Abstract
Purpose
To provide a brief summary and comparison of the most recent literature on available and theorized treatment modalities for classic lattice corneal dystrophy (LCD). This paper aims to support practitioners in their management of this disease.
Methods
A search was carried out on available literature through PubMed and Google Scholar of English language articles up to January 2023 that relate to the treatment of LCD. Due to scarcity of literature regarding specific novel therapies for LCD, results from other corneal pathologies (granular corneal dystrophy, corneal scarring) are sometimes included for contrast, which is clearly denoted.
Results
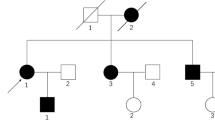
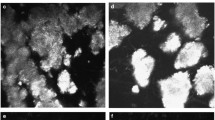
LCD is a slowly progressive disease that leads to recurrent epithelial corneal erosions, stromal haze, corneal opacification, substantial discomfort, and visual impairment. Due to its autosomal-dominant inheritance pattern, this disease can persist throughout ancestral lines and requires consistent treatment and follow-up. An optimal management plan is necessary to (1) prolong years of life with best achievable visual acuity; (2) treat painful recurrent corneal erosions as they occur; (3) ensure proper follow-up throughout the life of a patient, as well as monitor at-risk offspring; and (4) monitor efficacy of treatment.
Conclusions
This paper addresses (1) treatment for early disease including corneal epithelial debridement, photo therapeutic keratectomy (PTK), femtosecond laser–assisted lamellar keratectomy (FLK), and others; (2) treatment for late disease including full thickness keratoplasties and anterior lamellar keratoplasties; and (3) potential future treatment considerations including a wide variety of topical/systemic, genetic, and regenerative approaches.



Similar content being viewed by others
References
Weiss JS, Moller HU, Aldave AJ, Seitz B, Bredrup C, Kivela T, Munier FL, Rapuano CJ, Nischal KK, Kim EK, Sutphin J, Busin M, Labbe A, Kenyon KR, Kinoshita S, Lisch W (2015) IC3D classification of corneal dystrophies–edition 2. Cornea 34:117–159. https://doi.org/10.1097/ICO.0000000000000307
Munier FL, Frueh BE, Othenin-Girard P, Uffer S, Cousin P, Wang MX, Heon E, Black GC, Blasi MA, Balestrazzi E, Lorenz B, Escoto R, Barraquer R, Hoeltzenbein M, Gloor B, Fossarello M, Singh AD, Arsenijevic Y, Zografos L, Schorderet DF (2002) BIGH3 mutation spectrum in corneal dystrophies. Invest Ophthalmol Vis Sci 43:949–954
Kheir V, Cortes-Gonzalez V, Zenteno JC, Schorderet DF (2019) Mutation update: TGFBI pathogenic and likely pathogenic variants in corneal dystrophies. Hum Mutat 40:675–693. https://doi.org/10.1002/humu.23737
Fukuoka H, Kawasaki S, Yamasaki K, Matsuda A, Fukumoto A, Murakami A, Kinoshita S (2010) Lattice corneal dystrophy type IV (p.Leu527Arg) is caused by a founder mutation of the TGFBI gene in a single Japanese ancestor. Invest Ophthalmol Vis Sci 51:4523–4530. https://doi.org/10.1167/iovs.10-5343
Runager K, Basaiawmoit RV, Deva T, Andreasen M, Valnickova Z, Sorensen CS, Karring H, Thogersen IB, Christiansen G, Underhaug J, Kristensen T, Nielsen NC, Klintworth GK, Otzen DE, Enghild JJ (2011) Human phenotypically distinct TGFBI corneal dystrophies are linked to the stability of the fourth FAS1 domain of TGFBIp. J Biol Chem 286:4951–4958. https://doi.org/10.1074/jbc.M110.181099
