Abstract
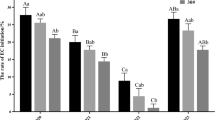
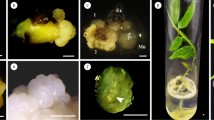
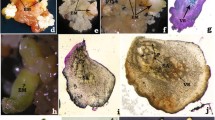
Taxodium hybrid ‘zhongshanshan’ is a valuable bottomland timber tree species with both ecological and economic significance. However, the lack of an efficient propagation method has hindered large-scale reproduction of superior clones. In this study, we aimed to establish an effective propagation system by investigating somatic embryogenesis (SE) of T. hybrid ‘zhongshanshan’. The results demonstrated successful induction of embryogenic calli through treatment of immature embryos with 2 mg L−1 2,4-dichlorophenoxyacetic acid (2,4-D), 0.5 mg L−1 6-Benzylaminopurine (6-BA) and 30 g L−1 maltose. Furthermore, the combination of 190 g L−1 polyethylene glycol (PEG) and 8 mg L−1 abscisic acid (ABA) exhibited a positive impact on somatic embryo induction and maturation. The embryogenic callus of T. hybrid ‘zhongshanshan’ exhibited a distinct embryonal-suspensor mass structure, ultimately develo** into somatic embryos. In addition, we observed that endogenous hormones affected the whole process of somatic embryogenesis. Indoleacetic acid (IAA), Gibberellic acid (GA) and Methyl jasmonate (MeJA) displayed elevated levels during the initial stages and cotyledonary embryos stage, while cytokinin (CTK) and salicylic acid (SA) contents declined throughout SE. The ABA content exhibited a steady increase throughout the SE process. These findings represented a critical technical basis for the efficient propagation of T. hybrid ‘zhongshanshan’, which also provided a good foundation for investigating SE in other conifer species.







Similar content being viewed by others
Abbreviations
- 2,4-D:
-
2,4-Dichlorophenoxyacetic acid
- 6-BA:
-
6-Benzylaminopurine
- CH:
-
Casein hydrolysate
- CTK:
-
Cytokinin
- GA:
-
Gibberellic acid
- IAA:
-
Indoleacetic acid
- MeJA:
-
Methyl jasmonate
- PGRs:
-
Plant growth regulators
- SA:
-
Salicylic acid
- SE:
-
Somatic embryogenesis
- ABA:
-
Abscisic acid
- PSK:
-
Phytosulfokine
- GSH:
-
Glutathione
- GSSG:
-
Glutathione oxidized
References
Belide S, Vanhercke T, Petrie JR, Singh SP (2017) Robust genetic transformation of sorghum (Sorghum bicolor L.) using differentiating embryogenic callus induced from immature embryos. Plant Methods 13:1–12. https://doi.org/10.1186/s13007-017-0260-9
Borjian L, Arak H (2013) A study on the effect of different concentration of plant hormones (BAP, NAA, 2, 4-D, and Kinetin) on callus induction in Brassica napus. International Research Journal of Applied and Basic Sciences 5:519–521
Chen S (2010) Optimization of somatic embryogenesis in Picea likiangensis (Franch.) Pritz and proteomic analysis on somatic embryos at developmental stages. Thesis.
