Abstract
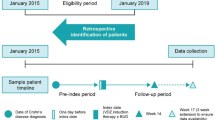
Ustekinumab is an effective therapy for adult Crohn’s disease (CD), but data in paediatric CD patients are scarce. The aim of the study was to describe the real-life effectiveness and safety of ustekinumab in paediatric CD. This is a multicentre review of children with Crohn's disease treated with ustekinumab. The aim of our study was to describe the effectiveness and safety of ustekinumab in paediatric real-life practice. This is a study of the Paediatric IBD (inflammatory bowel disease) Porto group of ESPGHAN. Corticosteroid (CS)- and exclusive enteral nutrition (EEN)-free remission, defined as weighted Paediatric Crohn’s Disease Activity Index (wPCDAI) < 12.5, and physician global assessment (PGA) were determined at weeks 12 and 52. A total of 101 children were included at a median age of 15.4 years (IQR 12.7–17.2) with a median follow-up of 7.4 months (IQR 5.6–11.8). Ninety-nine percent had received prior anti-TNF, 63% ≥ 2 anti-TNFα therapies and 22% vedolizumab. Baseline median wPCDAI was 39 (IQR 25–57.5) (71 (70%) patients with moderate-severe activity). Weeks 12 and 52 CS- and EEN-free remission were both 40.5%. Clinical response at week 6, iv induction route and older age at onset of ustekinumab treatment were predictive factors associated with clinical remission at week 12. Seven minor adverse events probably related to ustekinumab were reported. One patient died from an unrelated cause.
Conclusion: Our results suggest that ustekinumab is effective and safe in children with chronically active or refractory CD.
What is Known: • Ustekinumab is an effective therapy for adult moderate to severe Crohn’s disease (CD). • Off-label use of ustekinumab in children is increasing especially in anti-TNF refractory CD. | |
What is New: • Is the largest cohort of real-world use of ustekinumab in paediatric CD to date. • Clinical response at week 6, iv induction and older age at onset of ustekinumab were predictive factors associated with clinical response at week 12. |




Similar content being viewed by others
Data availability
No datasets were generated or analyzed during the current study.
References
Argollo MC, Allocca M, Furfaro F, Peyrin-Biroulet L, Danese S (2019) Interleukin-23 blockers: born to be first-line biologic agents in inflammatory bowel disease? Curr Pharm Des 25(1):25–31
Hanauer SB, Sandborn WJ, Feagan BG, Gasink C, Jacobstein D, Zou B et al (2020) IM-UNITI: three-year efficacy, safety, and immunogenicity of ustekinumab treatment of Crohn’s disease. J Crohns Colitis 14(1):23–32
Sands BE, Sandborn WJ, Panaccione R, O’Brien CD, Zhang H, Johanns J et al (2019) Ustekinumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med 381(13):1201–1214
Bishop C, Simon H, Suskind D, Lee D, Wahbeh G (2016) Ustekinumab in pediatric Crohn disease patients. J Pediatr Gastroenterol Nutr 63(3):348–351
Cameron FL, Garrick V, Russell RK (2016) Ustekinumab in treatment of refractory paediatric Crohn disease. J Pediatr Gastroenterol Nutr 62(3):e30
Rinawi F, Rosenbach Y, Assa A, Shamir R (2016) Ustekinumab for resistant pediatric Crohn disease. J Pediatr Gastroenterol Nutr 62(4):e34–e35
Fusillo SJ, Chang V, Stein RE, Maxwell EC, Conrad MA, Albenberg L et al (2018) 329 - Ustekinumab responders versus non-responders in refractory pediatric inflammatory bowel disease. Gastroenterology 154(6):S–82
Chavannes M, Martinez-Vinson C, Hart L, Kaniki N, Chao CY, Lawrence S et al (2019) Management of paediatric patients with medically refractory Crohn’s disease using ustekinumab: a multi-centred cohort study. J Crohns Colitis 13(5):578–584
