Abstract
Purpose
To evaluate risk factors of relapse in pediatric patients with clinical stage I (CS1) testicular yolk sac tumors.
Methods
With retrospective analysis, the medical records of children with pure testicular yolk sac tumors who were referred to Sun Yat-sen University Cancer Center and The First Affiliated Hospital from January 1995 to December 2015 were selected and recorded. Histopathology and staging were retrieved and multivariate analysis was performed with SPSS 20.0 software.
Results
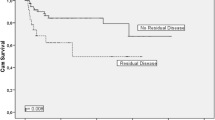
90 children with CS1 testicular yolk sac tumors were selected, and 21 of them underwent chemotherapy following initial orchiectomy. The median age of them was 17 months. With a median follow-up of 61 months (range 11–183 months), 84 patients were alive and 3 patients died, whereas the status was unknown in 3 patients. 30 patients experienced relapse within a median time of 4 months, including only 1 patient who underwent primary chemotherapy, and 28 of these patients underwent salvage chemotherapy. According to adjusted analysis, lymphovascular invasion (LVI) (P < 0.001), necrosis (P = 0.003) and primary chemotherapy (P = 0.008) were independent predictors of event-free survival. The 4-year event-free survival of high- and low-risk patients was 46.5% and 85.1%, respectively (P < 0.001).
Conclusions
LVI and necrosis were independent risk factors for relapse in pediatric patients with CS1 testicular yolk sac tumors, and primary chemotherapy was effective. Thus, individualized management might be feasible for these patients according to risk classification.




Similar content being viewed by others
Data Availability
The authenticity of this article has been validated by uploading the key raw data onto the Research Data Deposit public platform (http://www.researchdata.org.cn), with the approval RDD number as RDDA2017000163.
Abbreviations
- CS1:
-
Clinical stage I
- LVI:
-
Lymphovascular invasion
- RPLND:
-
Retroperitoneal lymph node dissection
- YST:
-
Yolk sac tumor
- RIO:
-
Radical inguinal orchiectomy
- GCT:
-
Germ cell tumor
- NSGCT:
-
Nonseminomatous germ cell tumor
- PEB:
-
Cisplatin, etoposide and bleomycin
- EP:
-
Etoposide and cisplatin
- PVB:
-
Cisplatin, vinblastine and bleomycin
- VIP:
-
Etoposide, ifosfamide and cisplatin
- OS:
-
Overall survival
- EFS:
-
Event-free survival
References
Schlatter M, Rescorla F, Giller R et al (2003) Excellent outcome in patients with stage I germ cell tumors of the testes: a study of the Children’s Cancer Group/Pediatric Oncology Group. J Pediatr Surg 38:319–324 (discussion 319–324)
Pohl HG, Shukla AR, Metcalf PD et al (2004) Prepubertal testis tumors: actual prevalence rate of histological types. J Urol 172:2370–2372
Lee SD (2004) Epidemiological and clinical behavior of prepubertal testicular tumors in Korea. J Urol 172:674–678
Ye YL, Qin ZK, Zhou FJ et al (2008) Clinical analysis of stage I pediatric testicular yolk sac tumors: a report of ten cases. Ai Zheng 27:1226–1228
Cornejo KM, Frazier L, Lee RS, Kozakewich HP, Young RH (2015) Yolk sac tumor of the testis in infants and children: a clinicopathologic analysis of 33 cases. Am J Surg Pathol 39:1121–1131
Ye YL, Sun XZ, Zheng FF et al (2012) Clinical analysis of management of pediatric testicular germ cell tumors. Urology 79:892–897
Grady RW, Ross JH, Kay R (1995) Patterns of metastatic spread in prepubertal yolk sac tumor of the testis. J Urol 153:1259–1261
Rogers PC, Olson TA, Cullen JW et al (2004) Treatment of children and adolescents with stage II testicular and stages I and II ovarian malignant germ cell tumors: a Pediatric Intergroup Study–Pediatric Oncology Group 9048 and Children’s Cancer Group 8891. J Clin Oncol 22:3563–3569
Liu HC, Liang DC, Chen SH et al (1998) The stage I yolk sac tumor of testis in children younger than 2 years, chemotherapy or not? Pediatr Hematol Oncol 15:223–228
