Abstract
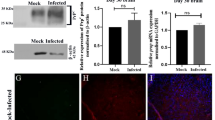
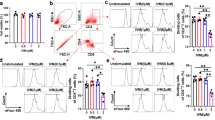
Adiponectin (APN) is an immunomodulatory adipocytokine that improves outcome in patients with virus-negative inflammatory cardiomyopathy and mice with autoimmune myocarditis. Here, we investigated whether APN modulates cardiac inflammation and injury in coxsackievirus B3 (CVB3) myocarditis. Myocarditis was induced by CVB3 infection of APN-KO and WT mice. APN reconstitution was performed by adenoviral gene transfer. Expression analyses were performed by qRT-PCR and immunoblot. Cardiac histology was analyzed by H&E-stain and immunohistochemistry. APN-KO mice exhibited diminished subacute myocarditis with reduced viral load, attenuated inflammatory infiltrates determined by NKp46, F4/80 and CD3/CD4/CD8 expression and reduced IFNβ, IFNγ, TNFα, IL-1β and IL-12 levels. Moreover, myocardial injury assessed by necrotic lesions and troponin I release was attenuated resulting in preserved left ventricular function. Those changes were reversed by APN reconstitution. APN had no influence on adhesion, uptake or replication of CVB3 in cardiac myocytes. In acute CVB3 myocarditis, cardiac viral load did not differ between APN-KO and WT mice. However, APN-KO mice displayed an enhanced acute immune response, i.e. increased expression of myocardial CD14, IFNβ, IFNγ, IL-12, and TNFα resulting in increased cardiac infiltration with pro-inflammatory M1 macrophages and activated NK cells. Up-regulation of cardiac CD14 expression, type I and II IFNs and inflammatory cell accumulation in APN-KO mice was inhibited by APN reconstitution. Our observations indicate that APN promotes CVB3 myocarditis by suppression of toll-like receptor-dependent innate immune responses, polarization of anti-inflammatory M2 macrophages and reduction of number and activation of NK cells resulting in attenuated acute anti-viral immune responses.






Similar content being viewed by others
References
Badorff C, Lee GH, Lamphear BJ, Martone ME, Campbell KP, Rhoads RE, Knowlton KU (1999) Enteroviral protease 2A cleaves dystrophin: evidence of cytoskeletal disruption in an acquired cardiomyopathy. Nat Med 5:320–326. doi:10.1038/6543
Blyszczuk P, Kania G, Dieterle T, Marty RR, Valaperti A, Berthonneche C, Pedrazzini T, Berger CT, Dirnhofer S, Matter CM, Penninger JM, Luscher TF, Eriksson U (2009) Myeloid differentiation factor-88/interleukin-1 signaling controls cardiac fibrosis and heart failure progression in inflammatory dilated cardiomyopathy. Circ Res 105:912–920. doi:10.1161/CIRCRESAHA.109.199802
Bobbert P, Scheibenbogen C, Jenke A, Kania G, Wilk S, Krohn S, Stehr J, Kuehl U, Rauch U, Eriksson U, Schultheiss HP, Poller W, Skurk C (2011) Adiponectin expression in patients with inflammatory cardiomyopathy indicates favourable outcome and inflammation control. Eur Heart J 32:1134–1147. doi:10.1093/eurheartj/ehq498
Chiang CH, Lai JS, Hung SH, Lee LT, Sheu JC, Huang KC (2013) Serum adiponectin levels are associated with hepatitis B viral load in overweight to obese hepatitis B virus carriers. Obesity 21:291–296. doi:10.1002/oby.20000
Chow LH, Beisel KW, McManus BM (1992) Enteroviral infection of mice with severe combined immunodeficiency. Evidence for direct viral pathogenesis of myocardial injury. Lab Invest 66:24–31
Cooper LT Jr (2009) Myocarditis. N Eng J Med 360:1526–1538. doi:10.1056/NEJMra0800028
