Abstract
Introduction
Appendiceal neoplasms have a propensity for peritoneal dissemination. The standard of care for select individuals is CRS/HIPEC. In the current 8th AJCC Staging system, a finding of only intraperitoneal acellular mucin (M1a) is classified as Stage IVa. There is concern that the current AJCC system may over-stage patients.
Methods
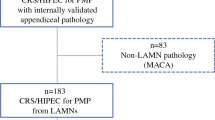
This was a single-institution retrospective review of 164 cases of mucinous appendiceal neoplasm. Patients undergoing CRS/HIPEC with M1a disease were compared to patients with peritoneal deposits containing tumor cells (well-differentiated adenocarcinoma; low-grade mucinous carcinoma peritonei—M1b,G1). Overall and recurrence-free survival were assessed.
Results
Median age was 51 years, 70% were female, and 75% White. Sixty-four patients had M1a disease and 100 M1b,G1 disease. M1a disease had a lower median PCI score (11 vs. 20, p = .0001) and a higher rate of complete CRS (62% vs. 50%, p = .021). Median follow-up was 7.6 years (IQR 5.6–10.5 years). For M1a disease, there were no recurrences and only one patient died during the study interval. In comparison, for M1b disease, 66/100 (66%) recurred with a 5-year RFS of 40.5% (HR 8.0, 95% CI 4.9–15.1, p < .0001), and 31/100 (31%) died with a 5-year OS of 84.8% (HR 4.5, 95% CI 2.2–9.2, p < .0001).
Conclusions
Acellular mucin (M1a disease) after CRS/HIPEC for appendiceal neoplasm is associated with longer OS and RFS compared to M1b, G1 disease. Current AJCC staging does not accurately reflect the differing outcomes of these two patient populations. The presence of acellular mucin in the peritoneal cavity should not be perceived as a metastatic equivalent.





Similar content being viewed by others
Data availability
Patient data for this study is maintained in a prospective clinical database supported by the University of Texas MD Anderson Cancer Center and managed by the institutional review board (IRB). Inquires for access to this information should be made to Dr. Christopher Scally. All software used for this analysis are available to the public for purchase.
References
Takeyama H, Murata K, Takeda T, Fujii M, Kagawa Y, Kawachi H, Yamaguchi T, Noura S, Masuishi T, Inoue A, Takii Y, Suto T, Sakamoto K, Tei M, Kishimoto M, Yao T, Sugihara K, study group of appendiceal neoplasms in Japan Society of Colorectal Cancer Research G (2022) Clinical Significance of Lymph Node Dissection and Lymph Node Metastasis in Primary Appendiceal Tumor Patients After Curative Resection: a Retrospective Multicenter Cohort Study. J Gastrointest Surg 26(1):128–140. https://doi.org/10.1007/s11605-021-05070-6
Sueda T, Murata K, Takeda T, Kagawa Y, Hasegawa J, Komori T, Noura S, Ikeda K, Tsujie M, Ohue M, Ota H, Ikenaga M, Hata T, Matsuda C, Mizushima T, Yamamoto H, Sekimoto M, Nezu R, Mori M, Doki Y (2019) Survival outcomes of appendiceal mucinous neoplasms by histological type and stage: Analysis of 266 cases in a multicenter collaborative retrospective clinical study. Ann Gastroenterol Surg 3(3):291–300. https://doi.org/10.1002/ags3.12241
Ronnett BM, Zahn CM, Kurman RJ, Kass ME, Sugarbaker PH, Shmookler BM, Shmookler BM. Disseminated peritoneal adenomucinosis and peritoneal mucinous carcinomatosis (1995) A clinicopathologic analysis of 109 cases with emphasis on distinguishing pathologic features, site of origin, prognosis, and relationship to “pseudomyxoma peritonei.” Am J Surg Pathol 19(12):1390–408. https://doi.org/10.1097/00000478-199512000-00006
Ronnett BM, Yan H, Kurman RJ, Shmookler BM, Wu L, Sugarbaker PH (2001) Patients with pseudomyxoma peritonei associated with disseminated peritoneal adenomucinosis have a significantly more favorable prognosis than patients with peritoneal mucinous carcinomatosis. Cancer 92(1):85–91. https://doi.org/10.1002/1097-0142(20010701)92:1%3c85::aid-cncr1295%3e3.0.co;2-r
Yantiss RK, Shia J, Klimstra DS, Hahn HP, Odze RD, Misdraji J (2009) Prognostic significance of localized extra-appendiceal mucin deposition in appendiceal mucinous neoplasms. Am J Surg Pathol 33(2):248–255. https://doi.org/10.1097/PAS.0b013e31817ec31e
