Abstract
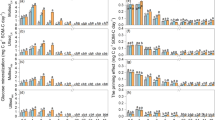
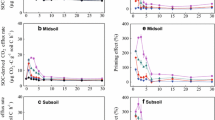
The priming effect (PE) induced by exogenous organic substrate input plays an important role in the soil carbon balance. However, little is known about how the chemical complexity of different exogenous organic substrates and soil type regulate the PE. Therefore, we conducted a 72-day incubation experiment involving the addition of three 13C-labelled substrates (glucose, starch and cellulose) to two forest soils with contrasting textures (i.e., a fine-textured clay soil and a coarse-textured sandy soil). Cellulose had a lag phase in the mineralization process but induced greater PE compared to glucose and starch in both soils. Moreover, exogenous organic substrate input generally induced negative PE and net soil C gain in the clay soil, but positive PE and net soil C loss in the sandy soil. The negative PE in the clay soil was negatively correlated with exogenous C mineralization, indicating that preferential substrate utilization was the mechanism controlling PE in the clay soil. However, the positive PE in the sandy soil was positively correlated with exogenous C mineralization, microbial biomass C (MBC), cellulase activity and soil inorganic N content, suggesting that co-metabolism was the mechanism regulating PE in the sandy soil. The significant difference in PE between the clay and sandy soils was mainly attributed to their significant differences in soil MBC, dissolved organic C (DOC) and SOC contents, followed by soil texture (particularly the clay content), available N content and pH. Overall, our results demonstrate that soil type is much more important than substrate type in regulating the PE and that the clay soil has a greater C storage potential than the sandy soil following the input of exogenous organic substrates.








Similar content being viewed by others
References
Arnosti C, Bell C, Moorhead D, Sinsabaugh R, Steen A, Stromberger M, Wallenstein M, Weintraub M (2014) Extracellular enzymes in terrestrial, freshwater, and marine environments: perspectives on system variability and common research needs. Biogeochem 117:5–21. https://doi.org/10.1007/s10533-013-9906-5
Aye NS, Butterly CR, Sale PWG, Tang C (2017) Residue addition and liming history interactively enhance mineralization of native organic carbon in acid soils. Biol Fert Soils 53:61–75. https://doi.org/10.1007/s00374-016-1156-y
Aye NS, Butterly CR, Sale PW, Tang C (2018) Interactive effects of initial pH and nitrogen status on soil organic carbon priming by glucose and lignocellulose. Soil Biol Biochem 123:33–44. https://doi.org/10.1016/j.soilbio.2018.04.027
Baldock JA, Nelson PN (2000) Soil Organic Matter. Sumner MEEEd. Handbook of Soil Science. CRC Press, Boca Raton, FL, pp 25–84
Bastida F, García C, Fierer N, Eldridge DJ, Bowker MA, Abades S, Alfaro FD, Berhe AA, Cutler NA, Gallardo A (2019) Global ecological predictors of the soil priming effect. Nat Commun 10:1–9. https://doi.org/10.1038/s41467-019-11472-7
Bei S, Li X, Kuyper TW, Chadwick DR, Zhang J (2022) Nitrogen availability mediates the priming effect of soil organic matter by preferentially altering the straw carbon-assimilating microbial community. Sci Total Environ 815:152882. https://doi.org/10.1016/j.scitotenv.2021.152882
Bernard L, Basile-Doelsch I, Derrien D, Fanin N, Fontaine S, Guenet B, Karimi B, Marsden C, Maron PA (2022) Advancing the mechanistic understanding of the priming effect on soil organic matter mineralisation. Funct Ecol 36:1355–1377. https://doi.org/10.1111/1365-2435.14038
Bingeman CW, Varner J, Martin W (1953) The effect of the addition of organic materials on the decomposition of an organic soil. Soil Sci Soc Am J 17:34–38. https://doi.org/10.2136/sssaj1953.03615995001700010008x
