Abstract
Purpose
Conditional survival represents the probability of subsequent survival given that patients have already survived a certain length of time. Several models predict biochemical recurrence (BCR) after radical prostatectomy. However, none of them include postoperative prostate-specific antigen (PSA). We aimed to analyze BCR-free survival evolution over time and develop a nomogram incorporating the postoperative PSA value to predict BCR-free survival.
Material and Methods
We included patients treated with robot-assisted radical prostatectomy (RARP) for prostate cancer between 2009 and 2021 and calculated conditional survival. Cox proportional hazard regression analysis was used to assess the predictive variables of BCR. We developed a nomogram predicting BCR-free survival three and five years after RARP. We used c-index and decision curve analyses to compare the nomogram with the Cancer of the Prostate Risk Assessment post-Surgical (CAPRA-S) score.
Results
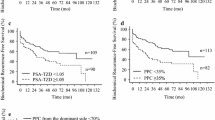
We included 718 patients. The overall 3- and 5-year BCR-free survival rates were 85.1% and 75.7%, respectively. The 5-year BCR-free survival rates increased to 78.9%, 82.9%, 85.2%, and 84.7% for patients surviving 1, 2, 3, and 4 years without BCR, respectively. We developed a nomogram including the pathological Gleason score and T stage, positive surgical margin, PSA ≥ 0.05 ng/mL at one year, and lymph node involvement to predict BCR at 3 and 5 years postoperatively. Our nomogram presented a higher c-index (0.89) than the CAPRA-S score (0.78; p = 0.001) and a positive net benefit at 3 and 5 years postoperatively in the decision curve analyses.
Conclusion
The 5-year conditional BCR-free survival increased with survival without BCR. The developed nomogram significantly improved the accuracy in predicting BCR-free survival after RARP.



Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. https://doi.org/10.3322/caac.21492
Okonogi N, Katoh H, Kawamura H et al (2015) Clinical outcomes of helical tomotherapy for super-elderly patients with localized and locally advanced prostate cancer: comparison with patients under 80 years of age. J Radiat Res 56(6):889–896. https://doi.org/10.1093/jrr/rrv040
He W, Goodkind D, Kowal P. (2015) An aging world. https://www.census.gov/content/dam/Census/library/publications/2016/demo/p95‐16‐1.pdf.
Bill-Axelson A, Holmberg L, Garmo H et al (2014) Radical prostatectomy or watchful waiting in early prostate cancer. N Engl J Med 370(10):932–942. https://doi.org/10.1056/NEJMoa1311593.PMID:24597866;PMCID:PMC4118145
Patel VR, Sivaraman A, Coelho RF et al (2011) Pentafecta: a new concept for reporting outcomes of robot-assisted laparoscopic radical prostatectomy. Eur Urol 59(5):702–707. https://doi.org/10.1016/j.eururo.2011.01.032
Simmons MN, Stephenson AJ, Klein EA (2007) Natural history of biochemical recurrence after radical prostatectomy: risk assessment for secondary therapy. Eur Urol 51(5):1175–1184. https://doi.org/10.1016/j.eururo.2007.01.015
Amling CL, Blute ML, Bergstralh EJ, Seay TM, Slezak J, Zincke H (2000) Long-term hazard of progression after radical prostatectomy for clinically localized prostate cancer: continued risk of biochemical failure after 5 years. J Urol 164(1):101–105. https://doi.org/10.1016/S0022-5347(05)67457-5
Han M, Partin AW, Pound CR, Epstein JI, Walsh PC (2001) Long-term biochemical disease-free and cancer-specific survival following anatomic radical retropubic prostatectomy: the 15-year Johns Hopkins experience. Urol Clin North Am 28(3):555–565. https://doi.org/10.1016/S0094-0143(05)70163-4
Hieke S, Kleber M, König C, Engelhardt M, Schumacher M (2015) Conditional survival: a useful concept to provide information on how prognosis evolves over time. Clin Cancer Res 21(7):1530–1536. https://doi.org/10.1158/1078-0432.CCR-14-2154
Henson DE, Ries LA (1994) On the estimation of survival. Semin Surg Oncol. 10(1):2–6. https://doi.org/10.1002/ssu.2980100103. (PMID: 8115782)
Kawakami M, Hasegawa M, Yamada K et al (2021) Conditional survival and time of biochemical recurrence of localized prostate cancer in japanese patients undergoing laparoscopic radical prostatectomies. Ann Surg Oncol 28(2):1247–1253. https://doi.org/10.1245/s10434-020-08770-6
Park SW, Hwang DS, Song WH, Nam JK, Lee HJ, Chung MK (2020) Conditional biochemical recurrence-free survival after radical prostatectomy in patients with high-risk prostate cancer. Prostate Int 8(4):173–177. https://doi.org/10.1016/j.prnil.2020.07.004
Llukani E, Lepor H (2017) Aetiology and management of earlier vs later biochemical recurrence after retropubic radical prostatectomy. BJU Int 120(4):505–510. https://doi.org/10.1111/bju.13816
García-Barreras S, Sanchez-Salas R, Mejia-Monasterio C et al (2019) Biochemical recurrence-free conditional probability after radical prostatectomy: a dynamic prognosis. Int J Urol 26(7):725–730. https://doi.org/10.1111/iju.13982
**a HZ, Bi H, Yan Y et al (2021) A novel nomogram provides improved accuracy for predicting biochemical recurrence after radical prostatectomy. Chin Med J (Engl) 134(13):1576–1583. https://doi.org/10.1097/CM9.0000000000001607
Koulikov D, Mohler MC, Mehedint DC, Attwood K, Wilding GE, Mohler JL.Low detectable prostate specific antigen after radical prostatectomy--treat or watch?. J Urol. 2014;192(5):1390–6. https://doi.org/10.1016/j.juro.2014.05.088. Epub 2014 May 21. PMID: 24859441; PMCID: PMC4586061.
