Abstract
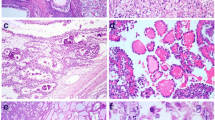
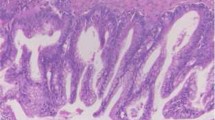
We found some clear cell glands appeared in the endometrioid cysts (ECs) of the ovary (EC-CCG). To explore the clinicopathological features, molecular biological changes, and prognosis in EC-CCG and analyze the association with ovarian clear cell borderline tumors (CCBT) and clear cell carcinoma (CCC). We retrospectively examined 35 cases of EC-CCG, compared them to 13 cases of clear cell cystadenomas, 14 cases of CCBT, and 49 cases of CCC. We analyzed the differences in clinicopathological features and prognosis between the four groups. Data on clinicopathology and survival were gathered. Immunohistochemistry (IHC) was performed in all cases, and we analyzed the molecular changes of 2 cases of EC-CCG and 1 case of CCC by whole-exome sequencing (WES). EC-CCG shared some common clinicopathological features with CCBT: they occurred before menopause, had an elevated serum CA125 level in some cases, had an ovarian cystic mass on B-ultrasound, and had a risk of recurrence. Microscopically, both diseases were based on typical EC, and clear cell glands in the EC cyst wall were seen in varying numbers. Some cases of EC-CCG had IHC results similar to those of CCBT and CCC, with positive expression of HNF1β and NapsinA; decreased expression of ER, PR, and ARID1A; and increased expression of Ki67 (> 5%). WES results revealed that EC-CCG had mutations in TP53BP1, ZNF462, FN1, and FTL (which was also mutated in CCC). In summary, we found that clear cell glands appearing around EC in the ovary have an association with CCC.







Similar content being viewed by others
Data availability
All data included in this study are available upon request by contact with the corresponding author.
References
Anglesio MS et al (2017) Cancer-associated mutations in endometriosis without cancer. N Engl J Med 376:1835–1848. https://doi.org/10.1056/NEJMoa1614814
Murakami K et al (2020) Endometriosis-associated ovarian cancer occurs early during follow-up of endometrial cysts. Int J Clin Oncol 25:51–58. https://doi.org/10.1007/s10147-019-01536-5
Ogawa S et al (2000) Ovarian endometriosis associated with ovarian carcinoma: a clinicopathological and immunohistochemical study. Gynecol Oncol 77:298–304
Kobayashi H et al (2009) Molecular pathogenesis of endometriosis-associated clear cell carcinoma of the ovary (review). Oncol Rep 22:233–240
Suryawanshi S et al (2013) Plasma microRNAs as novel biomarkers for endometriosis and endometriosis-associated ovarian cancer. Clin Cancer Res 19:1213–1224. https://doi.org/10.1158/1078-0432.CCR-12-2726
Muraoka A et al (2021) Impact of perioperative use of GnRH agonist or dienogest on ovarian reserve after cystectomy for endometriomas: a randomized controlled trial. Reprod Biol Endocrinol 19:179. https://doi.org/10.1186/s12958-021-00866-2
Roth LM, Langley FA, Fox H, Wheeler JE, Czernobilsky B (1984) Ovarian clear cell adenofibromatous tumors. Benign, of low malignant potential, and associated with invasive clear cell carcinoma. Cancer 53:1156–1163
Broi MGD, Ferriani RA, Navarro PA (2019) Ethiopathogenic mechanisms of endometriosis-related infertility. JBRA Assist Reprod 23:273–280. https://doi.org/10.5935/1518-0557.20190029
Guo S-W (2020) Cancer-associated mutations in endometriosis: shedding light on the pathogenesis and pathophysiology. Hum Reprod Update 26:423–449. https://doi.org/10.1093/humupd/dmz047
Herreros-Villanueva M, Chen C-C, Tsai E-M, Er T-K (2019) Endometriosis-associated ovarian cancer: what have we learned so far? Clin Chim Acta 493:63–72. https://doi.org/10.1016/j.cca.2019.02.016
Králíčková M, Laganà AS, Ghezzi F and Vetvicka V (2020) Endometriosis and risk of ovarian cancer: what do we know? Arch Gynecol Obstet 301. https://doi.org/10.1007/s00404-019-05358-8
Bulun SE, Wan Y, Matei D (2019) Epithelial mutations in endometriosis: link to ovarian cancer. Endocrinology 160:626–638. https://doi.org/10.1210/en.2018-00794
Wiedemeyer K et al (2022) Prognostic and theranostic biomarkers in ovarian clear cell carcinoma. Int J Gynecol Pathol 41:168–179. https://doi.org/10.1097/PGP.0000000000000780