Blagodatskaya E, Kuzyakov Y (2008) Mechanisms of real and apparent priming effects and their dependence on soil microbial biomass and community structure: critical review. Biol Fert Soils 45:115–131. https://doi.org/10.1007/s00374-008-0334-y
Blagodatskaya EV, Blagodatsky SA, Anderson TH, Kuzyakov Y (2007) Priming effects in Chernozem induced by glucose and N in relation to microbial growth strategies. Appl Soil Ecol 37:95–105. https://doi.org/10.1016/j.apsoil.2007.05.002
Blagodatskaya E, Yuyukina T, Blagodatsky S, Kuzyakov Y (2011) Turnover of soil organic matter and of microbial biomass under C3–C4 vegetation change: consideration of 13C fractionation and preferential substrate utilization. Soil Biol Biochem 43:159–166. https://doi.org/10.1016/j.soilbio.2010.09.028
Chen R, Senbayram M, Blagodatsky S, Myachina O, Dittert K, Lin X, Blagodatskaya E, Kuzyakov Y (2014) Soil C and N availability determine the priming effect: microbial N mining and stoichiometric decomposition theories. Global Change Biol 20:2356–2367. https://doi.org/10.1111/gcb.12475
Chen L, Liu L, Qin S, Yang G, Fang K, Zhu B, Kuzyakov Y, Chen P, Xu Y, Yang Y (2019) Regulation of priming effect by soil organic matter stability over a broad geographic scale. Nat Commun 10:1–10. https://doi.org/10.1038/s41467-019-13119-z
Chen Y, Li W, You Y, Ye C, Shu X, Zhang Q, Zhang K (2022) Soil properties and substrate quality determine the priming of soil organic carbon during vegetation succession. Plant Soil 471:559–575. https://doi.org/10.1007/s11104-021-05241-z
Chotte JL, Ladd JN, Amato M (1998) Sites of microbial assimilation, and turnover of soluble and particulate 14C-labelled substrates decomposing in a clay soil. Soil Biol Biochem 30:205–218. https://doi.org/10.1016/S0038-0717(97)00115-6
Cotrufo MF, Wallenstein MD, Boot CM, Denef K, Paul E (2013) The Microbial Efficiency-Matrix S tabilization (MEMS) framework integrates plant litter decomposition with soil organic matter stabilization: do labile plant inputs form stable soil organic matter? Global Change Biol 19:988–995. https://doi.org/10.1111/gcb.12113
Craine JM, Morrow C, Fierer N (2007) Microbial nitrogen limitation increases decomposition. Ecology 88:2105–2113. https://doi.org/10.1890/06-1847.1
Creamer CA, Jones DL, Baldock JA, Rui Y, Murphy DV, Hoyle FC, Farrell M (2016) Is the fate of glucose-derived carbon more strongly driven by nutrient availability, soil texture, or microbial biomass size? Soil Biol Biochem 103:201–212. https://doi.org/10.1016/j.soilbio.2016.08.025
Dalenberg J, Jager G (1981) Priming effect of small glucose additions to 14C-labelled soil. Soil Biol Biochem 13:219–223. https://doi.org/10.1016/0038-0717(81)90024-9
Dalenberg J, Jager G (1989) Priming effect of some organic additions to 14C-labelled soil. Soil Biol Biochem 21:443–448. https://doi.org/10.1016/0038-0717(89)90157-0
Davidson EA, Janssens IA (2006) Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature 440:165–173. https://doi.org/10.1038/nature04514
Deng S, Zheng X, Chen X, Zheng S, He X, Ge T, Kuzyakov Y, Wu J, Su Y, Hu Y (2021) Divergent mineralization of hydrophilic and hydrophobic organic substrates and their priming effect in soils depending on their preferential utilization by bacteria and fungi. Biol Fert Soils 57:65–76. https://doi.org/10.1007/s00374-020-01503-7
Di Lonardo DP, De Boer W, Klein Gunnewiek PJA, Hannula SE, Van der Wal A (2017) Priming of soil organic matter: Chemical structure of added compounds is more important than the energy content. Soil Biol and Biochem 108:41–54. https://doi.org/10.1016/j.soilbio.2017.01.017
Fang Y, Nazaries L, Singh BK, Singh BP (2018) Microbial mechanisms of carbon priming effects revealed during the interaction of crop residue and nutrient inputs in contrasting soils. Global Change Biol 24:2775–2790. https://doi.org/10.1111/gcb.14154
Feng J, Tang M, Zhu B (2021) Soil priming effect and its responses to nutrient addition along a tropical forest elevation gradient. Global Change Biol 27:2793–2806. https://doi.org/10.1111/gcb.15587
