Abstract
Purpose
Notch and Wnt/β-catenin signaling are responsible for regulation of intestinal stem cells (ISCs) proliferation and differentiation. The purpose of the study was to evaluate Wnt/β-catenin and Notch signaling roles in regulation of ISC differentiation following ischemia–reperfusion (IR) injury in a rat.
Methods
Rats were assigned into two groups: Sham rats underwent laparotomy without vascular intervention and IR rats underwent occlusion of SMA and portal vein for 20 min followed by 48 h of reperfusion. Wnt/β-catenin and Notch-related gene expression were determined using Real-Time PCR. Enterocyte proliferation, differentiation and Wnt-related proteins were determined by immunohistochemistry.
Results
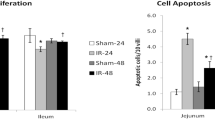
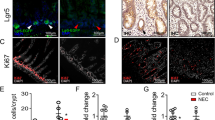
IR rats demonstrated a significant decrease in β-catenin gene expression, a decrease in cyclin D1 and β-catenin positive cells in jejunum and ileum compared to Sham rats. IR rats demonstrated a significant increase in Notch-related gene expression in jejunum and ileum compared to Sham rats. The number of secretory cells was higher mainly in the jejunum and number of absorptive cells was significantly lower in jejunum and lower in ileum in IR rats compared to Sham rats.
Conclusions
Intestinal stem-cell differentiation is toward secretory cells 48 h after IR injury; however, Wnt/β-catenin pathway inhibition and Notch-related gene expression stimulation suggest crosstalk between pathways.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Carden DL, Granger DN (2000) Pathophysiology of ischemia–reperfusion injury. J Pathol 190:255–266. https://doi.org/10.1002/(SICI)1096-9896(200002)190:3%3c255::AID-PATH526%3e3.0.CO;2-6
Mangino JE, Kotadia B, Mangino MJ (1996) Characterization of hypothermic intestinal ischemia–reperfusion injury in dogs. Effects Glycine Transpl 62:173–178. https://doi.org/10.1097/00007890-199607270-00005
Schoenberg MH, Poch B, Younes M et al (1991) Involvement of neutrophils in postischemic damage to the small intestine. Gut 32:905–912. https://doi.org/10.1136/gut.32.8.905
Yamamoto S, Tanabe M, Wakabayashi G et al (2001) The role of tumor necrosis factor-alpha and interleukin-1beta in ischemia–reperfusion injury of the rat small intestine. J Surg Res 99:134–141. https://doi.org/10.1006/jsre.2001.6106
Ikeda H, Suzuki Y, Suzuki M et al (1998) Apoptosis is a major mode of cell death caused by ischemia and ischemia/reperfusion injury to the rat intestinal epithelium. Gut 42:530–537. https://doi.org/10.1136/gut.42.4.530
Noda T, Iwakiri R, Fujimoto K et al (1998) Programmed cell death induced by ischemia–reperfusion in rat intestinal mucosa. Am J PhysiolGastrointest Liver Physiol 274:G270–G276. https://doi.org/10.1152/ajpgi.1998.274.2.G270
Ben-Shahar Y, Pollak Y, Bitterman A et al (2019) Sonic hedgehog signaling controls gut epithelium homeostasis following intestinal ischemia–reperfusion in a rat. Pediatr Surg Int 35:255–261. https://doi.org/10.1007/s00383-018-4406-2. (Epub 2018 Nov 1)
De Santa BP, van den Brink GR, Roberts DJ (2003) Development and differentiation of the intestinal epithelium. Cell Mol Life Sci 60:1322–1323. https://doi.org/10.1007/s00018-003-2289-3
Cheng H, Leblond CP (1974) Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine. Unitarian theory of the origin of the four epithelial cell types. Am J Anat 141:537–561. https://doi.org/10.1002/aja.1001410407
Vander Flier LG, Clevers H (2009) Stem cells, self-renewal, and differentiation in the intestinal epithelium. Ann Rev Physiol 71:241–260. https://doi.org/10.1146/annurev.physiol.010908.163145
Katoh M (2007) WNT signaling pathway and stem cell signaling network. Clin Cancer Res 13:4042–4045. https://doi.org/10.1158/1078-0432.CCR-06-2316
Turashvili G, Bouchal J, Burkadze G, Kolar Z (2006) Wnt signaling pathway in mammary gland development and carcinogenesis. Pathobiology 73:213–223. https://doi.org/10.1159/000098207
