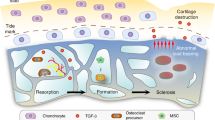
Abstract
In the pathogenesis of osteoarthritis, various signaling pathways may influence the bone joint through a common terminal pathway, thereby contributing to the pathological remodeling of the joint. Semaphorins (SEMAs) are cell-surface proteins actively involved in and primarily responsible for regulating chondrocyte function in the pathophysiological process of osteoarthritis (OA). The significance of the SEMA family in OA is increasingly acknowledged as pivotal. This review aims to summarize the mechanisms through which different members of the SEMA family impact various structures within joints. The findings indicate that SEMA3A and SEMA4D are particularly relevant to OA, as they participate in cartilage injury, subchondral bone remodeling, or synovitis. Additionally, other elements such as SEMA4A and SEMA5A may also contribute to the onset and progression of OA by affecting different components of the bone and joint. The mentioned mechanisms demonstrate the indispensable role of SEMA family members in OA, although the detailed mechanisms still require further exploration.






Similar content being viewed by others
Data availability
No publicly available data or shared data are cited. All original data supporting the conclusion of the current study are available from the corresponding author.
References
Azizi G, Boghozian R, Mirshafiey A (2014) The potential role of angiogenic factors in rheumatoid arthritis. Int J Rheum Dis 17(4):369–383. https://doi.org/10.1111/1756-185x.12280
Bastiaansen-Jenniskens YM, Wei W, Feijt C, Waarsing JH, Verhaar JA, Zuurmond AM, Hanemaaijer R, Stoop R, van Osch GJ (2013) Stimulation of fibrotic processes by the infrapatellar fat pad in cultured synoviocytes from patients with osteoarthritis: a possible role for prostaglandin f2α. Arthritis Rheum 65(8):2070–2080. https://doi.org/10.1002/art.37996
Bertuglia A, Lacourt M, Girard C, Beauchamp G, Richard H, Laverty S (2016) Osteoclasts are recruited to the subchondral bone in naturally occurring post-traumatic equine carpal osteoarthritis and may contribute to cartilage degradation. Osteoarthr Cartil 24(3):555–566. https://doi.org/10.1016/j.joca.2015.10.008
Bijlsma JW, Berenbaum F, Lafeber FP (2011) Osteoarthritis: an update with relevance for clinical practice. Lancet 377(9783):2115–2126. https://doi.org/10.1016/S0140-6736(11)60243-2
Boppart MD, Burkin DJ, Kaufman SJ (2011) Activation of AKT signaling promotes cell growth and survival in α7β1 integrin-mediated alleviation of muscular dystrophy. Biochim Biophys Acta 1812(4):439–446. https://doi.org/10.1016/j.bbadis.2011.01.002
Botter SM, van Osch GJ, Waarsing JH, van der Linden JC, Verhaar JA, Pols HA, van Leeuwen JP, Weinans H (2008) Cartilage damage pattern in relation to subchondral plate thickness in a collagenase-induced model of osteoarthritis. Osteoarthr Cartil 16(4):506–514. https://doi.org/10.1016/j.joca.2007.08.005
Bougeret C, Mansur IG, Dastot H, Schmid M, Mahouy G, Bensussan A, Boumsell L (1992) Increased surface expression of a newly identified 150-kDa dimer early after human T lymphocyte activation. J Immunol 148(2):318–323
Burrage PS, Mix KS, Brinckerhoff CE (2006) Matrix metalloproteinases: role in arthritis. Front Biosci 11:529–543. https://doi.org/10.2741/1817
Carr AJ, Robertsson O, Graves S, Price AJ, Arden NK, Judge A, Beard DJ (2012) Knee replacement. Lancet 379(9823):1331–1340. https://doi.org/10.1016/S0140-6736(11)60752-6
Catalano A (2010) The neuroimmune semaphorin-3A reduces inflammation and progression of experimental autoimmune arthritis. J Immunol 185(10):6373–6383. https://doi.org/10.4049/jimmunol.0903527
Charlier E, Deroyer C, Ciregia F, Malaise O, Neuville S, Plener Z, Malaise M, de Seny D (2019) Chondrocyte dedifferentiation and osteoarthritis (OA). Biochem Pharmacol 165:49–65. https://doi.org/10.1016/j.bcp.2019.02.036
Chen H, Wu G, Sun Q, Dong Y, Zhao H (2016) Hyperbaric oxygen protects mandibular condylar chondrocytes from interleukin-1β-induced apoptosis via the PI3K/AKT signaling pathway. Am J Transl Res 8(11):5108–5117
Chen Y, Zhang L, Liu WX, Wang K (2018) VEGF and SEMA4D have synergistic effects on the promotion of angiogenesis in epithelial ovarian cancer. Cell Mol Biol Lett 23:2. https://doi.org/10.1186/s11658-017-0058-9