Deonarain R, Cerullo D, Fuse K, Liu PP, Fish EN (2004) Protective role for interferon-beta in coxsackievirus B3 infection. Circulation 110:3540–3543. doi:10.1161/01.CIR.0000136824.73458.20
Esfandiarei M, McManus BM (2008) Molecular biology and pathogenesis of viral myocarditis. Annu Rev Pathol 3:127–155. doi:10.1146/annurev.pathmechdis.3.121806.151534
Fairweather D, Frisancho-Kiss S, Yusung SA, Barrett MA, Davis SE, Steele RA, Gatewood SJ, Rose NR (2005) IL-12 protects against coxsackievirus B3-induced myocarditis by increasing IFN-gamma and macrophage and neutrophil populations in the heart. J Immunol 174:261–269
Finberg RW, Kurt-Jones EA (2006) CD14: chaperone or matchmaker? Immunity 24:127–129. doi:10.1016/j.immuni.2006.01.010
Fujiu K, Nagai R (2013) Contributions of cardiomyocyte–cardiac fibroblast–immune cell interactions in heart failure development. Basic Res Cardiol 108:1–15. doi:10.1007/s00395-013-0357-x
Gebhard JR, Perry CM, Harkins S, Lane T, Mena I, Asensio VC, Campbell IL, Whitton JL (1998) Coxsackievirus B3-induced myocarditis: perforin exacerbates disease, but plays no detectable role in virus clearance. Am J Pathol 153:417–428. doi:10.1016/S0002-9440(10)65585-X
Godeny EK, Gauntt CJ (1986) Involvement of natural killer cells in coxsackievirus B3-induced murine myocarditis. J Immunol 137:1695–1702
Gorbea C, Makar KA, Pauschinger M, Pratt G, Bersola JL, Varela J, David RM, Banks L, Huang CH, Li H, Schultheiss HP, Towbin JA, Vallejo JG, Bowles NE (2010) A role for Toll-like receptor 3 variants in host susceptibility to enteroviral myocarditis and dilated cardiomyopathy. J Biol Chem 285:23208–23223. doi:10.1074/jbc.M109.047464
Hofmann U, Frantz S (2013) How can we cure a heart “in flame”? A translational view on inflammation in heart failure. Basic Res Cardiol 108:1–19. doi:10.1007/s00395-013-0356-y
Jenke A, Wilk S, Poller W, Eriksson U, Valaperti A, Rauch BH, Stroux A, Liu P, Schultheiss HP, Scheibenbogen C, Skurk C (2013) Adiponectin protects against Toll-like receptor 4 mediated cardiac inflammation and injury. Cardiovasc Res 99:422–431. doi:10.1093/cvr/cvt118
Kanda T, Saegusa S, Takahashi T, Sumino H, Morimoto S, Nakahashi T, Iwai K, Matsumoto M (2007) Reduced-energy diet improves survival of obese KKAy mice with viral myocarditis: induction of cardiac adiponectin expression. Int J Cardiol 119:310–318. doi:10.1016/j.ijcard.2006.07.181
Kandolf R, Sauter M, Aepinus C, Schnorr JJ, Selinka HC, Klingel K (1999) Mechanisms and consequences of enterovirus persistence in cardiac myocytes and cells of the immune system. Virus Res 62:149–158. doi:10.1016/S0168-1702(99)00041-6
Kania G, Blyszczuk P, Eriksson U (2009) Mechanisms of cardiac fibrosis in inflammatory heart disease. Trends Cardiovas Med 19:247–252. doi:10.1016/j.tcm.2010.02.005
Kindermann I, Kindermann M, Kandolf R, Klingel K, Bultmann B, Muller T, Lindinger A, Bohm M (2008) Predictors of outcome in patients with suspected myocarditis. Circulation 118:639–648. doi:10.1161/CIRCULATIONAHA.108.769489
Klingel K, Hohenadl C, Canu A, Albrecht M, Seemann M, Mall G, Kandolf R (1992) Ongoing enterovirus-induced myocarditis is associated with persistent heart muscle infection: quantitative analysis of virus replication, tissue damage, and inflammation. PNAS 89:314–318. doi:10.1073/pnas.89.1.314
Kuhl U, Pauschinger M, Noutsias M, Seeberg B, Bock T, Lassner D, Poller W, Kandolf R, Schultheiss HP (2005) High prevalence of viral genomes and multiple viral infections in the myocardium of adults with “idiopathic” left ventricular dysfunction. Circulation 111:887–893. doi:10.1161/01.CIR.0000155616.07901.35