Fontaine S, Mariotti A, Abbadie L (2003) The priming effect of organic matter: a question of microbial competition? Soil Biol Biochem 35:837–843. https://doi.org/10.1016/S0038-0717(03)00123-8
Frey SD, Lee J, Melillo JM, Six J (2013) The temperature response of soil microbial efficiency and its feedback to climate. Nat Clim Change 3:395–398. https://doi.org/10.1038/nclimate1796
Frøseth RB, Bleken MA (2015) Effect of low temperature and soil type on the decomposition rate of soil organic carbon and clover leaves, and related priming effect. Soil Biol Biochem 80:156–166. https://doi.org/10.1016/j.soilbio.2014.10.004
Garcia-Pausas J, Paterson E (2011) Microbial community abundance and structure are determinants of soil organic matter mineralisation in the presence of labile carbon. Soil Biol Biochem 43:1705–1713. https://doi.org/10.1016/j.soilbio.2011.04.016
Gaudel G, Poudel M, Mosongo PS, **ng L, Oljira AM, Zhang Y, Bizimana F, Liu B, Wang Y, Dong W (2021) Meta-analysis of the priming effect on native soil organic carbon in response to glucose amendment across soil depths (online). Plant Soil. https://doi.org/10.1007/s11104-021-05168-5
Guenet B, Camino-Serrano M, Ciais P, Tifafi M, Maignan F, Soong JL, Janssens IA (2018) Impact of priming on global soil carbon stocks. Global Change Biol 24:1873–1883. https://doi.org/10.1111/gcb.14069
Hamer U, Marschner B (2005) Priming effects in different soil types induced by fructose, alanine, oxalic acid and catechol additions. Soil Biol Biochem 37:445–454. https://doi.org/10.1016/j.soilbio.2004.07.037
Hicks LC, Meir P, Nottingham AT, Reay DS, Stott AW, Salinas N, Whitaker J (2019) Carbon and nitrogen inputs differentially affect priming of soil organic matter in tropical lowland and montane soils. Soil Biol Biochem 129:212–222. https://doi.org/10.1016/j.soilbio.2018.10.015
Jackson O, Quilliam RS, Stott A, Grant H, Subke JA (2019) Rhizosphere carbon supply accelerates soil organic matter decomposition in the presence of fresh organic substrates. Plant Soil 440:473–490. https://doi.org/10.1007/s11104-019-04072-3
Jagadamma S, Mayes MA, Steinweg JM, Schaeffer SM (2014) Substrate quality alters the microbial mineralization of added substrate and soil organic carbon. Biogeosciences 11:4665–4678. https://doi.org/10.5194/bg-11-4665-2014,2014
Joergensen RG (1996) The fumigation-extraction method to estimate soil microbial biomass: calibration of the kEC value. Soil Biol Biochem 28:25–31. https://doi.org/10.1016/0038-0717(95)00102-6
Joergensen RG, Mueller T (1996) The fumigation-extraction method to estimate soil microbial biomass: calibration of the kEN value. Soil Biol Biochem 28:33–37. https://doi.org/10.1016/0038-0717(95)00101-8
Juarez S, Nunan N, Duday AC, Pouteau V, Schmidt S, Hapca S, Falconer R, Otten W, Chenu C (2013) Effects of different soil structures on the decomposition of native and added organic carbon. Eur J Soil Biol 58:81–90. https://doi.org/10.1016/j.ejsobi.2013.06.005
Kuzyakov Y, Bol R (2006) Sources and mechanisms of priming effect induced in two grassland soils amended with slurry and sugar. Soil Biol Biochem 38:747–758. https://doi.org/10.1016/j.soilbio.2005.06.025
Kuzyakov Y, Friedel J, Stahr K (2000) Review of mechanisms and quantification of priming effects. Soil Biol Biochem 32:1485–1498. https://doi.org/10.1016/S0038-0717(00)00084-5
Lenka S, Trivedi P, Singh B, Singh BP, Pendall E, Bass A, Lenka NK (2019) Effect of crop residue addition on soil organic carbon priming as influenced by temperature and soil properties. Geoderma 347:70–79. https://doi.org/10.1016/j.geoderma.2019.03.039
Li JT, Wang JJ, Zeng DH, Zhao SY, Huang WL, Sun XK, Hu YL (2018) The influence of drought intensity on soil respiration during and after multiple drying-rewetting cycles. Soil Biol Biochem 127:82–89. https://doi.org/10.1016/j.soilbio.2018.09.018