Chen WG, Sun J, Shen WW, Yang SZ, Zhang Y, Hu X, Qiu H, Xu SC, Chu TW (2019) Sema4D expression and secretion are increased by HIF-1α and inhibit osteogenesis in bone metastases of lung cancer. Clin Exp Metastasis 36(1):39–56. https://doi.org/10.1007/s10585-018-9951-5
Cheng Q, Chen M, Liu M, Chen X, Zhu L, Xu J, Xue J, Wu H, Du Y (2022) Semaphorin 5A suppresses ferroptosis through activation of PI3K-AKT-mTOR signaling in rheumatoid arthritis. Cell Death Dis 13(7):608. https://doi.org/10.1038/s41419-022-05065-4
Chung L, Shimokawa K, Dinakarpandian D, Grams F, Fields GB, Nagase H (2000) Identification of the (183)RWTNNFREY(191) region as a critical segment of matrix metalloproteinase 1 for the expression of collagenolytic activity. J Biol Chem 275(38):29610–29617. https://doi.org/10.1074/jbc.M004039200
Cui Z, Crane J, **e H, ** X, Zhen G, Li C, **e L, Wang L, Bian Q, Qiu T, Wan M, **e M, Ding S, Yu B, Cao X (2016) Halofuginone attenuates osteoarthritis by inhibition of TGF-β activity and H-type vessel formation in subchondral bone. Ann Rheum Dis 75(9):1714–1721. https://doi.org/10.1136/annrheumdis-2015-207923
Dall’Ara E, Ohman C, Baleani M, Viceconti M (2011) Reduced tissue hardness of trabecular bone is associated with severe osteoarthritis. J Biomech 44(8):1593–1598. https://doi.org/10.1016/j.jbiomech.2010.12.022
de Wit J, Verhaagen J (2003) Role of semaphorins in the adult nervous system. Prog Neurobiol 71(2–3):249–267. https://doi.org/10.1016/j.pneurobio.2003.06.001
Elhabazi A, Marie-Cardine A, Chabbert-de Ponnat I, Bensussan A, Boumsell L (2003) Structure and function of the immune semaphorin CD100/SEMA4D. Crit Rev Immunol 23(1–2):65–81. https://doi.org/10.1615/critrevimmunol.v23.i12.40
Findlay DM, Atkins GJ (2014) Osteoblast-chondrocyte interactions in osteoarthritis. Curr Osteoporos Rep 12(1):127–134. https://doi.org/10.1007/s11914-014-0192-5
Fransès RE, McWilliams DF, Mapp PI, Walsh DA (2010) Osteochondral angiogenesis and increased protease inhibitor expression in OA. Osteoarthr Cartil 18(4):563–571. https://doi.org/10.1016/j.joca.2009.11.015
Fukuda T, Takeda S, Xu R, Ochi H, Sunamura S, Sato T, Shibata S, Yoshida Y, Gu Z, Kimura A, Ma C, Xu C, Bando W, Fujita K, Shinomiya K, Hirai T, Asou Y, Enomoto M, Okano H, Okawa A, Itoh H (2013) Sema3A regulates bone-mass accrual through sensory innervations. Nature 497(7450):490–493. https://doi.org/10.1038/nature12115
Gao H, Ma XX, Guo Q, **e LF, Zhong YC, Zhang XW (2018) Expression of circulating Semaphorin3A and its association with inflammation and bone destruction in rheumatoid arthritis. Clin Rheumatol 37(8):2073–2080. https://doi.org/10.1007/s10067-018-4070-x
Garcia S (2019) Role of semaphorins in immunopathologies and rheumatic diseases. Int J Mol Sci. https://doi.org/10.3390/ijms20020374
Gaur P, Bielenberg DR, Samuel S, Bose D, Zhou Y, Gray MJ, Dallas NA, Fan F, **a L, Lu J, Ellis LM (2009) Role of class 3 semaphorins and their receptors in tumor growth and angiogenesis. Clin Cancer Res 15(22):6763–6770. https://doi.org/10.1158/1078-0432.Ccr-09-1810
Geretti E, Shimizu A, Klagsbrun M (2008) Neuropilin structure governs VEGF and semaphorin binding and regulates angiogenesis. Angiogenesis 11(1):31–39. https://doi.org/10.1007/s10456-008-9097-1
Glyn-Jones S, Palmer AJ, Agricola R, Price AJ, Vincent TL, Weinans H, Carr AJ (2015) Osteoarthritis. Lancet 386(9991):376–387. https://doi.org/10.1016/s0140-6736(14)60802-3
Gomoll AH, Madry H, Knutsen G, van Dijk N, Seil R, Brittberg M, Kon E (2010) The subchondral bone in articular cartilage repair: current problems in the surgical management. Knee Surg Sports Traumatol Arthrosc 18(4):434–447. https://doi.org/10.1007/s00167-010-1072-x
Gras C, Eiz-Vesper B, Jaimes Y, Immenschuh S, Jacobs R, Witte T, Blasczyk R, Figueiredo C (2014) Secreted semaphorin 5A activates immune effector cells and is a biomarker for rheumatoid arthritis. Arthritis Rheumatol 66(6):1461–1471. https://doi.org/10.1002/art.38425
Hamilton JL, Nagao M, Levine BR, Chen D, Olsen BR, Im HJ (2016) Targeting VEGF and its receptors for the treatment of osteoarthritis and associated pain. J Bone Miner Res 31(5):911–924. https://doi.org/10.1002/jbmr.2828