Amin MB (2017) American Joint Committee on Cancer., American Cancer Society. AJCC cancer staging manual. Eight edition / editor-in-chief, Mahul B. Amin, MD, FCAP ; editors, Stephen B. Edge, MD, FACS and 16 others ; Donna M. Gress, RHIT, CTR - Technical editor ; Laura R. Meyer, CAPM - Managing editor. ed. Chicago IL: American Joint Committee on Cancer, Springer
Fournier K, Rafeeq S, Taggart M, Kanaby P, Ning J, Chen HC, Overman M, Raghav K, Eng C, Mansfield P, Royal R (2017) Low-grade Appendiceal Mucinous Neoplasm of Uncertain Malignant Potential (LAMN-UMP): Prognostic Factors and Implications for Treatment and Follow-up. Ann Surg Oncol 24(1):187–193. https://doi.org/10.1245/s10434-016-5588-2
Carr NJ, Cecil TD, Mohamed F, Sobin LH, Sugarbaker PH, Gonzalez-Moreno S, Taflampas P, Chapman S, Moran BJ, Peritoneal Surface Oncology Group I (2016) A Consensus for Classification and Pathologic Reporting of Pseudomyxoma Peritonei and Associated Appendiceal Neoplasia: The Results of the Peritoneal Surface Oncology Group International (PSOGI) Modified Delphi Process. Am J Surg Pathol 40(1):14–26. https://doi.org/10.1097/PAS.0000000000000535
Jacquet P, Sugarbaker PH (1996) Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. Cancer Treat Res 82:359–374. https://doi.org/10.1007/978-1-4613-1247-5_23
Sugarbaker PH (1999) Successful management of microscopic residual disease in large bowel cancer. Cancer Chemother Pharmacol 43(Suppl):S15-25. https://doi.org/10.1007/s002800051093
Sugarbaker PH (1999) Management of peritoneal-surface malignancy: the surgeon’s role. Langenbecks Arch Surg 384(6):576–587. https://doi.org/10.1007/s004230050246
Center UoTMAC. MD Anderson Price Transparency Charge Lists. (https://www.mdanderson.org/patients-family/becoming-our-patient/planning-for-care/insurance-billing-financial-support/health-care-disclosures.html)
Davison JM, Choudry HA, **pank JF, Ahrendt SA, Holtzman MP, Zureikat AH, Zeh HJ, Ramalingam L, Zhu B, Nikiforova M, Bartlett DL, Pai RK (2014) Clinicopathologic and molecular analysis of disseminated appendiceal mucinous neoplasms: identification of factors predicting survival and proposed criteria for a three-tiered assessment of tumor grade. Mod Pathol 27(11):1521–1539. https://doi.org/10.1038/modpathol.2014.37
Overman MJ, Fournier K, Hu CY, Eng C, Taggart M, Royal R, Mansfield P, Chang GJ (2013) Improving the AJCC/TNM staging for adenocarcinomas of the appendix: the prognostic impact of histological grade. Ann Surg 257(6):1072–1078. https://doi.org/10.1097/SLA.0b013e318269d680
Shetty S, Natarajan B, Thomas P, Govindarajan V, Sharma P, Loggie B (2013) Proposed classification of pseudomyxoma peritonei: influence of signet ring cells on survival. Am Surg 79(11):1171–1176. https://doi.org/10.1177/000313481307901120
Jackson SL, Fleming RA, Loggie BW, Geisinger KR (2001) Gelatinous ascites: a cytohistologic study of pseudomyxoma peritonei in 67 patients. Mod Pathol 14(7):664–671. https://doi.org/10.1038/modpathol.3880370
Pai RK, Beck AH, Norton JA, Longacre TA (2009) Appendiceal mucinous neoplasms: clinicopathologic study of 116 cases with analysis of factors predicting recurrence. Am J Surg Pathol 33(10):1425–1439. https://doi.org/10.1097/PAS.0b013e3181af6067
Baratti D, Kusamura S, Milione M, Bruno F, Guaglio M, Deraco M (2018) Validation of the Recent PSOGI Pathological Classification of Pseudomyxoma Peritonei in a Single-Center Series of 265 Patients Treated by Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy. Ann Surg Oncol 25(2):404–413. https://doi.org/10.1245/s10434-017-6252-1
Choudry HA, Pai RK, Shuai Y, Ramalingam L, Jones HL, **pank JF, Ahrendt SS, Holtzman MP, Zureikat AH, Zeh HJ, Bartlett DL (2018) Impact of Cellularity on Oncologic Outcomes Following Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemoperfusion for Pseudomyxoma Peritonei. Ann Surg Oncol 25(1):76–82. https://doi.org/10.1245/s10434-017-6214-7