Liu L, Estiarte M, Bengtson P, Li J, Asensio D, Wallander H, Peñuelas J (2022) Drought legacies on soil respiration and microbial community in a Mediterranean forest soil under different soil moisture and carbon inputs. Geoderma 405:115425. https://doi.org/10.1016/j.geoderma.2021.115425
Luo Z, Wang E, Sun OJ (2016) A meta-analysis of the temporal dynamics of priming soil carbon decomposition by fresh carbon inputs across ecosystems. Soil Biol Biochem 101:96–103. https://doi.org/10.1016/j.soilbio.2016.07.011
Mizuta K, Taguchi S, Sato S (2015) Soil aggregate formation and stability induced by starch and cellulose. Soil Biol Biochem 87:90–96. https://doi.org/10.1016/j.soilbio.2015.04.011
Perveen N, Barot S, Maire V, Cotrufo MF, Shahzad T, Blagodatskaya E, Stewart CE, Ding W, Siddiq MR, Dimassi B (2019) Universality of priming effect: an analysis using thirty five soils with contrasted properties sampled from five continents. Soil Biol Biochem 134:162–171. https://doi.org/10.1016/j.soilbio.2019.03.027
Qiao N, Schaefer D, Blagodatskaya E, Zou X, Xu X, Kuzyakov Y (2014) Labile carbon retention compensates for CO2 released by priming in forest soils. Global Change Biol 20:1943–1954. https://doi.org/10.1111/gcb.12458
Qiu Q, Wu L, Ouyang Z, Li B, Xu Y, Wu S, Gregorich EG (2016) Priming effect of maize residue and urea N on soil organic matter changes with time. Appl Soil Ecol 100:65–74. https://doi.org/10.1016/j.apsoil.2015.11.016
Rasmussen C, Southard RJ, Horwath WR (2007) Soil mineralogy affects conifer forest soil carbon source utilization and microbial priming. Soil Sci Soc Am J 71:1141–1150. https://doi.org/10.2136/sssaj2006.0375
Shahbaz M, Kuzyakov Y, Sanaullah M, Heitkamp F, Zelenev V, Kumar A, Blagodatskaya E (2017) Microbial decomposition of soil organic matter is mediated by quality and quantity of crop residues: mechanisms and thresholds. Biol Fert Soils 53:287–301. https://doi.org/10.1007/s00374-016-1174-9
Shen J, Bartha R (1996) Priming effect of substrate addition in soil-based biodegradation tests. Appl Environ Microb 62:1428–1430. https://doi.org/10.1128/aem.62.4.1428-1430.1996
Shen J, Bartha R (1997) Priming effect of glucose polymers in soil-based biodegradation tests. Soil Biol Biochem 29:1195–1198. https://doi.org/10.1016/S0038-0717(97)00031-X
Singh M, Sarkar B, Sarkar S, Churchman J, Bolan N, Mandal S, Menon M, Purakayastha TJ, Beerling DJ (2018) Stabilization of soil organic carbon as influenced by clay mineralogy. Adv Agron 148:33–84. https://doi.org/10.1016/bs.agron.2017.11.001
Stevenson FJ (1994) Humus chemistry: genesis, composition, reactions [M]. John Wiley & Sons, New York
Sullivan BW, Hart SC (2013) Evaluation of mechanisms controlling the priming of soil carbon along a substrate age gradient. Soil Biol Biochem 58:293–301. https://doi.org/10.1016/j.soilbio.2012.12.007
Sulman BN, Phillips RP, Oishi AC, Shevliakova E, Pacala SW (2014) Microbe-driven turnover offsets mineral-mediated storage of soil carbon under elevated CO2. Nat Clim Change 4:1099–1102. https://doi.org/10.1038/nclimate2436
Sun Z, Liu S, Zhang T, Zhao X, Chen S, Wang Q (2019) Priming of soil organic carbon decomposition induced by exogenous organic carbon input: a meta-analysis. Plant Soil 443:463–471. https://doi.org/10.1007/s11104-019-04240-5
Tuomela M, Hatakka A, Karjomaa S, Itävaara M (2002) Priming effect as determined by adding 14C-glucose to modified controlled composting test. Biodegradation 13:131–140. https://doi.org/10.1023/A:1020498209463
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring soil microbial biomass C. Soil Biol Biochem 19:703–707. https://doi.org/10.1016/0038-0717(87)90052-6
von Lützow M, Kögel-Knabner I, Ekschmitt K, Flessa H, Guggenberger G, Matzner E, Marschner B (2007) SOM fractionation methods: relevance to functional pools and to stabilization mechanisms. Soil Biol Biochem 39:2183–2207. https://doi.org/10.1016/j.soilbio.2007.03.007
