intraepithelial lesion (SIL) appearance. The present study evaluated the effects of the rs3761548 and rs2232365 intronic single-nucleotide variants (SNVs) and their haplotypes on HPV infection and SIL diagnosis in HPV-infected and -uninfected women.
Methods
HPV DNA-based detection in cervical specimens was performed by PCR. FOXP3 variants were genotyped by PCR-restriction fragment length polymorphism and haplotype recombination sites were inferred for 208 HPV-infected and 218 HPV-uninfected women diagnosed or not with low- or high-grade intraepithelial lesions of cervix. Case–control analyses were carried out by logistic regression adjusted for several socio-demographic, sexual lifestyle, and clinical data.
Results
The homozygous genotype of the rs3761548 variants (A/A) (related to decreased FOXP3 expression) may exert a protective role against HPV infection in women (ORAj: 0.60; 95% CI 0.36–0.99; p = 0.049) and was an independent predictor of protection against HSIL development (ORAdj: 0.28; 95% CI 0.11–0.68; p = 0.006). In addition, the homozygous genotype (G/G) of the rs2232365 variants (related to increased FOXP3 expression) was independently associated with the HPV infection (ORAdj: 2.10; 95% CI 1.06–4.15; p = 0.033). Haplotype analysis revealed no significant associations in our study.
Conclusions
Our results reveal the significant and independent associations between FOXP3 genetic variants and susceptibility to HPV infection and SIL diagnosis and their role as biomarkers of HPV infection and cervical lesion management.
Similar content being viewed by others
References
Banin-Hirata B, Guembarovski RL, Vitielo GAF et al (2017) FOXP3 Allelic variants and haplotype structures are associated with aggressive breast cancer subtypes. Dis Mark. https://doi.org/10.1155/2017/6359603
Bauer HM, Ting Y, Greer CE, Chambers JC, Tashiro CJ, Chimera J et al (1991) Genital human papillomavirus infection in female university students as determined by a PCR-based method. JAMA 265:472–477. https://doi.org/10.1001/jama.1991.03460040048027
Beiranvand E, Abediankenari S, Khani S, Hosseini HM, Zeinali S, Beiranvand B et al (2017) G allele at -924 A>G position of FoxP3 gene promoter as a risk factor for tuberculosis. BMC Infect Dis 17:4–9. https://doi.org/10.1186/s12879-017-2762-5
Castellsagué X, Muñoz N (2003) Chapter 3: cofactors in human papillomavirus carcinogenesis–role of parity, oral contraceptives, and tobacco smoking. J Natl Cancer Inst Monogr 2003:20–28
Chelimo C, Wouldes TA, Cameron LD, Elwood JM (2013) Risk factors for and prevention of human papillomaviruses (HPV), genital warts and cervical cancer. J Infect 66:207–217. https://doi.org/10.1016/j.**f.2012.10.024
Chen Y, Zhang H, Wang Y, **e H, Fei R, Wang X et al (2012) Association of FOXP3 gene polymorphism with chronic hepatitis B in Chinese population. Infect Int 1:191–195. https://doi.org/10.1186/1756-9966-32-39
Cheng Z, Guo Y, Ming L (2018) Functional Foxp3 polymorphisms and the susceptibility to cancer: an update meta-analysis. Medicine (Baltimore) 97:1–8. https://doi.org/10.1097/MD.0000000000011927
da Silva MC, Martins HPR, de Souza JL, Tognim MCB, Svidzinski TIE, Teixeira JJV et al (2012) Prevalence of HPV infection and genotypes in women with normal cervical cytology in the state of Paraná, Brazil. Arch Gynecol Obstet 286:1015–1022. https://doi.org/10.1007/s00404-012-2399-y
Ferlay J, Soerjomataram II, Dikshit R, Eser S, Mathers C, Rebelo M et al (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136:E356–E359. https://doi.org/10.1002/ijc.29210
Fontenot JD, Gavin MA, Rudensky AY (2003) Foxp3 programs the development and function of CD4+ CD25+ regulatory T cells. Nat Immunol 4:330–336. https://doi.org/10.1038/ni904
Forman D, De Martel C, Lacey CJ, Soerjomataram I, Lortet-Tieulent J, Bruni L et al (2012) Global burden of human papillomavirus and related diseases. Vaccine 30:F12–F23. https://doi.org/10.1016/j.vaccine.2012.07.055
He Y, Bo Q, Yong W, Qiu Z, Li Y, Li W (2013) FoxP3 genetic variants and risk of non-small cell lung cancer in the Chinese Han population. Gene 531:422–425. https://doi.org/10.1016/j.gene.2013.08.066
Jeon S, Lambert PF (1995) Integration of human papillomavirus type 16 DNA into the human genome leads to increased stability of E6 and E7 mRNAs: implications for cervical carcinogenesis. Proc Natl Acad Sci USA 92:1654–1658