Nikiforchin A, King MC, Baron E, MacDonald R, Sittig M, Nieroda C, Gushchin V, Sardi A (2020) Impact of Mucin Cellularity and Distribution on Survival in Newly Diagnosed Patients with Low-Grade Appendiceal Mucinous Neoplasm Treated with Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy. Ann Surg Oncol 27(13):4908–4917. https://doi.org/10.1245/s10434-020-08535-1
van Eden WJ, Kok NFM, Snaebjornsson P, Jozwiak K, Woensdregt K, Bottenberg PD, Boot H, Aalbers AGJ (2019) Factors influencing long-term survival after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for pseudomyxoma peritonei originating from appendiceal neoplasms. BJS Open 3(3):376–386. https://doi.org/10.1002/bjs5.50134
Reghunathan M, Kelly KJ, Valasek MA, Lowy AM, Baumgartner JM (2018) Histologic Predictors of Recurrence in Mucinous Appendiceal Tumors with Peritoneal Dissemination after HIPEC. Ann Surg Oncol 25(3):702–708. https://doi.org/10.1245/s10434-017-6310-8
Bhatt A, Mishra S, Prabhu R, Ramaswamy V, George A, Bhandare S, Shah M, Mehta S (2018) Can low grade PMP be divided into prognostically distinct subgroups based on histological features? A retrospective study and the importance of using the appropriate classification. Eur J Surg Oncol 44(7):1105–1111. https://doi.org/10.1016/j.ejso.2018.03.032
Carr NJ, McCarthy WF, Sobin LH (1995) Epithelial noncarcinoid tumors and tumor-like lesions of the appendix. A clinicopathologic study of 184 patients with a multivariate analysis of prognostic factors. Cancer 75(3):757–68. https://doi.org/10.1002/1097-0142(19950201)75:3%3c757::aid-cncr2820750303%3e3.0.co;2-f
Kong JC, Flood MP, Guerra GR, Liesegang A, Wong WJ, Mitchell C, Warrier SK, Naidu S, Meade B, Lutton N, Heriot AG (2021) Appendiceal pseudomyxoma peritonei: predictors of recurrence and iterative surgery. Colorectal Dis 23(9):2368–2375. https://doi.org/10.1111/codi.15778
Funding
Conrad Family Appendiceal Cancer Research Fund.
William Payne Family Appendiceal Cancer Research Fund.
Bidnick Family Appendiceal Cancer Research Fund.
Schroeder Family Appendiceal Cancer Research Fund.
National Cancer Institute (L30 CA171000 and K22 CA234406 to J.P.S.
Cancer Prevention & Research Institute of Texas (RR180035 to J.P.S.)
Cancer Center Support Grant P30 CA016672).
Sun and Do Lee GI Research and Care Fund.
Dwyer Endowment for Surgical Oncology.
Author information
Authors and Affiliations
Contributions
DJE wrote the main manuscript. DJE, KAR, KB, SR, YJC, and CPS performed chart reviews and data analysis for manuscript generation. KR, JPS, MJO, WCF, MWT, PFM, and RER provided expert opinion and manuscript review. CPS and KF contributed to study design, data analysis, and manuscript generation.
Corresponding author
Ethics declarations
Ethics approval
This retrospective study was reviewed and approved by the MD Anderson Cancer Center Institutional Review Board prior to initiation.
Study and publication consent
All patients included in this study provided signed consent for inclusion of their health information into a prospective clinical database maintained at the University of Texas MD Anderson Cancer Center. Consent includes publication of deidentified information for studies derived from this prospective database. All patient information for this study was derived from this database and has been deindentified.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Keith F. Fournier and Christopher P. Scally share co-senior authorship.
Synopsis:
We evaluated patients with appendiceal neoplasm and peritoneal dissemination in order to determine the prognostic significance of disseminated acellular mucin vs. cellular mucin. Among a cohort of 64 patients with only acellular mucin (AJCC Stage IVa, M1a disease), we detected zero clinically evident recurrences after cytoreductive surgery and HIPEC.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Erstad, D.J., Robinson, K.A., Beaty, K. et al. Prognostic significance of acellular mucin in patients undergoing cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (CRS/HIPEC) for appendiceal neoplasms. Langenbecks Arch Surg 408, 110 (2023). https://doi.org/10.1007/s00423-022-02732-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00423-022-02732-0