Hayashi M, Nakashima T, Taniguchi M, Kodama T, Kumanogoh A, Takayanagi H (2012) Osteoprotection by semaphorin 3A. Nature 485(7396):69–74. https://doi.org/10.1038/nature11000
Hayashi M, Nakashima T, Yoshimura N, Okamoto K, Tanaka S, Takayanagi H (2019) Autoregulation of osteocyte Sema3A orchestrates estrogen action and counteracts bone aging. Cell Metab 29(3):627-637.e625. https://doi.org/10.1016/j.cmet.2018.12.021
Henrotin Y, Pesesse L, Sanchez C (2009) Subchondral bone in osteoarthritis physiopathology: state-of-the art and perspectives. Biomed Mater Eng 19(4–5):311–316. https://doi.org/10.3233/bme-2009-0596
Hiligsmann M, Cooper C, Arden N, Boers M, Branco JC, Luisa Brandi M, Bruyère O, Guillemin F, Hochberg MC, Hunter DJ, Kanis JA, Kvien TK, Laslop A, Pelletier JP, Pinto D, Reiter-Niesert S, Rizzoli R, Rovati LC, Severens JL, Silverman S, Tsouderos Y, Tugwell P, Reginster JY (2013) Health economics in the field of osteoarthritis: an expert’s consensus paper from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO). Semin Arthritis Rheum 43(3):303–313. https://doi.org/10.1016/j.semarthrit.2013.07.003
Honda K, Ohno S, Tanimoto K, Ijuin C, Tanaka N, Doi T, Kato Y, Tanne K (2000) The effects of high magnitude cyclic tensile load on cartilage matrix metabolism in cultured chondrocytes. Eur J Cell Biol 79(9):601–609. https://doi.org/10.1078/0171-9335-00089
Hong BK, You S, Yoo SA, Park D, Hwang D, Cho CS, Kim WU (2017) MicroRNA-143 and -145 modulate the phenotype of synovial fibroblasts in rheumatoid arthritis. Exp Mol Med 49(8):e363. https://doi.org/10.1038/emm.2017.108
Igea A, Carvalheiro T, Malvar-Fernández B, Martinez-Ramos S, Rafael-Vidal C, Niemantsverdriet E, Varadé J, Fernández-Carrera A, Jimenez N, McGarry T, Rodriguez-Trillo A, Veale D, Fearon U, Conde C, Pego-Reigosa JM, González-Fernández Á, Reedquist KA, Radstake T, van der Helm-Van MA, García S (2022) Central role of semaphorin 3B in a serum-induced arthritis model and reduced levels in patients with rheumatoid arthritis. Arthritis Rheumatol 74(6):972–983. https://doi.org/10.1002/art.42065
Imhof H, Sulzbacher I, Grampp S, Czerny C, Youssefzadeh S, Kainberger F (2000) Subchondral bone and cartilage disease: a rediscovered functional unit. Invest Radiol 35(10):581–588. https://doi.org/10.1097/00004424-200010000-00004
Ishii T, Ruiz-Torruella M, Yamamoto K, Yamaguchi T, Heidari A, Pierrelus R, Leon E, Shindo S, Rawas-Qalaji M, Pastore MR, Ikeda A, Nakamura S, Mawardi H, Kandalam U, Hardigan P, Witek L, Coelho PG, Kawai T (2022) Locally secreted semaphorin 4D is engaged in both pathogenic bone resorption and retarded bone regeneration in a ligature-induced mouse model of periodontitis. Int J Mol Sci. https://doi.org/10.3390/ijms23105630
Janssen BJ, Malinauskas T, Weir GA, Cader MZ, Siebold C, Jones EY (2012) Neuropilins lock secreted semaphorins onto plexins in a ternary signaling complex. Nat Struct Mol Biol 19(12):1293–1299. https://doi.org/10.1038/nsmb.2416
Joosten LA, Helsen MM, Saxne T, van De Loo FA, Heinegard D, van Den Berg WB (1999) IL-1 alpha beta blockade prevents cartilage and bone destruction in murine type II collagen-induced arthritis, whereas TNF-alpha blockade only ameliorates joint inflammation. J Immunol 163(9):5049–5055
Joyal JS, Sitaras N, Binet F, Rivera JC, Stahl A, Zaniolo K, Shao Z, Polosa A, Zhu T, Hamel D, Djavari M, Kunik D, Honoré JC, Picard E, Zabeida A, Varma DR, Hickson G, Mancini J, Klagsbrun M, Costantino S, Beauséjour C, Lachapelle P, Smith LE, Chemtob S, Sapieha P (2011) Ischemic neurons prevent vascular regeneration of neural tissue by secreting semaphorin 3A. Blood 117(22):6024–6035. https://doi.org/10.1182/blood-2010-10-311589
Kamei H, Ishii T, Nishii Y (2022) Semaphorin 3A regulates alveolar bone remodeling on orthodontic tooth movement. Sci Rep 12(1):9243. https://doi.org/10.1038/s41598-022-13217-x
Kapoor M, Martel-Pelletier J, Lajeunesse D, Pelletier JP, Fahmi H (2011) Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat Rev Rheumatol 7(1):33–42. https://doi.org/10.1038/nrrheum.2010.196
Knäuper V, López-Otin C, Smith B, Knight G, Murphy G (1996) Biochemical characterization of human collagenase-3. J Biol Chem 271(3):1544–1550. https://doi.org/10.1074/jbc.271.3.1544