Chen T, Sheng Y, Hao Z, Long X, Fu F, Liu Y, Tang Z, Asif A, Peng Y, Liu Y (2021) Transcriptome and proteome analysis suggest enhanced photosynthesis in tetraploid Liriodendron sino-americanum. Tree Physiol 41:1953–1971. https://doi.org/10.1093/treephys/tpab039
Chen T, Yang D, Fan R, Zheng R, Lu Y, Cheng T, Shi J, Chen J (2022) γ-Aminobutyric acid a novel candidate for rapid induction in somatic embryogenesis of Liriodendron hybrid. Plant Growth Regul 96:293–302. https://doi.org/10.1007/s10725-021-00776-8
Cruz KZCM, Almeida FA, Vale EM, Botini N, Vettorazzi RG, Santos RC, Santa-Catarina C, Silveira V (2022) PEG induces maturation of somatic embryos of Passiflora edulis Sims ‘UENF Rio Dourado’by differential accumulation of proteins and modulation of endogenous contents of free polyamines. Plant Cell Tissue Organ Cult 150:527–541. https://doi.org/10.1007/s11240-022-02301-9
De Almeida NV, Rivas EB, Cardoso JC (2022) Somatic embryogenesis from flower tepals of Hippeastrum aiming regeneration of virus-free plants. Plant Sci 317:111191. https://doi.org/10.1016/j.plantsci.2022.111191
De Freitas Fraga HP, Do Nascimento Vieira L, Puttkammer CC, Dos Santos HP, De Andrade Garighan J, Guerra MP (2016) Glutathione and abscisic acid supplementation influences somatic embryo maturation and hormone endogenous levels during somatic embryogenesis in Podocarpus lambertii Klotzsch ex Endl. Plant Sci 253:98–106. https://doi.org/10.1016/j.plantsci.2016.09.012
Deo PC, Tyagi AP, Taylor M, Harding R, Becker D (2010) Factors affecting somatic embryogenesis and transformation in modern plant breeding. South Pac J Nat Appl Sci 28:27–40. https://doi.org/10.1071/SP10002
Efendi D, Sukma D, Pusparani R (2015) Induction and proliferation of durian (Durio zibethinus) embryonic culture in media supplemented with auxin and cytokinin. In: International symposium on durian and other humid tropical fruits, vol 1186, pp 23–28. https://doi.org/10.17660/ActaHortic.2017.1186.3
Fehér A, Pasternak TP, Dudits D (2003) Transition of somatic plant cells to an embryogenic state. Plant Cell Tissue Organ Cult 74(3):201–228. https://doi.org/10.1023/A:1024033216561
Gaj MD (2004) Factors influencing somatic embryogenesis induction and plant regeneration with particular reference to Arabidopsis thaliana (L.) Heynh. Plant Growth Regul 43:27–47. https://doi.org/10.1023/B:GROW.0000038275.29262.fb
Gao F, Peng C, Wang H, Shen H, Yang L (2021) Selection of culture conditions for callus induction and proliferation by somatic embryogenesis of Pinus koraiensis. J for Res 32:483–491. https://doi.org/10.1007/s11676-020-01147-1
Gautier F, Eliášová K, Leplé J-C, Vondráková Z, Lomenech A-M, Le Metté C, Label P, Costa G, Trontin J-F, Teyssier C (2018) Repetitive somatic embryogenesis induced cytological and proteomic changes in embryogenic lines of Pseudotsuga menziesii [Mirb]. BMC Plant Biol 18:164. https://doi.org/10.1186/s12870-018-1337-y
Grzyb M, Kalandyk A, Waligórski P, Mikuła A (2017) The content of endogenous hormones and sugars in the process of early somatic embryogenesis in the tree fern Cyathea delgadii Sternb. Plant Cell Tissue Organ Cult 129:387–397. https://doi.org/10.1007/s11240-017-1185-8
Guan Y, Li S-G, Fan X-F, Su Z-H (2016) Application of somatic embryogenesis in woody plants. Front Plant Sci 7:938. https://doi.org/10.3389/fpls.2016.00938