Murakami T, Otsubo S, Namitome R et al (2009) Clinical factors affecting perioperative outcomes in robot-assisted radical prostatectomy. Mol Clin Oncol 2018:575–581. https://doi.org/10.3892/mco.2018.1718
Cookson MS, Aus G, Burnett AL et al (2007) Variation in the definition of biochemical recurrence in patients treated for localized prostate cancer: The American Urological Association prostate guidelines for localized prostate cancer update panel report and recommendations for a standard in the reporting of surgical outcomes. J Urol 177(2):540–545. https://doi.org/10.1016/j.juro.2006.10.097
Naito S, Kuroiwa K, Kinukawa N et al (2008) Validation of partin tables and development of a preoperative nomogram for Japanese patients with clinically localized prostate cancer using 2005 international society of urological pathology consensus on gleason grading: data from the clinicopathological. J Urol 180(3):904–910. https://doi.org/10.1016/j.juro.2008.05.047
D’Amico AV, Whittington R, Bruce Malkowicz S et al (1998) Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. J Am Med Assoc 280(11):969–974. https://doi.org/10.1001/jama.280.11.969
Chikamatsu S, Shiota M, Onozawa M et al (2021) Dynamics of conditional survival and risk factors in androgen deprivation therapy for prostate cancer using a multi-institutional Japan-wide database. Int J Urol 28(9):927–935. https://doi.org/10.1111/iju.14605
Zlotnik A, Abraira V (2015) A general-purpose nomogram generator for predictive logistic regression models. Stata J 15(2):537–546. https://doi.org/10.1177/1536867x1501500212
Cooperberg MR, Hilton JF, Carroll PR (2011) The CAPRA-S score: a straightforward tool for improved prediction of outcomes after radical prostatectomy. Cancer 117(22):5039–5046. https://doi.org/10.1002/cncr.26169
Ploussard G, de la Taille A, Moulin M, Allorys Y, Abbou C, Salomon L (2016) Conditional disease-free survival after radical prostatectomy: recurrence risk evolution over time. Urology 94:173–179. https://doi.org/10.1016/j.urology.2016.04.031
Sokoll LJ, Zhang Z, Chan DW et al (2016) Do ultrasensitive prostate specific antigen measurements have a role in predicting long-term biochemical recurrence-free survival in men after radical prostatectomy? J Urol 195(2):330–336. https://doi.org/10.1016/j.juro.2015.08.080
Eisenberg ML, Davies BJ, Cooperberg MR, Cowan JE, Carroll PR (2010) Prognostic implications of an undetectable ultrasensitive prostate-specific antigen level after radical prostatectomy. Eur Urol 57(4):622–630. https://doi.org/10.1016/j.eururo.2009.03.077
Mir MC, Li J, Klink JC et al (2014) Optimal definition of biochemical recurrence after radical prostatectomy depends on pathologic risk factors: identifying candidates for early salvage therapy. Eur Urol 66(2):204–210. https://doi.org/10.1016/j.eururo.2013.08.022
Mir MC, Li J, Klink JC, Kattan MW, Klein EA, Stephenson AJ (2014) Optimal definition of biochemical recurrence after radical prostatectomy depends on pathologic risk factors: identifying candidates for early salvage therapy. Eur Urol 66(2):204–210. https://doi.org/10.1016/j.eururo.2013.08.022
Zhao Y, Tao Z, Li L, Zheng J, Chen X (2022) Predicting biochemical-recurrence-free survival using a three-metabolic-gene risk score model in prostate cancer patients. BMC Cancer 22(1):1–11. https://doi.org/10.1186/s12885-022-09331-8
Fredsøe J, Rasmussen AKI, Mouritzen P, Borre M, Ørntoft T, Sørensen KD (2019) A five-microRNA model (pCaP) for predicting prostate cancer aggressiveness using cell-free urine. Int J Cancer 145(9):2558–2567. https://doi.org/10.1002/ijc.32296
Acknowledgements
We thank Ms. Shihoko Nishikawa and Ms. Maki Isoda for their excellent administrative support.
Funding
NonE.
Author information
Authors and Affiliations
Contributions
Conceptualization, LB; MS, writing—original draft preparation, LB; writing—review and editing, MS; evidence acquisition: MS; DT; FK; TM; KL; KM; EK; JI; ME. supervision, ME All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None to declare.
Ethical approval
Approval of the research protocol by the Institutional Review Board Number 2021–121.
Informed consent
N/A
Registry and Registration No. of the study/trial
N/A.
Animal studies
N/A.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Blas, L., Shiota, M., Takamatsu, D. et al. Novel nomogram to predict biochemical recurrence-free survival after radical prostatectomy. World J Urol 41, 43–50 (2023). https://doi.org/10.1007/s00345-022-04245-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-022-04245-3