Leblond A, Allanore Y, Avouac J (2017) Targeting synovial neoangiogenesis in rheumatoid arthritis. Autoimmun Rev 16(6):594–601. https://doi.org/10.1016/j.autrev.2017.04.005
Lefèvre S, Knedla A, Tennie C, Kampmann A, Wunrau C, Dinser R, Korb A, Schnäker EM, Tarner IH, Robbins PD, Evans CH, Stürz H, Steinmeyer J, Gay S, Schölmerich J, Pap T, Müller-Ladner U, Neumann E (2009) Synovial fibroblasts spread rheumatoid arthritis to unaffected joints. Nat Med 15(12):1414–1420. https://doi.org/10.1038/nm.2050
Lefèvre S, Schwarz M, Meier FMP, Zimmermann-Geller B, Tarner IH, Rickert M, Steinmeyer J, Sauerbier M, Rehart S, Müller-Ladner U, Neumann E (2017) Disease-specific effects of matrix and growth factors on adhesion and migration of rheumatoid synovial fibroblasts. J Immunol 198(12):4588–4595. https://doi.org/10.4049/jimmunol.1600989
Lei J, Fu Y, Zhuang Y, Zhang K (2020) Sema4D aggravated LPS-Induced injury via activation of the MAPK signaling pathway in ATDC5 chondrocytes. Biomed Res Int 2020:8691534. https://doi.org/10.1155/2020/8691534
Lerner A, Neidhöfer S, Reuter S, Matthias T (2018) MMP3 is a reliable marker for disease activity, radiological monitoring, disease outcome predictability, and therapeutic response in rheumatoid arthritis. Best Pract Res Clin Rheumatol 32(4):550–562. https://doi.org/10.1016/j.berh.2019.01.006
Li B, Aspden RM (1997) Material properties of bone from the femoral neck and calcar femorale of patients with osteoporosis or osteoarthritis. Osteoporos Int 7(5):450–456. https://doi.org/10.1007/s001980050032
Li JY, Wang TT, Li C, Wang ZF, Li S, Ma L, Zheng LL (2020) Semaphorin 3A-hypoxia inducible factor 1 subunit alpha co-overexpression enhances the osteogenic differentiation of induced pluripotent stem cells-derived mesenchymal stem cells in vitro. Chin Med J (engl) 133(3):301–309. https://doi.org/10.1097/cm9.0000000000000612
Liacini A, Sylvester J, Li WQ, Zafarullah M (2002) Inhibition of interleukin-1-stimulated MAP kinases, activating protein-1 (AP-1) and nuclear factor kappa B (NF-kappa B) transcription factors down-regulates matrix metalloproteinase gene expression in articular chondrocytes. Matrix Biol 21(3):251–262. https://doi.org/10.1016/s0945-053x(02)00007-0
Liang J, Xu L, Zhou F, Liu AM, Ge HX, Chen YY, Tu M (2018) MALAT1/miR-127-5p regulates osteopontin (OPN)-mediated proliferation of human chondrocytes through PI3K/Akt pathway. J Cell Biochem 119(1):431–439. https://doi.org/10.1002/jcb.26200
Liu YQ, Han XF, Bo JX, Ma HP (2016) Wedelolactone enhances osteoblastogenesis but inhibits osteoclastogenesis through Sema3A/NRP1/PlexinA1 pathway. Front Pharmacol 7:375. https://doi.org/10.3389/fphar.2016.00375
Liu B, Zhang M, Zhao J, Zheng M, Yang H (2018) Imbalance of M1/M2 macrophages is linked to severity level of knee osteoarthritis. Exp Ther Med 16(6):5009–5014. https://doi.org/10.3892/etm.2018.6852
Liu L, Yang L, Liu X, Liu M, Liu J, Feng X, Nie Z, Luo J (2022) SEMA4D/PlexinB1 promotes AML progression via activation of PI3K/Akt signaling. J Transl Med 20(1):304. https://doi.org/10.1186/s12967-022-03500-w
Llamas-Granda P, Martin-Rodríguez L, Largo R, Herrero-Beaumont G, Mediero A (2021) Tenofovir modulates semaphorin 4D signaling and regulates bone homeostasis, which can be counteracted by dipyridamole and adenosine A2A receptor. Int J Mol Sci. https://doi.org/10.3390/ijms222111490
Loeser RF, Collins JA, Diekman BO (2016) Ageing and the pathogenesis of osteoarthritis. Nat Rev Rheumatol 12(7):412–420. https://doi.org/10.1038/nrrheum.2016.65
Lu L, Huang J, Zhang X, Zhang J, Zhang M, **g L, Yu S, Wang M (2014) Changes of temporomandibular joint and semaphorin 4D/Plexin-B1 expression in a mouse model of incisor malocclusion. J Oral Facial Pain Headache 28(1):68–79. https://doi.org/10.11607/jop.1082
Lu J, Zhang H, Cai D, Zeng C, Lai P, Shao Y, Fang H, Li D, Ouyang J, Zhao C, **e D, Huang B, Yang J, Jiang Y, Bai X (2018) Positive-feedback regulation of subchondral H-type vessel formation by chondrocyte promotes osteoarthritis development in mice. J Bone Miner Res 33(5):909–920. https://doi.org/10.1002/jbmr.3388
Luque MCA, Galuppo MK, Capelli-Peixoto J, Stolf BS (2018) CD100 effects in macrophages and its roles in atherosclerosis. Front Cardiovasc Med 5:136. https://doi.org/10.3389/fcvm.2018.00136