van de Wetering M, Sancho E, Verweij C, de Lau W, Oving I, Hurlstone A, van der Horn K, Batlle E, Coudreuse D, Haramis AP, Tjon-Pon-Fong M, Moerer P, van den Born M, Soete G, Pals S, Eilers M, Medema R, Clevers H (2002) The beta-catenin/TCF4 complex imposes a crypt progenitor phenotype on colorectal cancer cells. Cell 18(111):241–250. https://doi.org/10.1016/s0092-8674(02)01014-0
Riccio O, van Gijn ME, Bezdek AC, Pellegrinet L, van Es JH, Zimber-Strobl U, Strobl LJ, Honjo T, Clevers H, Radtke F (2008) Loss of intestinal crypt progenitor cells owing to inactivation of both Notch1 and Notch2 is accompanied by derepression of CDK inhibitors p27 Kip1and p57 Kip2. EMBO Rep 9:377–383. https://doi.org/10.1038/embor.2008.7. (Epub 2008 Feb 15)
Schroder N, Gossler A (2002) Expression of Notch pathway components in fetal and adult mouse small intestine. Gene Expr Patterns 2:247–250. https://doi.org/10.1016/s1567-133x(02)00060-1
Fre S, Huyghe M, Mourikis P, Robine S, Louvard D (2005) Artavanis-Tsakonas S. Notch signals control the fate of immature progenitor cells in the intestine. Nature 435:964–968. https://doi.org/10.1038/nature03589
Myers RB, Fredenburgh JL, Grizzle WE (2008) Carbohydrates. Theory and Practice of Histological Techniques(Bancroft JD, Gamble M, edi-tors). Philadelphia, PA, Churchill Livingstone Elsevier, pp 161–186
Haegebarth A, Clevers H (2009) Wntsignaling, Lgr5, and stem cells in the intestine. Am J Pathol 174:15–21. https://doi.org/10.2353/ajpath.2009.080758
Clevers H (2006) Wnt/beta-catenin signaling in development and disease. Cell 127:469–480. https://doi.org/10.1016/j.cell.2006.10.018
MacDonald BT, Tamai K, He X (2009) Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell 17:9–26. https://doi.org/10.1016/j.devcel.2009.06.016
Eisenmann DM (2005) Wnt signaling. WormBook. https://doi.org/10.1895/wormbook.1.7.1
Kopan R, Ilagan MXG (2009) The canonical Notch signaling pathway: unfolding the activation mechanism. Cell 137:216–233. https://doi.org/10.1016/j.cell.2009.03.045
Bjerknes M, Cheng H (1999) Clonal analysis of mouse intestinal epithelial progenitors. Gastroenterology 116(7–14):9869596. https://doi.org/10.1016/S0016-5085(99)70222-21:STN:280:DyaK1M%2Fosl2mtA%3D%3D
Wong MH, Saam JR, Stappenbeck TS, Rexer CH, Gordon JI (2000) Genetic mosaic analysis based on Cre recombinase and navigated laser capture microdissection. Proc Natl Acad Sci USA 97(12601–12606):11050178. https://doi.org/10.1073/pnas.2302379971:CAS:528:DC%2BD3cXotFygtLo%3D
Marshman E, Booth C, Potten CS (2002) The intestinal epithelial stem cell. BioEssays 24(91–98):11782954. https://doi.org/10.1002/bies.10028
Booth D, Potten CS (2001) Protection against mucosal injury by growth factors and cytokines. J Natl Cancer Inst Monogr 29:16–20. https://doi.org/10.1093/oxfordjournals.jncimonographs.a003433
Podolsky DK (1999) Mucosal immunity and inflammation. V. Innate mechanisms of mucosal defense and repair: the best offense is a good defense. Am J Physiol. https://doi.org/10.1152/ajpgi.1999.277.3.G495
Pellegrinet L, Rodilla V, Liu Z, Chen S, Koch U, Espinosa L, Kaestner KH, Kopan R, Lewis J, Radtke F (2011) Dll1- and Dll4-mediated Notch signaling are required for homeostasis of intestinal stem cells. Gastroenterology 140:1230–1240. https://doi.org/10.1053/j.gastro.2011.01.005
Van Es JH, van Gijn ME, Riccio O, vanden Born M, Vooijs M, (2005) Notch/gamma-secretase inhibition turns proliferativecells in intestinal crypts and adenomas into goblet cells. Nature 435:959–963. https://doi.org/10.1038/nature03659
Kinzler KW, Vogelstein B (1996) Lessons from hereditary colorectal cancer. Cell 87(159–170):8861899. https://doi.org/10.1016/S0092-8674(00)81333-11:CAS:528:DyaK28XmsVGhs7g%3D
Su LK, Kinzler KW, Vogelstein B, Preisinger AC, Moser AR, Luongo C et al (1992) Multiple intestinal neoplasia caused by a mutation in the murine homolog of the APC gene. Science 256:668–670. https://doi.org/10.1126/science.1350108
Sansom OJ, Reed KR, Hayes AJ, Ireland H, Brinkmann H, Newton IP et al (2004) Loss of Apc in vivo immediately perturbs Wnt signaling, differentiation, and migration. Genes Dev 18:1385–1390. https://doi.org/10.1101/gad.287404