Han J, Zhang M, Lin HY, Huang FY, Lin YY, Tan GH, Zheng ZY (2019) Impaired autophagic degradation of transforming growth factor-beta-induced protein by macrophages in lattice corneal dystrophy. Invest Ophthalmol Vis Sci 60:978–989. https://doi.org/10.1167/iovs.18-25838
Poulsen ET, Runager K, Risor MW, Dyrlund TF, Scavenius C, Karring H, Praetorius J, Vorum H, Otzen DE, Klintworth GK, Enghild JJ (2014) Comparison of two phenotypically distinct lattice corneal dystrophies caused by mutations in the transforming growth factor beta induced (TGFBI) gene. Proteomics Clin Appl 8:168–177. https://doi.org/10.1002/prca.201300058
Courtney DG, Poulsen ET, Kennedy S, Moore JE, Atkinson SD, Maurizi E, Nesbit MA, Moore CB, Enghild JJ (2015) Protein composition of TGFBI-R124C- and TGFBI-R555W-associated aggregates suggests multiple mechanisms leading to lattice and granular corneal dystrophy. Invest Ophthalmol Vis Sci 56:4653–4661. https://doi.org/10.1167/iovs.15-16922
Grothe HL, Little MR, Sjogren PP, Chang AA, Nelson EF, Yuan C (2013) Altered protein conformation and lower stability of the dystrophic transforming growth factor beta-induced protein mutants. Mol Vis 19:593–603
Underhaug J, Koldso H, Runager K, Nielsen JT, Sorensen CS, Kristensen T, Otzen DE, Karring H, Malmendal A, Schiott B, Enghild JJ, Nielsen NC (2013) Mutation in transforming growth factor beta induced protein associated with granular corneal dystrophy type 1 reduces the proteolytic susceptibility through local structural stabilization. Biochim Biophys Acta 1834:2812–2822. https://doi.org/10.1016/j.bbapap.2013.10.008
Kolozsvari L, Nogradi A, Hopp B, Bor Z (2002) UV absorbance of the human cornea in the 240- to 400-nm range. Invest Ophthalmol Vis Sci 43:2165–2168
Kitamoto K, Taketani Y, Fujii W, Inamochi A, Toyono T, Miyai T, Yamagami S, Kuroda M, Usui T, Ouchi Y (2020) Generation of mouse model of TGFBI-R124C corneal dystrophy using CRISPR/Cas9-mediated homology-directed repair. Sci Rep 10:2000. https://doi.org/10.1038/s41598-020-58876-w
Ellies P, Renard G, Valleix S, Boelle PY, Dighiero P (2002) Clinical outcome of eight BIGH3-linked corneal dystrophies. Ophthalmology 109:793–797. https://doi.org/10.1016/s0161-6420(01)01025-9
Mohamed A, Chaurasia S, Ramappa M, Murthy SI, Garg P (2018) Outcomes of keratoplasty in lattice corneal dystrophy in a large cohort of Indian eyes. Indian J Ophthalmol 66:666–672. https://doi.org/10.4103/ijo.IJO_1150_17
Meisler DM, Fine M (1984) Recurrence of the clinical signs of lattice corneal dystrophy (type I) in corneal transplants. Am J Ophthalmol 97:210–214. https://doi.org/10.1016/s0002-9394(14)76092-1
Snead DR, Mathews BN (2002) Differences in amyloid deposition in primary and recurrent corneal lattice dystrophy type 1. Cornea 21:308–311. https://doi.org/10.1097/00003226-200204000-00014
Lisch W, Seitz B (2014) Lattice corneal dystrophy type 1: an epithelial or stromal entity? Cornea 33:1109–1112. https://doi.org/10.1097/ICO.0000000000000202
Ramsay RM (1960) Familial corneal dystrophy-lattice type. Trans Can Opthalmolog Soc 23:222–229