Lyu Z, ** H, Yan Z, Hu K, Jiang H, Peng H, Zhuo H (2020) Effects of NRP1 on angiogenesis and vascular maturity in endothelial cells are dependent on the expression of SEMA4D. Int J Mol Med 46(4):1321–1334. https://doi.org/10.3892/ijmm.2020.4692
Ma X, Lv J, Sun X, Ma J, **ng G, Wang Y, Sun L, Wang J, Li F, Li Y, Zhao Z (2016) Naringin ameliorates bone loss induced by sciatic neurectomy and increases Semaphorin 3A expression in denervated bone. Sci Rep 6:24562. https://doi.org/10.1038/srep24562
Mapp PI, Walsh DA (2012) Mechanisms and targets of angiogenesis and nerve growth in osteoarthritis. Nat Rev Rheumatol 8(7):390–398. https://doi.org/10.1038/nrrheum.2012.80
Marcu KB, Otero M, Olivotto E, Borzi RM, Goldring MB (2010) NF-kappaB signaling: multiple angles to target OA. Curr Drug Targets 11(5):599–613. https://doi.org/10.2174/138945010791011938
Mathiessen A, Conaghan PG (2017) Synovitis in osteoarthritis: current understanding with therapeutic implications. Arthritis Res Ther 19(1):18. https://doi.org/10.1186/s13075-017-1229-9
McInnes IB, Buckley CD, Isaacs JD (2016) Cytokines in rheumatoid arthritis—sha** the immunological landscape. Nat Rev Rheumatol 12(1):63–68. https://doi.org/10.1038/nrrheum.2015.171
Mediero A, Wilder T, Shah L, Cronstein BN (2018) Adenosine A(2A) receptor (A2AR) stimulation modulates expression of semaphorins 4D and 3A, regulators of bone homeostasis. Faseb j 32(7):3487–3501. https://doi.org/10.1096/fj.201700217R
Milam SB, Zardeneta G, Schmitz JP (1998) Oxidative stress and degenerative temporomandibular joint disease: a proposed hypothesis. J Oral Maxillofac Surg 56(2):214–223. https://doi.org/10.1016/s0278-2391(98)90872-2
Miyabe Y, Lian J, Miyabe C, Luster AD (2019) Chemokines in rheumatic diseases: pathogenic role and therapeutic implications. Nat Rev Rheumatol 15(12):731–746. https://doi.org/10.1038/s41584-019-0323-6
Mort JS, Billington CJ (2001) Articular cartilage and changes in arthritis: matrix degradation. Arthritis Res 3(6):337–341. https://doi.org/10.1186/ar325
Movila A, Mawardi H, Nishimura K, Kiyama T, Egashira K, Kim JY, Villa A, Sasaki H, Woo SB, Kawai T (2016) Possible pathogenic engagement of soluble Semaphorin 4D produced by γδT cells in medication-related osteonecrosis of the jaw (MRONJ). Biochem Biophys Res Commun 480(1):42–47. https://doi.org/10.1016/j.bbrc.2016.10.012
Müller-Ladner U, Kriegsmann J, Franklin BN, Matsumoto S, Geiler T, Gay RE, Gay S (1996) Synovial fibroblasts of patients with rheumatoid arthritis attach to and invade normal human cartilage when engrafted into SCID mice. Am J Pathol 149(5):1607–1615
Muraoka T, Hagino H, Okano T, Enokida M, Teshima R (2007) Role of subchondral bone in osteoarthritis development: a comparative study of two strains of guinea pigs with and without spontaneously occurring osteoarthritis. Arthritis Rheum 56(10):3366–3374. https://doi.org/10.1002/art.22921
Negishi-Koga T, Shinohara M, Komatsu N, Bito H, Kodama T, Friedel RH, Takayanagi H (2011) Suppression of bone formation by osteoclastic expression of semaphorin 4D. Nat Med 17(11):1473–1480. https://doi.org/10.1038/nm.2489
Neuhold LA, Killar L, Zhao W, Sung ML, Warner L, Kulik J, Turner J, Wu W, Billinghurst C, Meijers T, Poole AR, Babij P, DeGennaro LJ (2001) Postnatal expression in hyaline cartilage of constitutively active human collagenase-3 (MMP-13) induces osteoarthritis in mice. J Clin Invest 107(1):35–44. https://doi.org/10.1172/jci10564
Neve A, Corrado A, Cantatore FP (2011) Osteoblast physiology in normal and pathological conditions. Cell Tissue Res 343(2):289–302. https://doi.org/10.1007/s00441-010-1086-1
Nygaard G, Firestein GS (2020) Restoring synovial homeostasis in rheumatoid arthritis by targeting fibroblast-like synoviocytes. Nat Rev Rheumatol 16(6):316–333. https://doi.org/10.1038/s41584-020-0413-5
Okubo M, Kimura T, Fujita Y, Mochizuki S, Niki Y, Enomoto H, Suda Y, Toyama Y, Okada Y (2011) Semaphorin 3A is expressed in human osteoarthritic cartilage and antagonizes vascular endothelial growth factor 165-promoted chondrocyte migration: an implication for chondrocyte cloning. Arthritis Rheum 63(10):3000–3009. https://doi.org/10.1002/art.30482