Kuhl U, Pauschinger M, Schwimmbeck PL, Seeberg B, Lober C, Noutsias M, Poller W, Schultheiss HP (2003) Interferon-beta treatment eliminates cardiotropic viruses and improves left ventricular function in patients with myocardial persistence of viral genomes and left ventricular dysfunction. Circulation 107:2793–2798. doi:10.1161/01.CIR.0000072766.67150.51
Lamphear BJ, Yan R, Yang F, Waters D, Liebig HD, Klump H, Kuechler E, Skern T, Rhoads RE (1993) Map** the cleavage site in protein synthesis initiation factor eIF-4 gamma of the 2A proteases from human Coxsackievirus and rhinovirus. J Biol Chem 268:19200–19203
Meznarich J, Malchodi L, Helterline D, Ramsey SA, Bertko K, Plummer T, Plawman A, Gold E, Stempien-Otero A (2013) Urokinase plasminogen activator induces pro-fibrotic/m2 phenotype in murine cardiac macrophages. PLoS One 8:e57837. doi:10.1371/journal.pone.0057837
Negishi H, Osawa T, Ogami K, Ouyang X, Sakaguchi S, Koshiba R, Yanai H, Seko Y, Shitara H, Bishop K, Yonekawa H, Tamura T, Kaisho T, Taya C, Taniguchi T, Honda K (2008) A critical link between Toll-like receptor 3 and type II interferon signaling pathways in antiviral innate immunity. PNAS 105:20446–20451. doi:10.1073/pnas.0810372105
Ohashi K, Parker JL, Ouchi N, Higuchi A, Vita JA, Gokce N, Pedersen AA, Kalthoff C, Tullin S, Sams A, Summer R, Walsh K (2010) Adiponectin promotes macrophage polarization toward an anti-inflammatory phenotype. J Biol Chem 285:6153–6160. doi:10.1074/jbc.M109.088708
Ouchi N, Kihara S, Arita Y, Okamoto Y, Maeda K, Kuriyama H, Hotta K, Nishida M, Takahashi M, Muraguchi M, Ohmoto Y, Nakamura T, Yamashita S, Funahashi T, Matsuzawa Y (2000) Adiponectin, an adipocyte-derived plasma protein, inhibits endothelial NF-kappaB signaling through a cAMP-dependent pathway. Circulation 102:1296–1301
Pei H, Qu Y, Lu X, Yu Q, Lian K, Liu P, Yan W, Liu J, Ma Y, Liu Y, Li C, Li W, Lau W, Zhang H, Tao L (2012) Cardiac-derived adiponectin induced by long-term insulin treatment ameliorates myocardial ischemia/reperfusion injury in type 1 diabetic mice via AMPK signaling. Basic Res Cardiol 108:1–11. doi:10.1007/s00395-012-0322-0
Porta C, Rimoldi M, Raes G, Brys L, Ghezzi P, Di Liberto D, Dieli F, Ghisletti S, Natoli G, De Baetselier P, Mantovani A, Sica A (2009) Tolerance and M2 (alternative) macrophage polarization are related processes orchestrated by p50 nuclear factor kappaB. PNAS 106:14978–14983. doi:10.1073/pnas.0809784106
Riad A, Westermann D, Zietsch C, Savvatis K, Becher PM, Bereswill S, Heimesaat MM, Lettau O, Lassner D, Dorner A, Poller W, Busch M, Felix SB, Schultheiss HP, Tschope C (2011) TRIF is a critical survival factor in viral cardiomyopathy. J Immunol 186:2561–2570. doi:10.4049/jimmunol.1002029
Shibata R, Izumiya Y, Sato K, Papanicolaou K, Kihara S, Colucci WS, Sam F, Ouchi N, Walsh K (2007) Adiponectin protects against the development of systolic dysfunction following myocardial infarction. J Mol Cell Cardiol 42:1065–1074. doi:10.1016/j.yjmcc.2007.03.808
Shibata R, Sato K, Pimentel DR, Takemura Y, Kihara S, Ohashi K, Funahashi T, Ouchi N, Walsh K (2005) Adiponectin protects against myocardial ischemia-reperfusion injury through AMPK- and COX-2-dependent mechanisms. Nat Med 11:1096–1103. doi:10.1038/nm1295
Takahashi T, Saegusa S, Sumino H, Nakahashi T, Iwai K, Morimoto S, Kanda T (2005) Adiponectin replacement therapy attenuates myocardial damage in leptin-deficient mice with viral myocarditis. Journal Int Med Res 33:207–214. doi:10.1177/147323000503300208