Marles S (1994) Linkage studies of lattice corneal dystrophy type 1. Master’s thesis. University of Manitoba
Pereira-Souza AL, Ambrosio R Jr, Bandeira F, Salomao MQ, Souza Lima A, Wilson SE (2022) Topical losartan for treating corneal fibrosis (haze): first clinical experience. J Refract Surg 38:741–746. https://doi.org/10.3928/1081597X-20221018-02
Miller DD, Hasan SA, Simmons NL, Stewart MW (2019) Recurrent corneal erosion: a comprehensive review. Clin Ophthalmol 13:325–335. https://doi.org/10.2147/OPTH.S157430
Kato Y, Yagi H, Kaji Y, Oshika T, Goto Y (2013) Benzalkonium chloride accelerates the formation of the amyloid fibrils of corneal dystrophy-associated peptides. J Biol Chem 288:25109–25118. https://doi.org/10.1074/jbc.M113.477695
Bae SS, Chan CC (2018) Superficial keratectomy: indications and outcomes. Can J Ophthalmol 53:553–559. https://doi.org/10.1016/j.jcjo.2018.01.030
Cohen EJ (2007) Debridement for visual symptoms resulting from anterior basement membrane corneal dystrophy. Am J Ophthalmol 144:288–289. https://doi.org/10.1016/j.ajo.2007.05.024
Itty S, Hamilton SS, Baratz KH, Diehl NN, Maguire LJ (2007) Outcomes of epithelial debridement for anterior basement membrane dystrophy. Am J Ophthalmol 144:217–221. https://doi.org/10.1016/j.ajo.2007.04.024
Soong HK, Farjo Q, Meyer RF, Sugar A (2002) Diamond burr superficial keratectomy for recurrent corneal erosions. Br J Ophthalmol 86:296–298. https://doi.org/10.1136/bjo.86.3.296
Sridhar MS, Rapuano CJ, Cosar CB, Cohen EJ, Laibson PR (2002) Phototherapeutic keratectomy versus diamond burr polishing of Bowman’s membrane in the treatment of recurrent corneal erosions associated with anterior basement membrane dystrophy. Ophthalmology 109:674–679. https://doi.org/10.1016/s0161-6420(01)01027-2
Tzelikis PF, Rapuano CJ, Hammersmith KM, Laibson PR, Cohen EJ (2005) Diamond burr treatment of poor vision from anterior basement membrane dystrophy. Am J Ophthalmol 140:308–310. https://doi.org/10.1016/j.ajo.2005.01.036
Vo RC, Chen JL, Sanchez PJ, Yu F, Aldave AJ (2015) Long-term outcomes of epithelial debridement and diamond burr polishing for corneal epithelial irregularity and recurrent corneal erosion. Cornea 34:1259–1265. https://doi.org/10.1097/ICO.0000000000000554
Morita Y, Chikama T, Yamada N, Morishige N, Sonoda KH, Nishida T (2012) New mode of treatment for lattice corneal dystrophy type I: corneal epithelial debridement and fibronectin eye drops. Jpn J Ophthalmol 56:26–30. https://doi.org/10.1007/s10384-011-0104-5
Das S, Langenbucher A, Seitz B (2005) Delayed healing of corneal epithelium after phototherapeutic keratectomy for lattice dystrophy. Cornea 24:283–287. https://doi.org/10.1097/01.ico.0000138853.26332.55
Foerster CG, Langenbucher A, Cursiefen C, Kruse FE, Seitz B (2007) Delayed epithelial healing after keratoplasty for lattice corneal dystrophy. Cornea 26:1182–1183. https://doi.org/10.1097/ICO.0b013e318151f8cc
Kawamoto K, Morishige N, Yamada N, Chikama T, Nishida T (2006) Delayed corneal epithelial wound healing after penetrating keratoplasty in individuals with lattice corneal dystrophy. Am J Ophthalmol 142:173–174. https://doi.org/10.1016/j.ajo.2006.01.077