Olivotto E, Otero M, Marcu KB, Goldring MB (2015) Pathophysiology of osteoarthritis: canonical NF-κB/IKKβ-dependent and kinase-independent effects of IKKα in cartilage degradation and chondrocyte differentiation. RMD Open 1(Suppl 1):e000061. https://doi.org/10.1136/rmdopen-2015-000061
Oxford Textbook of Rheumatology (2013). Oxford University Press. https://doi.org/10.1093/med/9780199642489.001.0001
Pap T, Dankbar B, Wehmeyer C, Korb-Pap A, Sherwood J (2020) Synovial fibroblasts and articular tissue remodelling: role and mechanisms. Semin Cell Dev Biol 101:140–145. https://doi.org/10.1016/j.semcdb.2019.12.006
Pivec R, Johnson AJ, Mears SC, Mont MA (2012) Hip arthroplasty. Lancet 380(9855):1768–1777. https://doi.org/10.1016/S0140-6736(12)60607-2
Prasadam I, Farnaghi S, Feng JQ, Gu W, Perry S, Crawford R, **ao Y (2013) Impact of extracellular matrix derived from osteoarthritis subchondral bone osteoblasts on osteocytes: role of integrinβ1 and focal adhesion kinase signaling cues. Arthritis Res Ther 15(5):R150. https://doi.org/10.1186/ar4333
Purohit A, Sadanandam A, Myneni P, Singh RK (2014) Semaphorin 5A mediated cellular navigation: connecting nervous system and cancer. Biochim Biophys Acta 1846(2):485–493. https://doi.org/10.1016/j.bbcan.2014.09.006
Rawlings ND, Barrett AJ, Thomas PD, Huang X, Bateman A, Finn RD (2018) The MEROPS database of proteolytic enzymes, their substrates and inhibitors in 2017 and a comparison with peptidases in the PANTHER database. Nucleic Acids Res 46:D624–D632
Remst DF, Blom AB, Vitters EL, Bank RA, van den Berg WB, Blaney Davidson EN, van der Kraan PM (2014) Gene expression analysis of murine and human osteoarthritis synovium reveals elevation of transforming growth factor β-responsive genes in osteoarthritis-related fibrosis. Arthritis Rheumatol 66(3):647–656. https://doi.org/10.1002/art.38266
Rigoglou S, Papavassiliou AG (2013) The NF-κB signalling pathway in osteoarthritis. Int J Biochem Cell Biol 45(11):2580–2584. https://doi.org/10.1016/j.biocel.2013.08.018
Sabag AD, Smolkin T, Mumblat Y, Ueffing M, Kessler O, Gloeckner CJ, Neufeld G (2014) The role of the plexin-A2 receptor in Sema3A and Sema3B signal transduction. J Cell Sci 127(Pt 24):5240–5252. https://doi.org/10.1242/jcs.155960
Scanzello CR, Goldring SR (2012) The role of synovitis in osteoarthritis pathogenesis. Bone 51(2):249–257. https://doi.org/10.1016/j.bone.2012.02.012
Shindo S, Savitri IJ, Ishii T, Ikeda A, Pierrelus R, Heidari A, Okubo K, Nakamura S, Kandalam U, Rawas-Qalaji M, Leon E, Pastore MR, Hardigan P, Kawai T (2022) Dual-function semaphorin 4D released by platelets: suppression of osteoblastogenesis and promotion of osteoclastogenesis. Int J Mol Sci. https://doi.org/10.3390/ijms23062938
Soker S, Miao HQ, Nomi M, Takashima S, Klagsbrun M (2002) VEGF165 mediates formation of complexes containing VEGFR-2 and neuropilin-1 that enhance VEGF165-receptor binding. J Cell Biochem 85(2):357–368. https://doi.org/10.1002/jcb.10140
Steffensen B, Bigg HF, Overall CM (1998) The involvement of the fibronectin type II-like modules of human gelatinase A in cell surface localization and activation. J Biol Chem 273(32):20622–20628. https://doi.org/10.1074/jbc.273.32.20622
Steinmeyer J, Ackermann B, Raiss RX (1997) Intermittent cyclic loading of cartilage explants modulates fibronectin metabolism. Osteoarthr Cartil 5(5):331–341. https://doi.org/10.1016/s1063-4584(97)80037-4
Stöckl S, Reichart J, Zborilova M, Johnstone B, Grässel S (2022) Semaphorin 3A-neuropilin-1 signaling modulates MMP13 expression in human osteoarthritic chondrocytes. Int J Mol Sci. https://doi.org/10.3390/ijms232214180
Sumi C, Hirose N, Yanoshita M, Takano M, Nishiyama S, Okamoto Y, Asakawa Y, Tanimoto K (2018) Semaphorin 3A inhibits inflammation in chondrocytes under excessive mechanical stress. Mediators Inflamm 2018:5703651. https://doi.org/10.1155/2018/5703651
Sun J, Wei X, Wang Z, Liu Y, Lu J, Lu Y, Cui M, Zhang X, Li F (2018) Inflammatory milieu cultivated Sema3A signaling promotes chondrocyte apoptosis in knee osteoarthritis. J Cell Biochem 119(3):2891–2899. https://doi.org/10.1002/jcb.26470
Suri S, Walsh DA (2012) Osteochondral alterations in osteoarthritis. Bone 51(2):204–211. https://doi.org/10.1016/j.bone.2011.10.010
