Abstract
Purpose
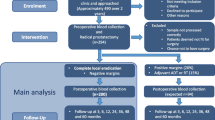
The detection of circulating tumor cells (CTCs) provides important prognostic information in men with metastatic prostate cancer. We aim to determine the rate of detection of CTCs in patients with high-risk non-metastatic prostate cancer using the CellSearch® method.
Method
Samples of peripheral blood (7.5 mL) were drawn from 36 men with newly diagnosed high-risk non-metastatic prostate cancer, prior to any initiation of therapy and analyzed for CTCs using the CellSearch® method.
Results
The median age was 70 years, median PSA was 14.1, and the median Gleason score was 9. The median 5-year risk of progression of disease using a validated nomogram was 39 %. Five out of 36 patients (14 %, 95 % CI 5–30 %) had CTCs detected in their circulation. Four patients had only 1 CTC per 7.5 mL of blood detected. One patient had 3 CTCs per 7.5 mL of blood detected, which included a circulating tumor microemboli. Both on univariate analysis and multivariate analysis, there were no correlations found between CTC positivity and the classic prognostic factors including PSA, Gleason score, T-stage and age.
Conclusion
This study demonstrates that patients with high-risk, non-metastatic prostate cancer present infrequently with small number of CTCs in peripheral blood. This finding is consistent with the limited literature available in this setting. Other CTC isolation and detection technologies with improved sensitivity and specificity may enable detection of CTCs with mesenchymal phenotypes, although none as yet have been validated for clinical use. Newer assays are emerging for detection of new putative biomarkers for prostate cancer. Correlation of disease control outcomes with CTC detection will be important.

Similar content being viewed by others
References
Attard G et al (2009) Characterization of ERG, AR and PTEN gene status in circulating tumor cells from patients with castration-resistant prostate cancer. Cancer Res 69:2912–2918. doi:10.1158/0008-5472.CAN-08-3667
Bastian PJ et al (2007) Prognostic value of preoperative serum cell-free circulating DNA in men with prostate cancer undergoing radical prostatectomy. Clin Cancer Res 13:5361–5367. doi:10.1158/1078-0432.CCR-06-2781
Cohen SJ et al (2008) Relationship of circulating tumor cells to tumor response, progression-free survival, and overall survival in patients with metastatic colorectal cancer. J Clin Oncol 26:3213–3221. doi:10.1200/JCO.2007.15.8923
Cristofanilli M et al (2004) Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med 351:781–791. doi:10.1056/NEJMoa040766
Danila DC et al (2007) Circulating tumor cell number and prognosis in progressive castration-resistant prostate cancer. Clin Cancer Res 13:7053–7058. doi:10.1158/1078-0432.CCR-07-1506
Darshan MS et al (2011) Taxane-induced blockade to nuclear accumulation of the androgen receptor predicts clinical responses in metastatic prostate cancer. Cancer Res 71:6019–6029. doi:10.1158/0008-5472.CAN-11-1417
Davis JW et al (2008) Circulating tumor cells in peripheral blood samples from patients with increased serum prostate specific antigen: initial results in early prostate cancer. J Urol 179:2187–2191. doi:10.1016/j.juro.2008.01.102 discussion 2191
de Bono JS et al (2008) Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res 14:6302–6309. doi:10.1158/1078-0432.CCR-08-0872
Even-Sapir E, Metser U, Mishani E, Lievshitz G, Lerman H, Leibovitch I (2006) The detection of bone metastases in patients with high-risk prostate cancer: 99 mTc-MDP Planar bone scintigraphy, single- and multi-field-of-view SPECT, 18F-fluoride PET, and 18F-fluoride PET/CT. J Nucl Med 47:287–297
Frisch SM, Francis H (1994) Disruption of epithelial cell-matrix interactions induces apoptosis. J Cell Biol 124:619–626
Hou JM et al (2012) Clinical significance and molecular characteristics of circulating tumor cells and circulating tumor microemboli in patients with small-cell lung cancer. J Clin Oncol 30:525–532. doi:10.1200/JCO.2010.33.3716
Kats-Ugurlu G et al (2009) Circulating tumour tissue fragments in patients with pulmonary metastasis of clear cell renal cell carcinoma. J Pathol 219:287–293. doi:10.1002/path.2613
Lecouvet FE et al (2012) Can whole-body magnetic resonance imaging with diffusion-weighted imaging replace Tc 99m bone scanning and computed tomography for single-step detection of metastases in patients with high-risk prostate cancer? Eur Urol 62:68–75. doi:10.1016/j.eururo.2012.02.020
Lilleby W, Stensvold A, Mills IG, Nesland JM (2013) Disseminated tumor cells and their prognostic significance in nonmetastatic prostate cancer patients. Int J Cancer 133:149–155. doi:10.1002/ijc.28002
Liotta LA, Saidel MG, Kleinerman J (1976) The significance of hematogenous tumor cell clumps in the metastatic process. Cancer Res 36:889–894
Ma X et al (2014) Prognostic role of circulating tumor cells and disseminated tumor cells in patients with prostate cancer: a systematic review and meta-analysis. Tumour Biol. doi:10.1007/s13277-014-1731-5
Molnar B, Ladanyi A, Tanko L, Sreter L, Tulassay Z (2001) Circulating tumor cell clusters in the peripheral blood of colorectal cancer patients. Clin Cancer Res 7:4080–4085
Resel Folkersma L, San Jose Manso L, Galante Romo I, Moreno Sierra J, Olivier Gomez C (2012) Prognostic significance of circulating tumor cell count in patients with metastatic hormone-sensitive prostate cancer. Urology 80:1328–1332. doi:10.1016/j.urology.2012.09.001
Schwarzenbach H, Alix-Panabieres C, Muller I, Letang N, Vendrell JP, Rebillard X, Pantel K (2009) Cell-free tumor DNA in blood plasma as a marker for circulating tumor cells in prostate cancer. Clin Cancer Res 15:1032–1038. doi:10.1158/1078-0432.CCR-08-1910
Selth LA et al (2013) Circulating microRNAs predict biochemical recurrence in prostate cancer patients. Br J Cancer 109:641–650. doi:10.1038/bjc.2013.369
Spizzo G et al (2011) EpCAM expression in primary tumour tissues and metastases: an immunohistochemical analysis. J Clin Pathol 64:415–420. doi:10.1136/jcp.2011.090274
Stott SL et al (2010) Isolation and characterization of circulating tumor cells from patients with localized and metastatic prostate cancer. Sci Transl Med 2:25ra23. doi:10.1126/scitranslmed.3000403
Thalgott M et al (2013) Detection of circulating tumor cells in different stages of prostate cancer. J Cancer Res Clin Oncol 139:755–763. doi:10.1007/s00432-013-1377-5
Wang MC, Valenzuela LA, Murphy GP, Chu TM (1979) Purification of a human prostate specific antigen. Invest Urol 17:159–163
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Loh, J., Jovanovic, L., Lehman, M. et al. Circulating tumor cell detection in high-risk non-metastatic prostate cancer. J Cancer Res Clin Oncol 140, 2157–2162 (2014). https://doi.org/10.1007/s00432-014-1775-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-014-1775-3