Nemeth G, Felszeghy S, Kenyeres A, Szentmary N, Berta A, Suveges I, Modis L (2008) Cell adhesion molecules in stromal corneal dystrophies. Histol Histopathol 23:945–952. https://doi.org/10.14670/HH-23.945
Resch MD, Schlotzer-Schrehardt U, Hofmann-Rummelt C, Kruse FE, Seitz B (2009) Alterations of epithelial adhesion molecules and basement membrane components in lattice corneal dystrophy (LCD). Graefes Arch Clin Exp Ophthalmol 247:1081–1088. https://doi.org/10.1007/s00417-009-1046-1
Dinh R, Rapuano CJ, Cohen EJ, Laibson PR (1999) Recurrence of corneal dystrophy after excimer laser phototherapeutic keratectomy. Ophthalmology 106:1490–1497. https://doi.org/10.1016/S0161-6420(99)90441-4
Reddy JC, Rapuano CJ, Nagra PK, Hammersmith KM (2013) Excimer laser phototherapeutic keratectomy in eyes with corneal stromal dystrophies with and without a corneal graft. Am J Ophthalmol 155(1111–1118):e1112. https://doi.org/10.1016/j.ajo.2012.12.016
Sauvageot P, Julio G, Bolanos JV, Carrera M, de Toledo JA, Barraquer RI (2022) Recurrence and visual outcomes of phototherapeutic keratectomy in lattice corneal dystrophy: a cohort study. J Refract Surg 38:43–49. https://doi.org/10.3928/1081597X-20211104-01
Ellies P, Bejjani RA, Bourges JL, Boelle PY, Renard G, Dighiero P (2003) Phototherapeutic keratectomy for BIGH3-linked corneal dystrophy recurring after penetrating keratoplasty. Ophthalmology 110:1119–1125. https://doi.org/10.1016/S0161-6420(03)00229-X
Hieda O, Sotozono C, Nakamura Y, Wakimasu K, Kinoshita S (2021) Surgical outcomes of re-excimer laser phototherapeutic keratectomy (re-PTK). Sci Rep 11:11503. https://doi.org/10.1038/s41598-021-91121-6
Das S, Langenbucher A, Seitz B (2005) Excimer laser phototherapeutic keratectomy for granular and lattice corneal dystrophy: a comparative study. J Refract Surg 21:727–731. https://doi.org/10.3928/1081-597X-20051101-12
Stewart OG, Pararajasegaram P, Cazabon J, Morrell AJ (2002) Visual and symptomatic outcome of excimer phototherapeutic keratectomy (PTK) for corneal dystrophies. Eye (Lond) 16:126–131. https://doi.org/10.1038/sj.eye.6700049
Orndahl MJ, Fagerholm PP (1998) Treatment of corneal dystrophies with phototherapeutic keratectomy. J Refract Surg 14:129–135. https://doi.org/10.3928/1081-597X-19980301-11
Nagpal R, Maharana PK, Roop P, Murthy SI, Rapuano CJ, Titiyal JS, Vajpayee RB, Sharma N (2020) Phototherapeutic keratectomy. Surv Ophthalmol 65:79–108. https://doi.org/10.1016/j.survophthal.2019.07.002
Steger B, Romano V, Biddolph S, Willoughby CE, Batterbury M, Kaye SB (2016) Femtosecond laser-assisted lamellar keratectomy for corneal opacities secondary to anterior corneal dystrophies: an interventional case series. Cornea 35:6–13. https://doi.org/10.1097/ICO.0000000000000665
Lee J, Kim JH, Lee D, Chang JW, Shin JY, Seo JW, Seo MH, Moon NJ (2018) Long-term clinical outcome of femtosecond laser-assisted lamellar keratectomy with phototherapeutic keratectomy in anterior corneal stromal dystrophy. Br J Ophthalmol 102:31–36. https://doi.org/10.1136/bjophthalmol-2017-310189
Arora T, Arora S, Sharma V (2016) Femtosecond laser-assisted lamellar keratectomy for anterior corneal dystrophies. Cornea 35:e7-8. https://doi.org/10.1097/ICO.0000000000000744