Takada H, Ibaragi S, Eguchi T, Okui T, Obata K, Masui M, Morisawa A, Takabatake K, Kawai H, Yoshioka N, Hassan NMM, Shimo T, Hu GF, Nagatsuka H, Sasaki A (2017) Semaphorin 4D promotes bone invasion in head and neck squamous cell carcinoma. Int J Oncol 51(2):625–632. https://doi.org/10.3892/ijo.2017.4050
Tang MW, Malvar Fernández B, Newsom SP, van Buul JD, Radstake T, Baeten DL, Tak PP, Reedquist KA, García S (2018) Class 3 semaphorins modulate the invasive capacity of rheumatoid arthritis fibroblast-like synoviocytes. Rheumatology (oxford) 57(5):909–920. https://doi.org/10.1093/rheumatology/kex511
Teng Y, Yin Z, Li J, Li K, Li X, Zhang Y (2017) Adenovirus-mediated delivery of Sema3A alleviates rheumatoid arthritis in a serum-transfer induced mouse model. Oncotarget 8(39):66270–66280. https://doi.org/10.18632/oncotarget.19915
Termini CM, Pang A, Fang T, Roos M, Chang VY, Zhang Y, Setiawan NJ, Signaevskaia L, Li M, Kim MM, Tabibi O, Lin PK, Sasine JP, Chatterjee A, Murali R, Himburg HA, Chute JP (2021) Neuropilin 1 regulates bone marrow vascular regeneration and hematopoietic reconstitution. Nat Commun 12(1):6990. https://doi.org/10.1038/s41467-021-27263-y
Valencia X, Higgins JM, Kiener HP, Lee DM, Podrebarac TA, Dascher CC, Watts GF, Mizoguchi E, Simmons B, Patel DD, Bhan AK, Brenner MB (2004) Cadherin-11 provides specific cellular adhesion between fibroblast-like synoviocytes. J Exp Med 200(12):1673–1679. https://doi.org/10.1084/jem.20041545
Varshavsky A, Kessler O, Abramovitch S, Kigel B, Zaffryar S, Akiri G, Neufeld G (2008) Semaphorin-3B is an angiogenesis inhibitor that is inactivated by furin-like pro-protein convertases. Cancer Res 68(17):6922–6931. https://doi.org/10.1158/0008-5472.Can-07-5408
Verlinden L, Vanderschueren D, Verstuyf A (2016) Semaphorin signaling in bone. Mol Cell Endocrinol 432:66–74. https://doi.org/10.1016/j.mce.2015.09.009
Walsh DA, McWilliams DF, Turley MJ, Dixon MR, Fransès RE, Mapp PI, Wilson D (2010) Angiogenesis and nerve growth factor at the osteochondral junction in rheumatoid arthritis and osteoarthritis. Rheumatology (oxford) 49(10):1852–1861. https://doi.org/10.1093/rheumatology/keq188
Wang L, Song G, Zheng Y, Tan W, Pan J, Zhao Y, Chang X (2015) Expression of Semaphorin 4A and its potential role in rheumatoid arthritis. Arthritis Res Ther 17(1):227. https://doi.org/10.1186/s13075-015-0734-y
Wang X, Wu Q, Zhang R, Fan Z, Li W, Mao R, Du Z, Yao X, Ma Y, Yan Y, Sun W, Wu H, Wei W, Hu Y, Hong Y, Hu H, Koh YW, Duan W, Chen X, Ouyang H (2023) Stage-specific and location-specific cartilage calcification in osteoarthritis development. Ann Rheum Dis 82(3):393–402. https://doi.org/10.1136/ard-2022-222944
Wong M, Siegrist M, Cao X (1999) Cyclic compression of articular cartilage explants is associated with progressive consolidation and altered expression pattern of extracellular matrix proteins. Matrix Biol 18(4):391–399. https://doi.org/10.1016/s0945-053x(99)00029-3
Woolf AD, Pfleger B (2003) Burden of major musculoskeletal conditions. Bull World Health Organ 81(9):646–656
Worzfeld T, Offermanns S (2014) Semaphorins and plexins as therapeutic targets. Nat Rev Drug Discov 13(8):603–621. https://doi.org/10.1038/nrd4337
Wu JH, Li YN, Chen AQ, Hong CD, Zhang CL, Wang HL, Zhou YF, Li PC, Wang Y, Mao L, **a YP, He QW, ** HJ, Yue ZY, Hu B (2020) Inhibition of Sema4D/PlexinB1 signaling alleviates vascular dysfunction in diabetic retinopathy. EMBO Mol Med 12(2):e10154. https://doi.org/10.15252/emmm.201810154
Wu Z, Ma D, Yang H, Gao J, Zhang G, Xu K, Zhang L (2021) Fibroblast-like synoviocytes in rheumatoid arthritis: Surface markers and phenotypes. Int Immunopharmacol 93:107392. https://doi.org/10.1016/j.intimp.2021.107392
**ao C, Lv C, Sun S, Zhao H, Ling H, Li M, Qin Y, Zhang J, Wang J, Yang X (2021) TSP1 is the essential domain of SEMA5A involved in pannus formation in rheumatoid arthritis. Rheumatology (oxford) 60(12):5833–5842. https://doi.org/10.1093/rheumatology/keab133
**ng Q, Feng J, Zhang X (2021) Glucocorticoids suppressed osteoblast differentiation by decreasing Sema3A expression via the PIK3/Akt pathway. Exp Cell Res 403(1):112595. https://doi.org/10.1016/j.yexcr.2021.112595
Xu H, Wang W, Liu X, Huang W, Zhu C, Xu Y, Yang H, Bai J, Geng D (2023) Targeting strategies for bone diseases: signaling pathways and clinical studies. Signal Transduct Target Ther 8(1):202. https://doi.org/10.1038/s41392-023-01467-8
