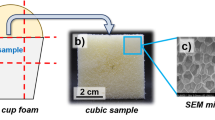
Abstract
Polyurethane (PU) foams are created via the chemical reactions arising after the blending of two initially liquid components (polyols and isocyanates). They are widely used for thermal insulation, for which a small pore size is required. Some of the most efficient pore size–reducing agents have proven to be per- and polyfluorinated carbons (FCs) which are simply added in small quantities to the initially liquid mixture. However, despite their long-standing use, their modes of action have only recently begun to be studied in detail. One widely accepted explanation of their action is that they supposedly suppress diffusional gas exchange between bubbles in the liquid-foam state of the nascent PU foam (foam coarsening). However, using a new double-syringe mixing technique, we show that FCs actually act at a much earlier state of the process: they facilitate the entrainment of tiny air bubbles into PU foam systems during the initial blending process. These bubbles serve as sites for heterogeneous nucleation during the foaming process, and their large number leads to a significant reduction of the characteristic pore size. More importantly, we also demonstrate that the same overall relation is found between the air bubble density and the final pore size for systems with and without FC. Combined with a detailed analysis of the pore size distribution, we argue that the main pore size–reducing effect of FCs is to facilitate air entrainment and that foam aging–related effects only play a minor role.







Similar content being viewed by others
Availability of data and materials
Data are available upon contacting the authors.
References
Eling B, Tomović Ž, Schädler V (2020) Current and future trends in polyurethanes: an industrial perspective. Macromol Chem Phys 221(14):1–11. https://doi.org/10.1002/macp.202000114
Gibson LJ, Ashby MF (1997) Cellular solids; Cambridge University Press: Cambridge. https://doi.org/10.1017/CBO9781139878326
Schuetz MA, Glicksman LR (1984) A basic study of heat transfer through foam insulation. J Cell Plast 20(2):114–121. https://doi.org/10.1177/0021955X8402000203
Glicksman L, Schuetz M, Sinofsky M (1987) Radiation heat transfer in foam insulation. Int J Heat Mass Transf 30(1):187–197. https://doi.org/10.1016/0017-9310(87)90071-8
Estravís S, Tirado-Mediavilla J, Santiago-Calvo M, Ruiz-Herrero JL, Villafañe F, Rodríguez-Pérez MÁ (2016) Rigid polyurethane foams with infused nanoclays: relationship between cellular structure and thermal conductivity. Eur Polym J 80:1–15. https://doi.org/10.1016/j.eurpolymj.2016.04.026
Kim TS, Lee Y, Hwang CH, Song KH, Kim WN (2021) Cryogenic thermal insulating and mechanical properties of rigid polyurethane foams blown with hydrofluoroolefin: effect of perfluoroalkane. J Cell Plast 0(0):1–17. https://doi.org/10.1177/0021955X211062633
Kang JW, Kim JM, Kim MS, Kim YH, Kim WN, Jang W, Shin DS (2009) Effects of nucleating agents on the morphological, mechanical and thermal insulating properties of rigid polyurethane foams. Macromol Res 17(11):856–862. https://doi.org/10.1007/BF03218626
Londrigan ME, Snider SC, Trout KG (1993) K-factor improvement via perfluorinated hydrocarbons. J Cell Plast 29(6):544–555. https://doi.org/10.1177/0021955X9302900603
Yu-Hallada LC, McLellan KP, Wierzbicki RJ, Reichel CJ (1993) Improved rigid insulating polyurethane foams prepared with HCFCs and perfluoroalkanes. J Cell Plast 29(6):589–596. https://doi.org/10.1177/0021955X9302900606