Lu SY, Sun XF, Zhen ZJ et al (2015) Survival analysis of children with stage II testicular malignant germ cell tumors treated with surgery or surgery combined with adjuvant chemotherapy. Chin J Cancer 34:86–93
Wei Y, Wu S, Lin T et al (2014) Testicular yolk sac tumors in children: a review of 61 patients over 19 years. World J Surg Oncol 12:400
Abouassaly R, Fossa SD, Giwercman A et al (2011) Sequelae of treatment in long-term survivors of testis cancer. Eur Urol 60:516–526
Travis LB, Fossa SD, Schonfeld SJ et al (2005) Second cancers among 40,576 testicular cancer patients: focus on long-term survivors. J Natl Cancer Inst 97:1354–1365
Brock PR, Knight KR, Freyer DR et al (2012) Platinum-induced ototoxicity in children: a consensus review on mechanisms, predisposition, and protection, including a new International Society of Pediatric Oncology Boston ototoxicity scale. J Clin Oncol 30:2408–2417
Vergouwe Y, Steyerberg EW, Eijkemans MJ, Albers P, Habbema JD (2003) Predictors of occult metastasis in clinical stage I nonseminoma: a systematic review. J Clin Oncol 21:4092–4099
Cost NG, Lubahn JD, Adibi M et al (2014) Risk stratification of pubertal children and postpubertal adolescents with clinical stage I testicular nonseminomatous germ cell tumors. J Urol 191:1485–1490
Rescorla FJ, Ross JH, Billmire DF et al (2015) Surveillance after initial surgery for Stage I pediatric and adolescent boys with malignant testicular germ cell tumors: Report from the Children’s Oncology Group. J Pediatr Surg 50:1000–1003
Ross JH (2009) Prepubertal testicular tumors. Urology 74:94–99
Ahmed HU, Arya M, Muneer A, Mushtaq I, Sebire NJ (2010) Testicular and paratesticular tumours in the prepubertal population. Lancet Oncol 11:476–483
Frazier AL, Hale JP, Rodriguez-Galindo C et al (2015) Revised risk classification for pediatric extracranial germ cell tumors based on 25 years of clinical trial data from the United Kingdom and United States. J Clin Oncol 33:195–201
Rink M, Robinson BD, Green DA et al (2012) Impact of histological variants on clinical outcomes of patients with upper urinary tract urothelial carcinoma. J Urol 188:398–404
Seitz C, Gupta A, Shariat SF et al (2010) Association of tumor necrosis with pathological features and clinical outcome in 754 patients undergoing radical nephroureterectomy for upper tract urothelial carcinoma: an international validation study. J Urol 184:1895–1900
Nichols CR, Roth B, Albers P et al (2013) Active surveillance is the preferred approach to clinical stage I testicular cancer. J Clin Oncol 31:3490–3493
de Wit R, Bosl GJ (2013) Optimal management of clinical stage I testis cancer: one size does not fit all. J Clin Oncol 31:3477–3479
Tandstad T, Stahl O, Hakansson U et al (2014) One course of adjuvant BEP in clinical stage I nonseminoma mature and expanded results from the SWENOTECA group. Ann Oncol 25:2167–2172
Tandstad T, Dahl O, Cohn-Cedermark G et al (2009) Risk-adapted treatment in clinical stage I nonseminomatous germ cell testicular cancer: the SWENOTECA management program. J Clin Oncol 27:2122–2128
Lopes LF, Macedo CR, Aguiar Sdos S et al (2016) Lowered cisplatin dose and no bleomycin in the treatment of pediatric germ cell tumors: results of the GCT-99 protocol from the brazilian germ cell pediatric oncology cooperative group. J Clin Oncol 34:603–610
Grantham EC, Caldwell BT, Cost NG (2016) Current urologic care for testicular germ cell tumors in pediatric and adolescent patients. Urol Oncol 34:65–75
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest was declared.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Ye, Yl., Zheng, Ff., Chen, D. et al. Relapse in children with clinical stage I testicular yolk sac tumors after initial orchiectomy. Pediatr Surg Int 35, 383–389 (2019). https://doi.org/10.1007/s00383-018-04426-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-018-04426-5