Kawashima M, Kawakita T, Den S, Shimmura S, Tsubota K, Shimazaki J (2006) Comparison of deep lamellar keratoplasty and penetrating keratoplasty for lattice and macular corneal dystrophies. Am J Ophthalmol 142:304–309. https://doi.org/10.1016/j.ajo.2006.03.057
Akhtar S, Meek KM, Ridgway AE, Bonshek RE, Bron AJ (1999) Deposits and proteoglycan changes in primary and recurrent granular dystrophy of the cornea. Arch Ophthalmol 117:310–321. https://doi.org/10.1001/archopht.117.3.310
Spelsberg H, Reinhard T, Henke L, Berschick P, Sundmacher R (2004) Penetrating limbo-keratoplasty for granular and lattice corneal dystrophy: survival of donor limbal stem cells and intermediate-term clinical results. Ophthalmology 111:1528–1533. https://doi.org/10.1016/j.ophtha.2004.01.030
Lang SJ, Eberwein P, Reinshagen H, Reinhard T, Sundmacher R (2015) Simultaneous transplantation of limbal stem cells may reduce recurrences of granular dystrophy after corneal transplantation: 2 long-term case reports. Medicine (Baltimore) 94:e789. https://doi.org/10.1097/MD.0000000000000789
Vandemeulebroecke N, Spelsberg H (2008) Homologous penetrating central limbokeratoplasty versus conventional homologous penetrating keratoplasty and hard contact lens in granular and lattice corneal dystrophy. Invest Ophthalmol Vis Sci 49:2334
Espandar L, Carlson AN (2013) Lamellar keratoplasty: a literature review. J Ophthalmol 2013:894319. https://doi.org/10.1155/2013/894319
Jimenez-Alfaro I, Perez-Santonja JJ, Gomez Telleria G, Bueno Palacin JL, Puy P (2001) Therapeutic lamellar keratoplasty with an automated microkeratome. J Cataract Refract Surg 27:1161–1165. https://doi.org/10.1016/s0886-3350(00)00889-0
Vajpayee RB, Vasudendra N, Titiyal JS, Tandon R, Sharma N, Sinha R (2006) Automated lamellar therapeutic keratoplasty (ALTK) in the treatment of anterior to mid-stromal corneal pathologies. Acta Ophthalmol Scand 84:771–773. https://doi.org/10.1111/j.1600-0420.2006.00722.x
Tan DT, Ang LP (2006) Modified automated lamellar therapeutic keratoplasty for keratoconus: a new technique. Cornea 25:1217–1219. https://doi.org/10.1097/01.ico.0000248388.39767.42
Yoo SH, Kymionis GD, Koreishi A, Ide T, Goldman D, Karp CL, O’Brien TP, Culbertson WW, Alfonso EC (2008) Femtosecond laser-assisted sutureless anterior lamellar keratoplasty. Ophthalmology 115:1303–1307. https://doi.org/10.1016/j.ophtha.2007.10.037. (1307 e1301)
Shetty R, Nagaraja H, Veluri H, Shivanna Y, Kugar T, Nujits R, Shetty B (2014) Sutureless femtosecond anterior lamellar keratoplasty: a 1-year follow-up study. Indian J Ophthalmol 62:923–926. https://doi.org/10.4103/0301-4738.143928
Shousha MA, Yoo SH, Kymionis GD, Ide T, Feuer W, Karp CL, O’Brien TP, Culbertson WW, Alfonso E (2011) Long-term results of femtosecond laser-assisted sutureless anterior lamellar keratoplasty. Ophthalmology 118:315–323. https://doi.org/10.1016/j.ophtha.2010.06.037
Yuen LH, Mehta JS, Shilbayeh R, Lim L, Tan DT (2011) Hemi-automated lamellar keratoplasty (HALK). Br J Ophthalmol 95:1513–1518. https://doi.org/10.1136/bjophthalmol-2011-300195
Fuest M, Liu YC, Arundhati A, Li L, Tan D, Mehta JS (2018) Long-term outcomes of hemi-automated lamellar keratoplasty. Clin Exp Ophthalmol 46:1017–1027. https://doi.org/10.1111/ceo.13331