Katoh H, Zheng P, Liu Y (2013) FOXP3: genetic and epigenetic implications for autoimmunity. J Autoimmun 41:72–78. https://doi.org/10.1016/j.jaut.2012.12.004
Kobayashi A, Weinberg V, Darragh T, Smith-McCune K (2008) Evolving immunosuppressive microenvironment during human cervical carcinogenesis. Mucosal Immunol 1:412–420. https://doi.org/10.1038/mi.2008.33
Koukouikila-Koussounda F, Ntoumi F, Ndounga M, Tong HV, Abena AA, Velavan TP (2013) Genetic evidence of regulatory gene variants of the STAT6, IL10R and FOXP3 locus as a susceptibility factor in uncomplicated malaria and parasitaemia in Congolese children. Malar J 12:1. https://doi.org/10.1186/1475-2875-12-9
Krishnan S, Dunbar MS, Minnis AM, Medlin CA, Gerdts CE, Padian NS (2008) Poverty, gender inequities, and women’s risk of human immunodeficiency virus/AIDS. Ann N Y Acad Sci 1136:101–110. https://doi.org/10.1196/annals.1425.013
Kwon T-K, Chung EJ, Lee N, Roh EY, Song EY (2017) Associations of FoxP3 gene polymorphisms with severe recurrent respiratory papillomatosis in Korean patients. J Otolaryngol Head Neck Surg 46:10–13. https://doi.org/10.1186/s40463-017-0197-z
Lins TC, Vieira RG, Abreu BS, Grattapaglia D, Pereira RW (2010) Genetic composition of Brazilian population samples based on a set of twenty eight ancestry informative SNPs. Am J Hum Biol 22:187–192. https://doi.org/10.1002/ajhb.20976
Lu L, Barbi J, Pan F (2017) The regulation of immune tolerance by FOXP3. Nat Rev Immunol 17:703–717. https://doi.org/10.1038/nri.2017.75
Machiela MJ, Chanock SJ (2015) LDlink: A web-based application for exploring population-specific haplotype structure and linking correlated alleles of possible functional variants. Bioinformatics 31:3555–3557. https://doi.org/10.1093/bioinformatics/btv402
Maruyama T, Li J, Vaque JP, Konkel JE, Wang W, Zhang B et al (2011) Control of regulatory T cell and Th17 cell differentiation by inhibitory helix-loop-helix protein Id3. Nat Immunol 12:86–95. https://doi.org/10.1038/ni.1965
Molling JW, De Gruijl TD, Glim J, Moreno M, Rozendaal L, Meijer CJLM et al (2007) CD4+ CD25hi regulatory T-cell frequency correlates with persistence of human papillomavirus type 16 and T helper cell responses in patients with cervical intraepithelial neoplasia. Int J Cancer 121:1749–1755. https://doi.org/10.1002/ijc.22894
Morshed K, Polz-Gruszka D, Szymañski M, Polz-Dacewicz M (2014) Human papillomavirus (HPV)—structure, epidemiology and pathogenesis. Otolaryngol Pol 68:213–219. https://doi.org/10.1016/j.otpol.2014.06.001
Mudvari P, Movassagh M, Kowsari K, Seyfi A, Kokkinaki M, Edwards NJ et al (2015) SNPlice: variants that modulate Intron retention from RNA-sequencing data. Bioinformatics 31:1191–1198. https://doi.org/10.1093/bioinformatics/btu804
Müller S, Poehnert D, Müller JA, Scheumann GWF, Koch M, Lück R (2010) Regulatory T cells in peripheral blood, lymph node, and thyroid tissue in patients with medullary thyroid carcinoma. World J Surg 34:1481–1487. https://doi.org/10.1007/s00268-010-0484-6
Oda JMM, Hirata BKB, Guembarovski RL, Watanabe MAE (2013) Genetic polymorphism in FOXP3 gene: imbalance in regulatory T-cell role and development of human diseases. J Genet 92:163–171
Okuyama NCM, Cezar-dos-Santos F, Pereira ÉR, Trugilo KP, Cebinelli GCM, Sena MM et al (2018) Genetic variant in CXCL12 gene raises susceptibility to HPV infection and squamous intraepithelial lesions development: a case-control study. J Biomed Sci 25:1–10. https://doi.org/10.1186/s12929-018-0472-y
Paradowska-Gorycka A, Jurkowska M, Felis-Giemza A, Romanowska-Prochnicka K, Manczak M, Maslinski S et al (2015) Genetic polymorphisms of Foxp3 in patients with rheumatoid arthritis. J Rheumatol 42:170–180. https://doi.org/10.3899/jrheum.131381
Park K, Cho KJ, Lee M, Yoon DH, Kim S-B (2013) Importance of FOXP3 in prognosis and its relationship with p16 in tonsillar squamous cell carcinoma. Anticancer Res 33:5667–5674
Passerini L, Santoni De Sio FR, Roncarolo MG, Bacchetta R (2014) Forkhead box P3: the peacekeeper of the immune system. Int Rev Immunol 33:129–145. https://doi.org/10.3109/08830185.2013.863303
Santiago E, Camacho L, Junquera ML, Vázquez F (2006) Full HPV ty** by a single restriction enzyme. J Clin Virol 37:38–46. https://doi.org/10.1016/j.jcv.2006.06.001