Yang QZ, Ma XB, Li YH, Li YN, Zhong YC, Zhang XW (2020) Correlation analysis of Sema4D with rheumatoid arthritis disease activity, bone destruction and rheumatoid arthritis-related interstitial lung disease. Zhonghua Yi Xue Za Zhi 100(20):1567–1572. https://doi.org/10.3760/cma.j.cn112137-20191130-02613
Yoshida Y, Ogata A, Kang S, Ebina K, Shi K, Nojima S, Kimura T, Ito D, Morimoto K, Nishide M, Hosokawa T, Hirano T, Shima Y, Narazaki M, Tsuboi H, Saeki Y, Tomita T, Tanaka T, Kumanogoh A (2015) Semaphorin 4D contributes to rheumatoid arthritis by inducing inflammatory cytokine production: pathogenic and therapeutic implications. Arthritis Rheumatol 67(6):1481–1490. https://doi.org/10.1002/art.39086
Yu W, Bai Y, Han N, Wang F, Zhao M, Huang L, Li X (2013) Inhibition of pathological retinal neovascularization by semaphorin 3A. Mol vis 19:1397–1405
Zeng CY, Wang XF, Hua FZ (2022) HIF-1α in osteoarthritis: from pathogenesis to therapeutic implications. Front Pharmacol 13:927126. https://doi.org/10.3389/fphar.2022.927126
Zhang Y, Liu B, Ma Y, ** B (2013) Sema 4D/CD100-plexin B is a multifunctional counter-receptor. Cell Mol Immunol 10(2):97–98. https://doi.org/10.1038/cmi.2012.65
Zhang X, Xu X, Xu T, Qin S (2014) β-Ecdysterone suppresses interleukin-1β-induced apoptosis and inflammation in rat chondrocytes via inhibition of NF-κB signaling pathway. Drug Dev Res 75(3):195–201. https://doi.org/10.1002/ddr.21170
Zhang M, Ishikawa S, Inagawa T, Ikemoto H, Guo S, Sunagawa M, Hisamitsu T (2017) Influence of mechanical force on bone matrix proteins in ovariectomised mice and osteoblast-like MC3T3-E1 cells. In Vivo 31(1):87–95. https://doi.org/10.21873/invivo.11029
Zhang H, Wei Q, **ang X, Zhou B, Chen J, Li J, Li Q, **ong H, Liu F (2019) Semaphorin 4A acts in a feed-forward loop with NF-κB pathway to exacerbate catabolic effect of IL-1β on chondrocytes. Int Immunopharmacol 69:88–94. https://doi.org/10.1016/j.intimp.2019.01.006
Zhang H, Lu Y, Wu B, **a F (2021) Semaphorin 3A mitigates lipopolysaccharide-induced chondrocyte inflammation, apoptosis and extracellular matrix degradation by binding to Neuropilin-1. Bioengineered 12(2):9641–9654. https://doi.org/10.1080/21655979.2021.1974806
Zhang Z, Lu L, Ye T, Yu S, Zhang J, Zhang M, He F, Liu Q, Yang H, Feng J (2022) Inhibition of Semaphorin 4D/Plexin-B1 signaling inhibits the subchondral bone loss in early-stage osteoarthritis of the temporomandibular joint. Arch Oral Biol 135:105365. https://doi.org/10.1016/j.archoralbio.2022.105365
Zhou H, Yang YH, Binmadi NO, Proia P, Basile JR (2012) The hypoxia-inducible factor-responsive proteins semaphorin 4D and vascular endothelial growth factor promote tumor growth and angiogenesis in oral squamous cell carcinoma. Exp Cell Res 318(14):1685–1698. https://doi.org/10.1016/j.yexcr.2012.04.019
Zhu Z, Luo Y, Yu J, Gao J, Zhang Y, **ao C, Zhang C, Wang G, Liu Y, Fu M, Yao X, Li W (2016) Sema4D is required in both the adaptive and innate immune responses of contact hypersensitivity. Mol Immunol 78:98–104. https://doi.org/10.1016/j.molimm.2016.09.003
Zou T, Jiang S, Dissanayaka WL, Heng BC, Xu J, Gong T, Huang X, Zhang C (2019) Sema4D/PlexinB1 promotes endothelial differentiation of dental pulp stem cells via activation of AKT and ERK1/2 signaling. J Cell Biochem 120(8):13614–13624. https://doi.org/10.1002/jcb.28635
Funding
This work was supported by the funding of NSFC grants (82330029) to Qianming Chen and was supported by the Zhejiang Provincial Natural Science Foundation of China under grant no. LQ24H140004 to Zu** Wu, LQ21H140004 to Li Xu, and LQ24H140003 to Qian Chen.
Author information
Authors and Affiliations
Ethics declarations
Conflict of interest
The authors report no conflicts of interest.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Peng, W., Chen, Q., Zheng, F. et al. The emerging role of the semaphorin family in cartilage and osteoarthritis. Histochem Cell Biol (2024). https://doi.org/10.1007/s00418-024-02303-y
Accepted:
Published:
DOI: https://doi.org/10.1007/s00418-024-02303-y