Gupta PK, Durzan DJ (1985) Shoot multiplication from mature trees of Douglas-fir (Pseudotsuga menziesii) and sugar pine (Pinus lambertiana). Plant Cell Rep 4(4):177–179. https://doi.org/10.1007/BF00269282
Hao Z, Shi J, Wu H, Yan Y, **ng K, Zheng R, Shi J, Chen J (2023) Phytosulfokine contributes to suspension culture of Cunninghamia lanceolata through its impact on redox homeostasis. BMC Plant Biol 23(1):480. https://doi.org/10.1186/s12870-023-04496-1
Hazubska-Przybył T, Ratajczak E, Obarska A, Pers-Kamczyc E (2020) Different roles of auxins in somatic embryogenesis efficiency in two Picea species. Int J Mol Sci 21:3394. https://doi.org/10.3390/ijms21093394
Horstman A, Bemer M, Boutilier K (2017) A transcriptional view on somatic embryogenesis. Regeneration 4:201–216. https://doi.org/10.1002/reg2.91
Hu R, Sun Y, Wu B, Duan H, Zheng H, Hu D, Lin H, Tong Z, Xu J, Li Y (2017) Somatic embryogenesis of immature Cunninghamia lanceolata (Lamb.) hook zygotic embryos. Sci Rep 7:56. https://doi.org/10.1038/s41598-017-00156-1
Hua J, Han L, Wang Z, Gu C, Yin Y (2017) Morpho-anatomical and photosynthetic responses of Taxodium hybrid “Zhongshanshan” 406 to prolonged flooding. Flora 231:29–37. https://doi.org/10.1016/j.flora.2017.04.007
Igasaki T, Akashi N, U**o-Ihara T, Matsubayashi Y, Sakagami Y, Shinohara K (2003a) Phytosulfokine stimulates somatic embryogenesis in Cryptomeria japonica. Plant Cell Physiol 44:1412–1416. https://doi.org/10.1093/pcp/pcg161
Igasaki T, Sato T, Akashi N, Mohri T, Maruyama E, Kinoshita I, Walter C, Shinohara K (2003b) Somatic embryogenesis and plant regeneration from immature zygotic embryos of Cryptomeria japonica D. Don. Plant Cell Rep 22:239–243. https://doi.org/10.1007/s00299-003-0687-5
Isah T (2016) Induction of somatic embryogenesis in woody plants. Acta Physiol Plant 38:1–22. https://doi.org/10.1007/s11738-016-2134-6
Jiang S, Chen X, Gao Y, Cui Y, Kong L, Zhao J, Zhang J (2021) Plant regeneration via somatic embryogenesis in Larix principis-rupprechtii Mayr. Forests 12:1335. https://doi.org/10.3390/f12101335
Jiménez VM (2001) Regulation of in vitro somatic embryogenesis with emphasis on to the role of endogenous hormones. Rev Bras Fisiol Veg 13:196–223. https://doi.org/10.1590/S0103-31312001000200008
Karami O, Philipsen C, Rahimi A, Nurillah AR, Boutilier K, Offringa R (2022) Endogenous auxin maintains embryonic cell identity and promotes somatic embryo development in Arabidopsis. Plant J 113(1):7–22. https://doi.org/10.1111/tpj.16024
Lelu-Walter MA, Bernier-Cardou M, Klimaszewska K (2006) Simplified and improved somatic embryogenesis for clonal propagation of Pinus pinaster (Ait.). Plant Cell Report 25:767–776. https://doi.org/10.1007/s00299-006-0115-8
Liang Y, Xu X, Shen H, Gao M, Zhao Y, Bai X (2022) Morphological and endogenous phytohormone changes during long-term embryogenic cultures in Korean pine. [Preprint] https://doi.org/10.21203/rs.3.rs-1286747/v1.
Lou Y (2009) Study on the sucrose metabolism in embryogenic callus of occurrence process of Picea mongolica. Thesis.
Loyola-Vargas VM, Ochoa-Alejo N (2016) Somatic embryogenesis. An overview. In: Somatic embryogenesis: fundamental aspects and applications, pp 1–8. https://doi.org/10.1007/978-3-319-33705-0_1.