Volkert O (1992) PUR foams prepared with emulsified perfluoroalkanes as blowing agents. J Cell Plast 28(5):486–495. https://doi.org/10.1177/0021955X9202800503
Lee Y, Geun Jang M, Hyung Choi K, Han C, Nyon Kim W (2016) Liquid-type nucleating agent for improving thermal insulating properties of rigid polyurethane foams by HFC-365mfc as a blowing agent. J Appl Polym Sci 133(25). https://doi.org/10.1002/app.43557
Brondi C, Di Maio E, Bertucelli L, Parenti V, Mosciatti T (2021) The effect of organofluorine additives on the morphology, thermal conductivity and mechanical properties of rigid polyurethane and polyisocyanurate foams. J Cell Plast 58(1):121–137. https://doi.org/10.1177/0021955X20987152
Hamann M, Andrieux S, Schütte M, Telkemeyer D, Ranft M, Drenckhan W (2023) Quantitative investigation of the pore size reducing effect of perfluorocarbons in polyurethane foaming. Colloid Polym Sci 301(7):763–773. https://doi.org/10.1007/s00396-023-05107-z
Hodnebrog, Etminan M, Fuglestvedt JS, Marston G, Myhre G, Nielsen CJ, Shine KP, Wallington TJ (2013) Global warming potentials and radiative efficiencies of halocarbons and related compounds: a comprehensive review. Rev Geophys 51(2):300–378. https://doi.org/10.1002/rog.20013
Jain AK, Briegleb BP, Minschwaner K, Wuebbles DJ (2000) Radiative forcings and global warming potentials of 39 greenhouse gases. J Geophys Res Atmos 105(D16):20773–20790. https://doi.org/10.1029/2000JD900241
Mühle J, Ganesan AL, Miller BR, Salameh PK, Harth CM, Greally BR, Rigby M, Porter LW, Steele LP, Trudinger CM, Krummel PB, O’Doherty S, Fraser PJ, Simmonds PG, Prinn RG, Weiss RF (2010) Perfluorocarbons in the global atmosphere: tetrafluoromethane, hexafluoroethane, and octafluoropropane. Atmos Chem Phys 10(11):5145–5164. https://doi.org/10.5194/acp-10-5145-2010
Klostermann M, Schiller C, Venzmer J, Eilbracht C (2018) Production of fine cell foams using a cell aging inhibitor. US 2018/0327563 A1
Creazzo JA (2007) Blowing agents for forming foam comprising unsaturated fluorocarbons. US 2007/0100010 A1
Brondi C, Mosciatti T, Di Maio E (2022) Ostwald ripening modulation by organofluorine additives in rigid polyurethane foams. Ind Eng Chem Res 61(40):14868–14880. https://doi.org/10.1021/acs.iecr.2c01829
Kanner B, Decker TG (1969) Urethane foam formation— role of the silicone surfactant. J Cell Plast 5(1):32–39. https://doi.org/10.1177/0021955X6900500104
Grünbauer HJM, Folmer JCW (1994) Polymer morphology of CO2-blown rigid polyurethane foams: its fractal nature. J Appl Polym Sci 54(7):935–949. https://doi.org/10.1002/app.1994.070540712
Reignier J, Alcouffe P, Méchin F, Fenouillot F (2019) The morphology of rigid polyurethane foam matrix and its evolution with time during foaming – new insight by cryogenic scanning electron microscopy. J Colloid Interface Sci 552:153–165. https://doi.org/10.1016/j.jcis.2019.05.032
Merillas B, Villafañe F, Rodríguez-Pérez MÁ (2021) Nanoparticles addition in Pu foams: the dramatic effect of trapped-air on nucleation. Polymers (Basel) 13(17):1–11. https://doi.org/10.3390/polym13172952
Hamann M, Andrieux S, Schütte M, Telkemeyer D, Ranft M, Drenckhan W (2023) Directing the pore size of rigid polyurethane foam via controlled air entrainment. J Cell Plast 0(0):1–14. https://doi.org/10.1177/0021955X231152680
Kong M, Bhattacharya RN, James C, Basu A (2005) A statistical approach to estimate the 3D size distribution of spheres from 2D size distributions. GSA Bull 117(1–2):244–249. https://doi.org/10.1130/B25000.1