Acar BT, Arslan OS, Buttanri IB, Sevim MS, Acar S (2011) Comparing deep anterior lamellar keratoplasty and automated lamellar therapeutic keratoplasty in patients with keratoconus. Jpn J Ophthalmol 55:327–332. https://doi.org/10.1007/s10384-011-0044-0
Reda A (2020) Deep anterior lamellar keratoplasty versus penetrating keratoplasty in the treatment of stromal corneal dystrophies. Int Eye Sci 20:1118–1125
Schmidinger G, Seiler TG, Donner R (2022) Femtosecond-laser assisted deep anterior lamellar keratoplasty (F-DALK). In: Armia A, Mazzotta C (eds) Keratoconus. Springer, pp 213–222
Chamberlain WD (2019) Femtosecond laser-assisted deep anterior lamellar keratoplasty. Curr Opin Ophthalmol 30:256–263. https://doi.org/10.1097/ICU.0000000000000574
Reinhart WJ, Musch DC, Jacobs DS, Lee WB, Kaufman SC, Shtein RM (2011) Deep anterior lamellar keratoplasty as an alternative to penetrating keratoplasty: a report by the American Academy of Ophthalmology. Ophthalmology 118:209–218. https://doi.org/10.1016/j.ophtha.2010.11.002
Borderie VM, Sandali O, Bullet J, Gaujoux T, Touzeau O, Laroche L (2012) Long-term results of deep anterior lamellar versus penetrating keratoplasty. Ophthalmology 119:249–255. https://doi.org/10.1016/j.ophtha.2011.07.057
Infantes Molina EJ, Celis Sanchez J, Tenias Burilllo JM, Diaz Valle D, Benitez-Del-Castillo JM, Mesa Varona D, Avendano-Cantos E (2019) Deep anterior lamellar keratoplasty versus penetrating keratoplasty in corneas showing a high or low graft rejection risk. Eur J Ophthalmol 29:295–303. https://doi.org/10.1177/1120672118797287
Marcon AS, Cohen EJ, Rapuano CJ, Laibson PR (2003) Recurrence of corneal stromal dystrophies after penetrating keratoplasty. Cornea 22:19–21. https://doi.org/10.1097/00003226-200301000-00005
Unal M, Arslan OS, Atalay E, Mangan MS, Bilgin AB (2013) Deep anterior lamellar keratoplasty for the treatment of stromal corneal dystrophies. Cornea 32:301–305. https://doi.org/10.1097/ICO.0b013e31825718ca
Courtney DG, Atkinson SD, Moore JE, Maurizi E, Serafini C, Pellegrini G, Black GC, Manson FD, Yam GH, Macewen CJ, Allen EH, McLean WH, Moore CB (2014) Development of allele-specific gene-silencing siRNAs for TGFBI Arg124Cys in lattice corneal dystrophy type I. Invest Ophthalmol Vis Sci 55:977–985. https://doi.org/10.1167/iovs.13-13279
Christie KA, Robertson LJ, Conway C, Blighe K, DeDionisio LA, Chao-Shern C, Kowalczyk AM, Marshall J, Turnbull D, Nesbit MA, Moore CBT (2020) Mutation-independent allele-specific editing by CRISPR-Cas9, a novel approach to treat autosomal dominant disease. Mol Ther 28:1846–1857. https://doi.org/10.1016/j.ymthe.2020.05.002
Kim EK, Kim S, Maeng YS (2019) Generation of TGFBI knockout ABCG2+/ABCB5+ double-positive limbal epithelial stem cells by CRISPR/Cas9-mediated genome editing. PLoS ONE 14:e0211864. https://doi.org/10.1371/journal.pone.0211864
Christie KA, Courtney DG, DeDionisio LA, Shern CC, De Majumdar S, Mairs LC, Nesbit MA, Moore CBT (2017) Towards personalised allele-specific CRISPR gene editing to treat autosomal dominant disorders. Sci Rep 7:16174. https://doi.org/10.1038/s41598-017-16279-4
Weiss JS, Willoughby CE, Abad-Morales V, Turunen JA, Lisch W (2022) Update on the corneal dystrophies-genetic testing and therapy. Cornea 41:1337–1344. https://doi.org/10.1097/ICO.0000000000002857