Hariri N, Qarmali M, Fadare O (2018) Endometrial serous carcinoma with clear-cell change: frequency and immunohistochemical analysis. Int J Surg Pathol 26:126–134. https://doi.org/10.1177/1066896917731862
Fadare O, Parkash V (2019) Pathology of endometrioid and clear cell carcinoma of the ovary. Surg Pathol Clin 12:529–564. https://doi.org/10.1016/j.path.2019.01.009
Lheureux S et al (2018) A Clinical and molecular phase II trial of oral ENMD-2076 in ovarian clear cell carcinoma (OCCC): a study of the Princess Margaret phase II consortium. Clin Cancer Res 24:6168–6174. https://doi.org/10.1158/1078-0432.CCR-18-1244
Cuella-Martin R et al (2016) 53BP1 integrates DNA repair and p53-dependent cell fate decisions via distinct mechanisms. Mol Cell 64:51–64. https://doi.org/10.1016/j.molcel.2016.08.002
Pal T, Permuth-Wey J, Kumar A, Sellers TA (2008) Systematic review and meta-analysis of ovarian cancers: estimation of microsatellite-high frequency and characterization of mismatch repair deficient tumor histology. Clin Cancer Res 14:6847–6854. https://doi.org/10.1158/1078-0432.CCR-08-1387
Ali-Fehmi R et al (2006) Patterns of loss of heterozygosity at 10q23.3 and microsatellite instability in endometriosis, atypical endometriosis, and ovarian carcinoma arising in association with endometriosis. Int J Gynecol Pathol 25:223–229
Yeon SY et al (2018) Frameshift mutations in repeat sequences of ANK3, HACD4, TCP10L, TP53BP1, MFN1, LCMT2, RNMT, TRMT6, METTL8 and METTL16 genes in colon cancers. Pathol Oncol Res 24:617–622. https://doi.org/10.1007/s12253-017-0287-2
Hurley RM et al (2019) 53BP1 as a potential predictor of response in PARP inhibitor-treated homologous recombination-deficient ovarian cancer. Gynecol Oncol 153:127–134. https://doi.org/10.1016/j.ygyno.2019.01.015
Kruszka P et al (2019) Phenotype delineation of ZNF462 related syndrome. Am J Med Genet A 179:2075–2082. https://doi.org/10.1002/ajmg.a.61306
Matalliotaki C et al (2019) Role of FN1 and GREB1 gene polymorphisms in endometriosis. Mol Med Rep 20:111–116. https://doi.org/10.3892/mmr.2019.10247
Matalliotakis M et al (2017) The role of gene polymorphisms in endometriosis. Mol Med Rep 16:5881–5886. https://doi.org/10.3892/mmr.2017.7398
Lou X et al (2013) SOX2 targets fibronectin 1 to promote cell migration and invasion in ovarian cancer: new molecular leads for therapeutic intervention. OMICS 17:510–518. https://doi.org/10.1089/omi.2013.0058
Bertoli S et al (2019) Ferritin heavy/light chain (FTH1/FTL) expression, serum ferritin levels, and their functional as well as prognostic roles in acute myeloid leukemia. Eur J Haematol 102:131–142. https://doi.org/10.1111/ejh.13183
Santoro A et al (2021) Clear cell endometrial carcinoma precursors: presentation of two cases and diagnostic issues. Diagn Pathol 16:95. https://doi.org/10.1186/s13000-021-01154-8
Acknowledgements
We thank Yiyao Liu (Tongji University, Shanghai First Maternity and Infant Hospital) and Wei Jiang (Neo Biology Company, Shanghai) for providing whole-exome sequencing technical support.
Funding
This work was supported by a grant from the Shanghai Pudong New Area Health Commission (PDYLT2022-11).
Author information
Authors and Affiliations
Contributions
JNQ and JY conceived the study, analyzed the results, made the figures, and wrote the manuscript together; they made equal contributions to this work; and they are co-first authors. SYZ collected clinical and pathology data. WCW and QPW performed immunohistochemistry experiments. BD participated in data analysis and provided critical revisions to the manuscript. All authors provided comments on the manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the ethics committee of Shanghai First Maternity and Infant Hospital Institutional (Number KS22293).
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qu, J., Yu, J., Zhong, S. et al. The presence of clear cell glands around the ovarian endometrioid cyst has an association with clear cell carcinoma. Virchows Arch 482, 1021–1034 (2023). https://doi.org/10.1007/s00428-022-03479-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-022-03479-1