Dayan JR, Dolinger M, Benkov K, Dunkin D, Jossen J, Lai J et al (2019) Real world experience with ustekinumab in children and young adults at a tertiary care pediatric inflammatory bowel disease center. J Pediatr Gastroenterol Nutr 69(1):61–67
Do P, Andersen J, Patel A, Semrin G, Sifuentes-Dominguez L, Luu P et al (2021) Augmented ustekinumab dosing is needed to achieve clinical response in patients with anti-TNF refractory pediatric Crohn’s disease: a retrospective chart review. F1000Research 9:316
Dolinger MT, Spencer EA, Lai J, Dunkin D, Dubinsky MC (2021) Dual biologic and small molecule therapy for the treatment of refractory pediatric inflammatory bowel disease. Inflamm Bowel Dis 27(8):1210–1214
Kim FS, Patel PV, Stekol E, Ali S, Hamandi H, Heyman MB et al (2021) Experience using ustekinumab in pediatric patients with medically refractory Crohn disease. J Pediatr Gastroenterol Nutr 73(5):610–614
Koudsi M, Martinez-Vinson C, Pigneur B, Willot S, Djamal D, Enaud R et al (2023) Ustekinumab use in pediatric inflammatory bowel disease: a French multicenter study from the pediatric GETAID. J Pediatr Gastroenterol Nutr 76(6):763–770
Rosh JR, Turner D, Griffiths A, Cohen SA, Jacobstein D, Adedokun OJ et al (2021) Ustekinumab in paediatric patients with moderately to severely active Crohn’s disease: pharmacokinetics, safety, and efficacy results from UniStar, a Phase 1 study. J Crohns Colitis 15(11):1931–1942
Turner D, Ricciuto A, Lewis A, D’Amico F, Dhaliwal J, Griffiths AM et al (2021) STRIDE-II: an update on the Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE) initiative of the International Organization for the study of IBD (IOIBD): determining therapeutic goals for treat-to-target strategies in IBD. Gastroenterology 160(5):1570–1583
Yerushalmy-Feler A, Pujol-Muncunill G, Martin-de-Carpi J, Kolho KL, Levine A, Olbjørn C et al (2022) Safety and potential efficacy of escalating dose of ustekinumab in pediatric Crohn disease (the speed-up study): a multicenter study from the Pediatric IBD Porto group of ESPGHAN. J Pediatr Gastroenterol Nutr 75(6):717–723
Takeuchi I, Arai K, Kyodo R, Sato T, Tokita K, Hirano Y et al (2021) Ustekinumab for children and adolescents with inflammatory bowel disease at a tertiary children’s hospital in Japan. J Gastroenterol Hepatol 36(1):125–130
Levine A, Koletzko S, Turner D, Escher JC, Cucchiara S, de Ridder L et al (2014) ESPGHAN revised Porto criteria for the diagnosis of inflammatory bowel disease in children and adolescents. J Pediatr Gastroenterol Nutr 58(6):795–806
Turner D, Levine A, Walters TD, Focht G, Otley A, Lopez VN et al (2017) Which PCDAI version best reflects intestinal inflammation in pediatric Crohn disease? J Pediatr Gastroenterol Nutr 64(2):254–260
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG (2009) Research electronic data capture (REDCap)–a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 42(2):377–381
Van Rheenen PF, Aloi M, Assa A, Bronsky J, Escher JC, Fagerberg UL et al (2021) The medical management of paediatric Crohn’s disease: an ECCO-ESPGHAN guideline update. J Crohns Colitis 15(2):171–194
Fang S, Zhang S, Zhang C, Wang L (2023) Effectiveness and safety of ustekinumab for pediatric inflammatory bowel disease: a systematic review. Pediatr Drugs 25(5):499–513
Cohen A, Ahmed N, Sant’Anna A, (2021) Ustekinumab for the treatment of refractory pediatric Crohn’s disease: a single-center experience. Intest Res 19(2):217–24