Andreu P, Colnot S, Godard C, Gad S, Chafey P, Niwa-Kawakita M et al (2005) Crypt-restricted proliferation and commitment to the Paneth cell lineage following Apc loss in the mouse intestine. Development 132:1443–1451. https://doi.org/10.1242/dev.01700. (Epub 2005 Feb 16)
Korinek V, Barker N, Moerer P, van Donselaar E, Huls G, Peters PJ et al (1998) Depletion of epithelial stem-cell compartments in the small intestine of mice lacking Tcf-4. Nat Genet 19:379–383. https://doi.org/10.1038/1270
Pinto D, Gregorieff A, Begthel H, Clevers H (2003) Canonical Wnt signals are essential for homeostasis of the intestinal epithelium. Genes Dev 17:1709–1713. https://doi.org/10.1101/gad.267103
Ireland H, Kemp R, Houghton C, Howard L, Clarke AR, Sansom OJ et al (2004) Inducible Cre-mediated control of gene expression in the murine gastrointestinal tract: effect of loss of beta-catenin. Gastroenterology 126:1236–1246. https://doi.org/10.1053/j.gastro.2004.03.020
Kuhnert F, Davis CR, Wang HT, Chu P, Lee M, Yuan J et al (2004) Essential requirement for Wnt signaling in proliferation of adult small intestine and colon revealed by adenoviral expression of Dickkopf-1. Proc Natl Acad Sci USA 101:266–271. https://doi.org/10.1073/pnas.2536800100
Artavanis-Tsakonas S, Rand MD, Lake RJ (1999) Notch signaling: cell fate control and signal integration in development. Science 284:770–776. https://doi.org/10.1126/science.284.5415.770
Gridley T (1997) Notch signaling in vertebrate development and disease. Mol Cell Neurosci 9:103–108. https://doi.org/10.1006/mcne.1997.0610
Nakamura T, Tsuchiya K, Watanabe MJ (2007) Crosstalk between Wnt and Notch signaling in intestinal epithelial cell fate decision. Gastroenterol 42:705–710. https://doi.org/10.1007/s00535-007-2087-z. (Epub 2007 Sep 25)
Jensen J, Pedersen EE, Galante P, Hald J, Heller RS, Ishibashi M et al (2000) Control of endodermal endocrine development by Hes-1. Nat Genet 24:36–44. https://doi.org/10.1038/71657
Sasai Y, Kageyama R, Tagawa Y, Shigemoto R, Nakanishi S (1992) Two mammalian helix-loop-helix factors structurally related to Drosophila hairy and Enhancer of split. Genes Dev 6:2620–2634. https://doi.org/10.1101/gad.6.12b.2620
Jarriault S, Brou C, Logeat F, Schroeter EH, Kopan R, Israel A (1995) Signalling downstream of activated mammalian Notch. Nature 377:355–358. https://doi.org/10.1038/377355a0
Gerbe F, van Es JH, Makrini L, Brulin B, Mellitzer G, Robine S, Romagnolo B, Shroyer NF, Bourgaux JF, Pignodel C, Clevers H, Jay P (2011) Distinct ATOH1 and Neurog3 requirements define tuft cells as a new secretory cell type in the intestinal epithelium. J Cell Biol 192:767–780. https://doi.org/10.1083/jcb.201010127
Ben-Shahar Y, Abassi Z, Pollak Y, Bitterman A, Kreizman-Shefer H, Koppelman T, Fuhrer AE, Hayari L, Sukhotnik I (2019) Accelerated cell turnover 48 h after intestinal ischemia is NOTCH independent. Pediatr Surg Int 35:1413–1420. https://doi.org/10.1007/s00383-019-04569-z. (Epub 2019 Oct 1PMID: 31576469)
Leow CC, Romero MS, Ross S, Polakis P, Gao WQ (2004) Hath1, down-regulated in colon adenocarcinomas, inhibits proliferation and tumorigenesis of colon cancer cells. Cancer Res 64:6050–6057. https://doi.org/10.1158/0008-5472.CAN-04-0290
Funding
No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Y. B. participated in design, operated the animals, analysed and interpreted the results and wrote the main manuscript text, V. V. and K. K. participated in design and operated the animals, Y. P. analysed and interpreted the results, I. S. participated in design, operated the animals, analysed and interpreted the results and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ben-Shahar, Y., Vasserman, V., Pollak, Y. et al. The mechanism of intestinal stem cells differentiation after ischemia–reperfusion injury in a rat model. Pediatr Surg Int 40, 23 (2024). https://doi.org/10.1007/s00383-023-05610-y
Accepted:
Published:
DOI: https://doi.org/10.1007/s00383-023-05610-y