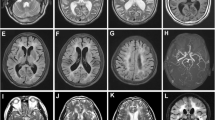
Abstract
Cerebral autosomal dominant arteriopathy with subcortical infarcts and leucoencephalopathy (CADASIL) represents the most common monogenic cause of adult-onset ischemic stroke and vascular dementia. It is caused by heterozygous missense mutations in the NOTCH3 gene, encoding a transmembrane receptor protein on vascular smooth muscle cells. Classical CADASIL mutations affect conserved cysteine residues of the Notch3 protein. By contrast, the role of non-canonical genetic variation in NOTCH3, in particular of variants causing a hypomorphic Notch3 protein, is subject to an ongoing scientific debate. In this context, we here report a novel NOTCH3 frameshift variant in exon 18 (NM_000435.2: c.2853_2857delTCCCG), causing a frameshift and introducing a premature stop codon, which was detected in a 43-year-old woman and her father. Both carriers of the variant were carefully evaluated, including serial follow-up in the index. Neither clinical nor imaging features provided convincing evidence for a classical CADASIL phenotype, thus reinforcing the concept of hypomorphic NOTCH3 variants most likely not being causative for CADASIL. Our finding, which is discussed in the light of the published literature, has practical implications for interpreting results of NOTCH3 molecular genetic testing as well as patient counseling.


Similar content being viewed by others
References
Chabriat H, Joutel A, Dichgans M et al (2009) CADASIL. The Lancet Neurol 8:643–653. https://doi.org/10.1016/S1474-4422(09)70127-9
Vahedi K, Chabriat H, Levy C et al (2004) Migraine with aura and brain magnetic resonance imaging abnormalities in patients with CADASIL. Arch Neurol 61:1237–1240. https://doi.org/10.1001/archneur.61.8.1237
Guey S, Mawet J, Hervé D et al (2015) Prevalence and characteristics of migraine in CADASIL. Cephalalgia. https://doi.org/10.1177/0333102415620909
Dichgans M, Holtmannspötter M, Herzog J et al (2002) Cerebral microbleeds in CADASIL: a gradient-echo magnetic resonance imaging and autopsy study. Stroke 33:67–71
Joutel A, Corpechot C, Ducros A et al (1996) Notch3 mutations in CADASIL, a hereditary adult-onset condition causing stroke and dementia. Nature 383:707–710. https://doi.org/10.1038/383707a0
Peters N, Opherk C, Bergmann T et al (2005) Spectrum of mutations in biopsy-proven CADASIL: implications for diagnostic strategies. Arch Neurol 62:1091–1094. https://doi.org/10.1001/archneur.62.7.1091
Duering M, Karpinska A, Rosner S et al (2011) Co-aggregate formation of CADASIL-mutant NOTCH3: a single-particle analysis. Hum Mol Genet 20:3256–3265. https://doi.org/10.1093/hmg/ddr237
Tikka S, Mykkänen K, Ruchoux M-M et al (2009) Congruence between NOTCH3 mutations and GOM in 131 CADASIL patients. Brain 132:933–939. https://doi.org/10.1093/brain/awn364
Joutel A, Andreux F, Gaulis S et al (2000) The ectodomain of the Notch3 receptor accumulates within the cerebrovasculature of CADASIL patients. J Clin Invest 105:597–605. https://doi.org/10.1172/JCI8047
Arboleda-Velasquez JF, Manent J, Lee JH et al (2011) Hypomorphic Notch 3 alleles link Notch signaling to ischemic cerebral small-vessel disease. Proc Natl Acad Sci USA 108:E128–E135. https://doi.org/10.1073/pnas.1101964108
Markus HS, Martin RJ, Simpson MA et al (2002) Diagnostic strategies in CADASIL. Neurology 59:1134–1138
Auer DP, Pütz B, Gössl C et al (2001) Differential lesion patterns in CADASIL and sporadic subcortical arteriosclerotic encephalopathy: MR imaging study with statistical parametric group comparison. Radiology 218:443–451. https://doi.org/10.1148/radiology.218.2.r01fe24443
O’Sullivan M, Jarosz JM, Martin RJ et al (2001) MRI hyperintensities of the temporal lobe and external capsule in patients with CADASIL. Neurology 56:628–634
Chabriat H, Joutel A, Dichgans M et al (2009) CADASIL. The Lancet Neurol 8:643–653. https://doi.org/10.1016/S1474-4422(09)70127-9
Rutten JW, Boon EMJ, Liem MK et al (2013) Hypomorphic NOTCH3 alleles do not cause CADASIL in humans. Hum Mutat 34:1486–1489. https://doi.org/10.1002/humu.22432
Smith CG, Naven M, Harris R et al (2013) Exome resequencing identifies potential tumor-suppressor genes that predispose to colorectal cancer. Hum Mutat 34:1026–1034. https://doi.org/10.1002/humu.22333
Moccia M, Mosca L, Erro R et al (2015) Hypomorphic NOTCH3 mutation in an Italian family with CADASIL features. Neurobiol Aging 36(547):e5–e11. https://doi.org/10.1016/j.neurobiolaging.2014.08.021
Dotti MT, De Stefano N, Bianchi S et al (2004) A novel NOTCH3 frameshift deletion and mitochondrial abnormalities in a patient with CADASIL. Arch Neurol 61:942–945. https://doi.org/10.1001/archneur.61.6.942
Weiming F, Yuliang W, Youjie L et al (2013) A novel Notch3 deletion mutation in a Chinese patient with cerebral autosomal dominant arteriopathy with subcortical infarcts and leucoencephalopathy (CADASIL). J Clin Neurosci 20:322–323. https://doi.org/10.1016/j.jocn.2012.02.026
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical standards
The study was performed in accordance with the 1964 Declaration of Helsinki and its later amendments. Patients provided their informed consent, including consent to the publication of clinical and genetic findings.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Schubert, V., Bender, B., Kinzel, M. et al. A novel frameshift variant in the CADASIL gene NOTCH3: pathogenic or not?. J Neurol 265, 1338–1342 (2018). https://doi.org/10.1007/s00415-018-8844-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-018-8844-5