Feagan BG, Sandborn WJ, Gasink C, Jacobstein D, Lang Y, Friedman JR et al (2016) Ustekinumab as induction and maintenance therapy for Crohn’s disease. N Engl J Med 375(20):1946–1960
Ma C, Fedorak RN, Kaplan GG, Dieleman LA, Devlin SM, Stern N et al (2017) Clinical, endoscopic and radiographic outcomes with ustekinumab in medically-refractory Crohn’s disease: real world experience from a multicentre cohort. Aliment Pharmacol Ther 45(9):1232–1243
Harris KA, Horst S, Gadani A, Nohl A, Annis K, Duley C et al (2016) Patients with refractory Crohn’s disease successfully treated with ustekinumab. Inflamm Bowel Dis 22(2):397–401
Alric H, Amiot A, Kirchgesner J, Tréton X, Allez M, Bouhnik Y et al (2020) The effectiveness of either ustekinumab or vedolizumab in 239 patients with Crohn’s disease refractory to anti-tumour necrosis factor. Aliment Pharmacol Ther 51(10):948–957
Greenup AJ, Rosenfeld G, Bressler B (2017) Ustekinumab use in Crohn’s disease: a Canadian tertiary care centre experience. Scand J Gastroenterol 52(12):1354–1359
Penagini F, Lonoce L, Abbattista L, Silvera V, Rendo G, Cococcioni L et al (2023) Dual biological therapy and small molecules in pediatric inflammatory bowel disease. Pharmacol Res 196:106935
Author information
Authors and Affiliations
Contributions
GPM, JMC and VMNL designed the data collection instruments, coordinated and supervised data collection, conducted the analyses, drafted the initial manuscript and reviewed and edited the manuscript.VMN-L, JM, GP, JD and RM-M conducted the analyses, drafted the initial manuscript and reviewed and edited the manuscript. All authors reviewed and edited the manuscript and approved the final manuscript as submitted and agree to be accountable for all aspects of the work. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interest
GPM: speakers fees and congress support from Abbvie, Nestlé Health Science, Janssen. VNL: consultant fees from Abbvie, Nestlé Health Science, Otsuka, Pfizer. DT: consultant fee, research grant, royalties, or honorarium from Janssen, Pfizer, Hospital for Sick Children, Ferring, Abbvie, Takeda, Atlantic Health, Shire, Celgene, Lilly, Roche, ThermoFisher, BMS. KLK: consultant fees from Abbvie, Biocodex, Ferring, Tillots Pharma, MSD. AL: Nestlé Health Science, Janssen, Abbvie, Takeda and Megapharm. JB: honoraria, speakers fees and congress support from Abbvie, MSD, Nestlé Health Science, Nutricia, Biocodex, Ferring and Walmark. AA: consultant and lectures fees from Abbvie. Research grants from Abbvie and Janssen. FK: educational Grant from Dr. Falk, Abbvie. DCW: honoraria, speakers fees and/or congress support from Abbvie, Nestlé Health Science, Ferring, Roche, and Predictimmune. Abbvie and Janssen. LC: educational Grant from Dr. Falk. LR: Shire, Malinckrodt, Nestlé, Celltrion, Abbvie, Pfizer. Grant from ZonMw, ECCO and Pfizer. JMC: consultation fees, research grants, or honoraria from Abbvie, MSD, Janssen, Adacyte, Otsuka, Celltrion, Roche, Celgene, Dr. Falk, Nestlé HS, Ferring, FAES, Kern Pharma. The remaining authors have no competing interest.
Additional information
Communicated by Peter de Winter
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pujol-Muncunill, G., Navas-López, V.M., Ledder, O. et al. STEP-CD study: ustekinumab use in paediatric Crohn’s disease—a multicentre retrospective study from paediatric IBD Porto group of ESPGHAN. Eur J Pediatr (2024). https://doi.org/10.1007/s00431-024-05588-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00431-024-05588-2