Takahashi T, Yu F, Saegusa S, Sumino H, Nakahashi T, Iwai K, Morimoto S, Kurabayashi M, Kanda T (2006) Impaired expression of cardiac adiponectin in leptin-deficient mice with viral myocarditis. Int Heart J 47:107–123. doi:10.1536/ihj.47.107
Tilg H, Moschen AR (2006) Adipocytokines: mediators linking adipose tissue, inflammation and immunity. Nat Rev Immunol 6:772–783. doi:10.1038/nri1937
Trapani JA, Smyth MJ (2002) Functional significance of the perforin/granzyme cell death pathway. Nat Rev Immunol 2:735–747. doi:10.1038/nri911
Wada H, Saito K, Kanda T, Kobayashi I, Fujii H, Fujigaki S, Maekawa N, Takatsu H, Fujiwara H, Sekikawa K, Seishima M (2001) Tumor necrosis factor-alpha (TNF-alpha) plays a protective role in acute viralmyocarditis in mice: a study using mice lacking TNF-alpha. Circulation 103:743–749
Wessely R, Klingel K, Knowlton KU, Kandolf R (2001) Cardioselective infection with coxsackievirus B3 requires intact type I interferon signaling: implications for mortality and early viral replication. Circulation 103:756–761
Wilk S, Jenke A, Stehr J, Yang CA, Bauer S, Goldner K, Kotsch K, Volk HD, Poller W, Schultheiss HP, Skurk C, Scheibenbogen C (2013) Adiponectin modulates natural killer cell function. Eur J Immunol 43:1024–1033. doi:10.1002/eji.201242382
Wilk S, Scheibenbogen C, Bauer S, Jenke A, Rother M, Guerreiro M, Kudernatsch R, Goerner N, Poller W, Elligsen-Merkel D, Utku N, Magrane J, Volk HD, Skurk C (2011) Adiponectin is a negative regulator of antigen-activated T cells. Eur J Immunol 41:2323–2332. doi:10.1002/eji.201041349
Woodruff JF, Woodruff JJ (1974) Involvement of T lymphocytes in the pathogenesis of coxsackie virus B3 heart disease. J Immunol 113:1726–1734
**ong D, Lee GH, Badorff C, Dorner A, Lee S, Wolf P, Knowlton KU (2002) Dystrophin deficiency markedly increases enterovirus-induced cardiomyopathy: a genetic predisposition to viral heart disease. Nat Med 8:872–877. doi:10.1038/nm737
Yajima T, Knowlton KU (2009) Viral myocarditis: from the perspective of the virus. Circulation 119:2615–2624. doi:10.1161/CIRCULATIONAHA.108.766022
Yan W, Zhang H, Liu P, Wang H, Liu J, Gao C, Liu Y, Lian K, Yang L, Sun L, Guo Y, Zhang L, Dong L, Lau W, Gao E, Gao F, **ong L, Wang H, Qu Y, Tao L (2013) Impaired mitochondrial biogenesis due to dysfunctional adiponectin-AMPK-PGC-1α signaling contributing to increased vulnerability in diabetic heart. Basic Res Cardiol 108:1–15. doi:10.1007/s00395-013-0329-1
Yoon S, Jung J, Kim T, Park S, Chwae YJ, Shin HJ, Kim K (2011) Adiponectin, a downstream target gene of peroxisome proliferator-activated receptor gamma, controls hepatitis B virus replication. Virology 409:290–298. doi:10.1016/j.virol.2010.10.024
Zacharioudaki V, Androulidaki A, Arranz A, Vrentzos G, Margioris AN, Tsatsanis C (2009) Adiponectin promotes endotoxin tolerance in macrophages by inducing IRAK-M expression. J Immunol 182:6444–6451. doi:10.4049/jimmunol.0803694
Acknowledgments
This work was supported by a Grant of Deutsche Forschungsgemeinschaft (SFB TR19, TP B7 to CS and CS as well as TP Z4 to KK).
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jenke, A., Holzhauser, L., Löbel, M. et al. Adiponectin promotes coxsackievirus B3 myocarditis by suppression of acute anti-viral immune responses. Basic Res Cardiol 109, 408 (2014). https://doi.org/10.1007/s00395-014-0408-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-014-0408-y