Sasagawa T, Takagi H, Makinoda S (2012) Immune responses against human papillomavirus (HPV) infection and evasion of host defense in cervical cancer. J Infect Chemother 18:807–815. https://doi.org/10.1007/s10156-012-0485-5
Shen Z, Chen L, Hao F, Wang G, Fan P, Liu Y (2010) Intron-1 rs3761548 is related to the defective transcription of Foxp3 in psoriasis through abrogating E47/c-Myb binding. J Cell Mol Med 14:226–241. https://doi.org/10.1111/j.1582-4934.2008.00370.x
Solé X, Guinó E, Valls J, Iniesta R, Moreno V (2006) SNPStats: a web tool for the analysis of association studies. Bioinformatics 22:1928–1929. https://doi.org/10.1093/bioinformatics/btl268
Solomon D, Davey D, Kurman R, Moriarty A, Connor DO, Raab S et al (2002) The 2001 Bethesda System: terminology for reporting results of cervical cytology. JAMA 287:2114–2119. https://doi.org/10.1001/jama.287.16.2114
Stephens M, Scheet P (2005) Accounting for decay of linkage disequilibrium in haplotype inference and missing-data imputation. Am J Hum Genet 76:449–462. https://doi.org/10.1086/428594
Stephens M, Smith NJ, Donnelly P (2001) A new statistical method for haplotype reconstruction from population data. Am J Hum Genet 68:978–989. https://doi.org/10.1086/319501
Trugilo KP, Cebinelli GCM, Berti FCB, Okuyama NCM, Cezar-dos-Santos F, Sena MM et al (2018) Polymorphisms in the TGFB1 signal peptide influence human papillomavirus infection and development of cervical lesions. Med Microbiol Immunol 208:49–58. https://doi.org/10.1007/s00430-018-0557-y
Waggoner SE (2003) Cervical cancer. Lancet 361:2217–2225. https://doi.org/10.1016/S0140-6736(03)13778-6
Walboomers JMM, Jacobs MV, Manos MM, Bosch FX, Kummer JA, Shah KV et al (1999) Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J Pathol 189:12–19. https://doi.org/10.1002/(SICI)1096-9896(199909)189:1%3c12:AID-PATH431%3e3.0.CO;2-F
Wang L, Liu R, Li W, Chen C, Katoh H, Chen GY et al (2009) Somatic single hits inactivate the X-linked tumor suppressor FOXP3 in the prostate. Cancer Cell 16:336–346. https://doi.org/10.1016/j.ccr.2009.08.016
Wang L, Liu R, Ribick M, Zheng P, Liu Y (2010) FOXP3 as X-linked tumor suppressor. Discov Med 10:322. https://doi.org/10.1002/ijc.22894
Wu Z, You Z, Zhang C, Li Z, Su X, Zhang X et al (2012) Association between functional polymorphisms of foxp3 gene and the occurrence of unexplained recurrent spontaneous abortion in a Chinese Han population. Clin Dev Immunol 2012:7. https://doi.org/10.1155/2012/896458
You D, Wang Y, Zhang Y, Li Q, Yu X, Yuan M et al (2018) Association of Foxp3 promoter polymorphisms with susceptibility to endometrial cancer in the Chinese Han women. Medicine (Baltimore). 97(18):e0582. https://doi.org/10.1097/MD.0000000000010582
Zeng C, Yao Y, Jie W, Zhang M, Hu X, Zhao Y et al (2013) Up-regulation of Foxp3 participates in progression of cervical cancer. Cancer Immunol Immunother 62:481–487. https://doi.org/10.1007/s00262-012-1348-8
Zuo T, Wang L, Morrison C, Chang X, Zhang H, Li W et al (2007) FOXP3 is an X-Linked breast cancer suppressor gene and an important repressor of the HER-2/ErbB2 oncogene. Cell 129:1275–1286. https://doi.org/10.1016/j.cell.2007.04.034
Acknowledgements
The authors would like to thank the women who made this study possible, Intermunicipal Consortium of Health of the Middle Paranapanema (Cismepar), University Hospital and Clinic Center of State University of Londrina, Municipal Health Department of Londrina—PR, Brazil, and nurses of these health services for their technical assistance with patient screening and cervical sample collection.
Funding
This work was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (470137/2013-4), Fundação Araucária—Programa Pesquisa para o SUS (34935.406.36850.19112012). Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001, and financial support for language editing from FAEPE/UEL-PUBLIC 2018.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (Institutional Ethics Committee Involving Humans of the State University of Londrina, Londrina, PR, Brazil, CEP/UEL 133/2012, CAAE 05505912.0.0000.5231) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cezar-dos-Santos, F., Ferreira, R.S., Okuyama, N.C.M. et al. FOXP3 immunoregulatory gene variants are independent predictors of human papillomavirus infection and cervical cancer precursor lesions. J Cancer Res Clin Oncol 145, 2013–2025 (2019). https://doi.org/10.1007/s00432-019-02951-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-019-02951-x