Maruyama TE, Hosoi Y (2019) Progress in somatic embryogenesis of Japanese pines. Front Plant Sci 10:31. https://doi.org/10.3389/fpls.2019.00031
Maruyama TE, Ueno S, Mori H, Kaneeda T, Moriguchi Y (2021) Factors influencing somatic embryo maturation in Sugi (Japanese cedar, Cryptomeria japonica (Thunb. ex Lf) D. Don). Plants 10:874. https://doi.org/10.3390/plants10050874
Montalbán IA, De Diego N, Moncaleán P (2010) Bottlenecks in Pinus radiata somatic embryogenesis: improving maturation and germination. Trees 24:1061–1071. https://doi.org/10.1007/s00468-010-0477-y
Pourkhaloee A, Khosh-Khui M, Barba-Gonzalez R (2023) Somatic embryogenesis of Tuberose (Agave amica L.) was improved by milk as a potential biostimulant in plant tissue culture. Int J Hortic Sci Technol 10:257–268. https://doi.org/10.22059/ijhst.2022.330660.504
Pullman GS, Zeng X, Copeland-Kamp B, Crockett J, Lucrezi J, May SW, Bucalo K (2015) Conifer somatic embryogenesis: improvements by supplementation of medium with oxidation–reduction agents. Tree Physiol 35:209–224. https://doi.org/10.1093/treephys/tpu117
Raza G, Singh MB, Bhalla PL (2019) Somatic embryogenesis and plant regeneration from commercial soybean cultivars. Plants 9:38. https://doi.org/10.3390/plants9010038
Seldimirova O, Kudoyarova G, Kruglova N, Galin I, Veselov D (2019) Somatic embryogenesis in Wheat and Barley Calli in vitro is determined by the level of indoleacetic and abscisic acids. Russ J Dev Biol 50:124–135. https://doi.org/10.1134/S1062360419030056
Shan H, Chun-xia Y, Zhen-jun G, Qiang D, **-jiang X, Huo-gen L (2022) Optimization of somatic embryogenesis for Pinus taeda. For Res 35:9–17. https://doi.org/10.13275/j.cnki.lykxyj.2022.03.002
Shi Q, Yin Y, Wang Z, Fan W, Hua J (2016) Physiological acclimation of Taxodium hybrid ‘Zhongshanshan 118’ plants to short-term drought stress and recovery. HortScience 51:1159–1166. https://doi.org/10.21273/HORTSCI10997-16
Shi Q, Zhou Z, Wang Z, Lu Z, Han J, Xue J, Creech D, Yin Y, Hua J (2022) Afforestation of Taxodium hybrid zhongshanshan influences soil bacterial community structure by altering soil properties in the Yangtze River Basin. China Plants 11:3456. https://doi.org/10.3390/plants11243456
Shoji M, Sato H, Nakagawa R, Funada R, Kubo T, Ogita S (2006) Influence of osmotic pressure on somatic embryo maturation in Pinus densiflora. J for Res 11:449–453. https://doi.org/10.1007/s10310-006-0227-6
Song Y, Li S, Bai X, Zhang H (2018) Screening and verification of the factors influencing somatic embryo maturation of Larix olgensis. J for Res 29:1581–1589. https://doi.org/10.1007/s11676-018-0694-6
Tret’yakova I, Park M (2018) Somatic polyembriogenesis of Larix sibirica in embryogenic in vitro culture. Russ J Dev Biol 49:222–233. https://doi.org/10.1134/S1062360418040069
Vahdati K, Bayat S, Ebrahimzadeh H, Jariteh M, Mirmasoumi M (2008) Effect of exogenous ABA on somatic embryo maturation and germination in Persian walnut (Juglans regia L.). Plant Cell Tissue Organ Cult 93:163–171. https://doi.org/10.1007/s11240-008-9355-3
Vondráková Z, Eliášová K, Fischerová L, Vágner M (2011) The role of auxins in somatic embryogenesis of Abies alba. Cent Eur J Biol 6:587–596. https://doi.org/10.2478/s11535-011-0035-7