Lee JH, Kim MJ, Ha SW, Kim HK (2016) Autologous platelet-rich plasma eye drops in the treatment of recurrent corneal erosions. Korean J Ophthalmol 30:101–107. https://doi.org/10.3341/kjo.2016.30.2.101
Anitua E, Muruzabal F, Tayebba A, Riestra A, Perez VL, Merayo-Lloves J, Orive G (2015) Autologous serum and plasma rich in growth factors in ophthalmology: preclinical and clinical studies. Acta Ophthalmol 93:e605-614. https://doi.org/10.1111/aos.12710
Gupta A, Monroy D, Ji Z, Yoshino K, Huang A, Pflugfelder SC (1996) Transforming growth factor beta-1 and beta-2 in human tear fluid. Curr Eye Res 15:605–614. https://doi.org/10.3109/02713689609008900
Nishida T, Nakamura M, Ofuji K, Reid TW, Mannis MJ, Murphy CJ (1996) Synergistic effects of substance P with insulin-like growth factor-1 on epithelial migration of the cornea. J Cell Physiol 169:159–166. https://doi.org/10.1002/(SICI)1097-4652(199610)169:1%3c159::AID-JCP16%3e3.0.CO;2-8
van Setten GB, Viinikka L, Tervo T, Pesonen K, Tarkkanen A, Perheentupa J (1989) Epidermal growth factor is a constant component of normal human tear fluid. Graefe’s Arch Clin Exp Ophthalmol 227:184–187
Liu L, Hartwig D, Harloff S, Herminghaus P, Wedel T, Geerling G (2005) An optimised protocol for the production of autologous serum eyedrops. Graefes Arch Clin Exp Ophthalmol 243:706–714. https://doi.org/10.1007/s00417-004-1106-5
Tsubota K, Goto E, Fujita H, Ono M, Inoue H, Saito I, Shimmura S (1999) Treatment of dry eye by autologous serum application in Sjogren’s syndrome. Br J Ophthalmol 83:390–395. https://doi.org/10.1136/bjo.83.4.390
Kamiya K, Takahashi M, Shoji N (2021) Effect of platelet-rich plasma on corneal epithelial healing after phototherapeutic keratectomy: an intraindividual contralateral randomized sstudy. Biomed Res Int 2021:5752248. https://doi.org/10.1155/2021/5752248
Chakrabarty K, Shetty R, Ghosh A (2018) Corneal cell therapy: with iPSCs, it is no more a far-sight. Stem Cell Res Ther 9:287. https://doi.org/10.1186/s13287-018-1036-5
Stenvang M, Schafer NP, Malmos KG, Perez AW, Niembro O, Sormanni P, Basaiawmoit RV, Christiansen G, Andreasen M, Otzen DE (2018) Corneal dystrophy mutations drive pathogenesis by targeting TGFBIp stability and solubility in a latent amyloid-forming domain. J Mol Biol 430:1116–1140. https://doi.org/10.1016/j.jmb.2018.03.001
Nielsen NS, Poulsen ET, Lukassen MV, Chao Shern C, Mogensen EH, Weberskov CE, DeDionisio L, Schauser L, Moore TCB, Otzen DE, Hjortdal J, Enghild JJ (2020) Biochemical mechanisms of aggregation in TGFBI-linked corneal dystrophies. Prog Retin Eye Res 77:100843. https://doi.org/10.1016/j.preteyeres.2020.100843
Stenvang M, Christiansen G, Otzen DE (2016) Epigallocatechin gallate remodels fibrils of lattice corneal dystrophy protein, facilitating proteolytic degradation and preventing formation of membrane-permeabilizing species. Biochemistry 55:2344–2357. https://doi.org/10.1021/acs.biochem.6b00063
Venkatraman A, Murugan E, Lin SJ, Peh GSL, Rajamani L, Mehta JS (2020) Effect of osmolytes on in-vitro aggregation properties of peptides derived from TGFBIp. Sci Rep 10:4011. https://doi.org/10.1038/s41598-020-60944-0