Wang Q, Wang S, He T, Liu L, Wu J (2014) Response of organic carbon mineralization and microbial community to leaf litter and nutrient additions in subtropical forest soils. Soil Biol Biochem 71:13–20. https://doi.org/10.1016/j.soilbio.2014.01.004
Wang H, Boutton TW, Xu W, Hu G, Jiang P, Bai E (2015a) Quality of fresh organic matter affects priming of soil organic matter and substrate utilization patterns of microbes. Sci Rep 5:1–13. https://doi.org/10.1038/srep10102
Wang H, Xu W, Hu G, Dai W, Jiang P, Bai E (2015b) The priming effect of soluble carbon inputs in organic and mineral soils from a temperate forest. Oecologia 178:1239–1250. https://doi.org/10.1007/s00442-015-3290-x
Wang Y, Jiao P, Guo W, Du D, Hu Y, Tan X, Liu X (2022) Changes in bulk and rhizosphere soil microbial diversity and composition along an age gradient of chinese fir (Cunninghamia lanceolate) plantations in subtropical China. Front Microbiol 12:777862. https://doi.org/10.3389/fmicb.2021.777862
Weng ZH, Van Zwieten L, Singh BP, Kimber S, Morris S, Cowie A, Macdonald LM (2015) Plant-biochar interactions drive the negative priming of soil organic carbon in an annual ryegrass field system. Soil Biol Biochem 90:111–121. https://doi.org/10.1016/j.soilbio.2015.08.005
Wu J, Brookes P, Jenkinson D (1993) Formation and destruction of microbial biomass during the decomposition of glucose and ryegrass in soil. Soil Biol Biochem 25:1435–1441. https://doi.org/10.1016/0038-0717(93)90058-J
Wutzler T, Reichstein M (2013) Priming and substrate quality interactions in soil organic matter models. Biogeosciences 10:2089–2103. https://doi.org/10.5194/bg-10-2089-2013,2013
**ao Q, Huang Y, Wu L, Tian Y, Wang Q, Wang B, Xu M, Zhang W (2021) Long-term manuring increases microbial carbon use efficiency and mitigates priming effect via alleviated soil acidification and resource limitation. Biol Fert Soils 57:925–934. https://doi.org/10.1007/s00374-021-01583-z
Xu Y, Liu K, Han Y, Jiang H, Yao S, Zhang B (2019) A soil texture manipulation doubled the priming effect following crop straw addition as estimated by two models. Soil till Res 186:11–22. https://doi.org/10.1016/j.still.2018.09.011
Yang Y, Sun K, Han L, Chen Y, Liu J, **ng B (2022) Biochar stability and impact on soil organic carbon mineralization depend on biochar processing, aging and soil clay content. Soil Biol Biochem 169:108657. https://doi.org/10.1016/j.soilbio.2022.108657
Yuan J, Ouyang Z, Zheng H, Su Y (2021) Ecosystem carbon storage following different approaches to grassland restoration in south-eastern Horqin Sandy Land, northern China. Glob Ecol Conserv 26:e01438. https://doi.org/10.1016/j.gecco.2020.e01438
Zhang L, Li M, Yuan F, Hu Y (2022) Addition effects of organic carbon with different chemical structure on the fate and accumulation of exogenous carbon in red and sandy soils. Chinese J Appl Ecol 33:2593–2601. https://doi.org/10.13287/j.1001-9332.202210.041
Zhao J, Chen S, Hu R, Li Y (2017) Aggregate stability and size distribution of red soils under different land uses integrally regulated by soil organic matter, and iron and aluminum oxides. Soil till Res 167:73–79. https://doi.org/10.1016/j.still.2016.11.007
Funding
This work was supported by the National Natural Science Foundation of China (grant numbers U1805243 and 42077094), the Forestry Peak Discipline Construction Project of Fujian Agriculture and Forestry University (72202200205), and the Science and Technology Innovation Special Fund Project of Fujian Agriculture and Forestry University (KFb22029XA).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qiu, Q., Li, M., Mgelwa, A.S. et al. Divergent mineralization of exogenous organic substrates and their priming effects depending on soil types. Biol Fertil Soils 59, 87–101 (2023). https://doi.org/10.1007/s00374-022-01682-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-022-01682-5