Pinto J, Solórzano E, Rodriguez-Perez MA, De Saja JA (2013) Characterization of the cellular structure based on user-interactive image analysis procedures. J Cell Plast 49(6):555–575. https://doi.org/10.1177/0021955X13503847
Gaillard T, Roché M, Honorez C, Jumeau M, Balan A, Jedrzejczyk C, Drenckhan W (2017) Controlled foam generation using cyclic diphasic flows through a constriction. Int J Multiph Flow 96:173–187. https://doi.org/10.1016/j.ijmultiphaseflow.2017.02.009
Chernyshev VS, Skliar M (2014) Surface tension of water in the presence of perfluorocarbon vapors. Soft Matter 10(12):1937–1943. https://doi.org/10.1039/c3sm52289j
Steck K, Hamann M, Andrieux S, Muller P, Kékicheff P, Stubenrauch C, Drenckhan W (2021) Fluorocarbon vapors slow down coalescence in foams. Adv Mater Interfaces 2100723. https://doi.org/10.1002/admi.202100723
Shi D, Liu X, Counil C, Krafft MP (2019) Fluorocarbon exposure mode markedly affects phospholipid monolayer behavior at the gas/liquid interface: impact on size and stability of microbubbles. Langmuir 35(31):10025–10033. https://doi.org/10.1021/acs.langmuir.8b03546
Gerber F, Krafft MP, Vandamme TF, Goldmann M, Fontaine P (2006) Fluidization of a dipalmitoyl phosphatidylcholine monolayer by fluorocarbon gases: potential use in lung surfactant therapy. Biophys J 90(9):3184–3192. https://doi.org/10.1529/biophysj.105.077008
Ando Y, Tabata H, Sanchez M, Cagna A, Koyama D, Krafft MP (2016) Microbubbles with a self-assembled poloxamer shell and a fluorocarbon inner gas. Langmuir 32(47):12461–12467. https://doi.org/10.1021/acs.langmuir.6b01883
Steck K, Dijoux J, Preisig N, Bouylout V, Stubenrauch C, Drenckhan W (2023) Fluorocarbon vapors slow down coalescence in foams: influence of surfactant concentration. Colloid Polym Sci 301(7):685–695. https://doi.org/10.1007/s00396-023-05129-7
Brondi C, Di Maio E, Bertucelli L, Parenti V, Mosciatti T (2021) Competing bubble formation mechanisms in rigid polyurethane foaming. Polymer (Guildf) 228:123877. https://doi.org/10.1016/j.polymer.2021.123877
Acknowledgements
We are grateful to Leandro Jacomine, Jonathan Dijoux, Aurélie Hourlier-Fargette, Marie-Pierre Krafft, Dominique Langevin, and Katja Steck for precious scientific discussions and technical help. The authors gratefully acknowledge the PLAMICS facility of the Institute Charles Sadron for providing access to the SEM.
Funding
This work has been financed by BASF and an ERC Consolidator Grant (agreement 819511 – METAFOAM). It also profited from an IdEx Unistra “Attractivity grant” (Chaire W. Drenckhan). Overall, it was conducted in the framework of the Interdisciplinary Institute HiFunMat, as part of the ITI 2021–2028 program of the University of Strasbourg, CNRS and Inserm, and was supported by IdEx Unistra (ANR-10-IDEX-0002) and SFRI (STRATUS project, ANR-20-SFRI-0012) under the framework of the French Investments for the Future Program.
Author information
Authors and Affiliations
Contributions
MH and GCC conducted and analyzed the experiments. All authors designed the study. MH and WD wrote the manuscript. All authors reviewed the manuscript. WD, MR, and MS managed the project and acquired the funding.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hamann, M., Cotte-Carluer, G., Andrieux, S. et al. Fluorocarbon-driven pore size reduction in polyurethane foams: an effect of improved bubble entrainment. Colloid Polym Sci 302, 585–596 (2024). https://doi.org/10.1007/s00396-023-05208-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-023-05208-9