Choi SI, Kim BY, Dadakhujaev S, Jester JV, Ryu H, Kim TI, Kim EK (2011) Inhibition of TGFBIp expression by lithium: implications for TGFBI-linked corneal dystrophy therapy. Invest Ophthalmol Vis Sci 52:3293–3300. https://doi.org/10.1167/iovs.10-6405
Nie D, Peng Y, Li M, Liu X, Zhu M, Ye L (2018) Lithium chloride (LiCl) induced autophagy and downregulated expression of transforming growth factor beta-induced protein (TGFBI) in granular corneal dystrophy. Exp Eye Res 173:44–50. https://doi.org/10.1016/j.exer.2018.04.008
Zhu F, Li M, Zhang C, Chen C, Ying F, Nie D (2021) In vivo confocal microscopy qualitative investigation of the relationships between lattice corneal dystrophy deposition and corneal nerves. BMC Ophthalmol 21:449. https://doi.org/10.1186/s12886-021-02149-1
Shetty R, Naidu JR, Nair AP, Vaidya TA, D’Souza S, Matalia H, Deshpande V, Sethu S, Ghosh A, Chakrabarty K (2020) Distinct ocular surface soluble factor profile in human corneal dystrophies. Ocul Surf 18:237–248. https://doi.org/10.1016/j.jtos.2019.11.007
Sciriha GG, Seiler TG, Donner R (2022) Identifying and categorizing compounds that reduce corneal transforming growth factor beta induced protein levels: a sco** review. Expert Rev Clin Pharmacol 15:1423–1442. https://doi.org/10.1080/17512433.2022.2142560
Sampaio LP, Hilgert GSL, Shiju TM, Murillo SE, Santhiago MR, Wilson SE (2022) Topical losartan inhibits corneal scarring fibrosis and collagen type IV deposition after Descemet’s membrane-endothelial excision in rabbits. Exp Eye Res 216:108940. https://doi.org/10.1016/j.exer.2022.108940
Sampaio LP, Hilgert GSL, Shiju TM, Santhiago MR, Wilson SE (2022) Topical losartan and corticosteroid additively inhibit corneal stromal myofibroblast generation and scarring fibrosis after alkali burn injury. Transl Vis Sci Technol 11:9. https://doi.org/10.1167/tvst.11.7.9
Salman M, Verma A, Singh VK, Jaffet J, Chaurasia S, Sahel DK, Ramappa M, Singh V (2022) New frontier in the management of corneal dystrophies: basics, development, and challenges in corneal gene therapy and gene editing. Asia Pac J Ophthalmol (Phila) 11:346–359. https://doi.org/10.1097/APO.0000000000000443
Acknowledgements
We are grateful to Jayne S. Weiss, MD and Anthony J. Aldave, MD for their critical review and comments during the preparation of this manuscript. As well, we would like to thank Jeffrey Judelson, MD and Walter Lisch, MD for providing images for use in this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. The first draft of the article was written by S. G., MD and G. R., MD. The final manuscript has been re-designed, completed, and edited by E. M., MD and G. R., MD. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This article does not require ethics approval as no studies with human participants, animals, or cell lines were performed by any of the authors.
Consent for publication
Informed consent for publication of the figure was obtained from Dr. Jeffrey Judelson.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Milovanova, E., Gomon, S. & Rocha, G. Classic lattice corneal dystrophy: a brief review and summary of treatment modalities. Graefes Arch Clin Exp Ophthalmol 262, 1667–1681 (2024). https://doi.org/10.1007/s00417-023-06297-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-023-06297-6