Vondrakova Z, Dobrev PI, Pesek B, Fischerova L, Vagner M, Motyka V (2018) Profiles of endogenous phytohormones over the course of Norway spruce somatic embryogenesis. Front Plant Sci 9:1283. https://doi.org/10.3389/fpls.2018.01283
Wang X, Peng D (2005) Effects of three kinds of cytokinins on callus induction and differentiation of ramie. Hubei Agric Sci 1:17–19
Wang Z, Hua J, Yin Y, Gu C, Yu C, Shi Q, Guo J, Xuan L, Yu F (2019) An integrated transcriptome and proteome analysis reveals putative regulators of adventitious root formation in Taxodium ‘Zhongshanshan.’ Int J Mol Sci 20:1225. https://doi.org/10.3390/ijms20051225
Wang D, Guo Y, Long X, Pan Y, Yang D, Li R, Lu Y, Chen Y, Shi J, Chen J (2020) Exogenous spermidine promotes somatic embryogenesis of Cunninghamia lanceolata by altering the endogenous phytohormone content. Phyton 89:27. https://doi.org/10.32604/phyton.2020.08971
Wang Z, Zhang F, Shi Q, Zhang R, Yin Y, Yu C (2022) Identification and characterization of salt-responsive microRNAs in Taxodium hybrid ‘Zhongshanshan 405’ by high-throughput sequencing. Forests 13:1685. https://doi.org/10.3390/f13101685
Wu G, Wei X, Wang X, Wei Y (2021) Changes in biochemistry and histochemical characteristics during somatic embryogenesis in Ormosia henryi Prain. Plant Cell Tissue Organ Cult 144:505–517. https://doi.org/10.1007/s11240-020-01973-5
Xuan L, Hua J, Zhang F, Wang Z, Pei X, Yang Y, Yin Y, Creech DL (2021) Identification and functional analysis of ThADH1 and ThADH4 genes involved in tolerance to waterlogging stress in Taxodium hybrid ‘Zhongshanshan 406.’ Genes 12:225. https://doi.org/10.3390/genes12020225
Ya R, Li J, Zhang N, Yu Q, Xu W (2022) Phenotypically abnormal cotyledonary Vitis vinifera embryos differ in anatomy, endogenous hormone levels and transcriptome profiles. Tree Physiol 43(3):467–485. https://doi.org/10.1093/treephys/tpac129
Yu C, Xu S, Yin Y (2016) Transcriptome analysis of the Taxodium ‘Zhongshanshan 405’roots in response to salinity stress. Plant Physiol Biochem 100:156–165. https://doi.org/10.1016/j.plaphy.2016.01.009
Zhang S, Yan S, An P, Cao Q, Wang C, Wang J, Zhang H, Zhang L (2021) Embryogenic callus induction from immature zygotic embryos and genetic transformation of Larix kaempferi 3x Larix gmelinii 9. PLoS ONE 16:e0258654. https://doi.org/10.1371/journal.pone.0258654
Zheng W, Zhang X, Yang Z, Wu J, Li F, Duan L, Liu C, Lu L, Zhang C, Li F (2014) AtWuschel promotes formation of the embryogenic callus in Gossypium hirsutum. PLoS ONE 9(1):e87502. https://doi.org/10.1371/journal.pone.0087502
Zhou X, Zheng R, Liu G, Xu Y, Zhou Y, Laux T, Zhen Y, Harding SA, Shi J, Chen J (2017) Desiccation treatment and endogenous IAA levels are key factors influencing high frequency somatic embryogenesis in Cunninghamia lanceolata (Lamb.) Hook. Front Plant Sci 8:2054. https://doi.org/10.3389/fpls.2017.02054
Zhu T, Wang J, Hu J, Ling J (2022) Mini review: Application of the somatic embryogenesis technique in conifer species. For Res. https://doi.org/10.48130/FR-2022-0018
Zuo J, Niu Q, Frugis G, Chua N (2002) The WUSCHEL gene promotes vegetative-to-embryonic transition in Arabidopsis. Plant J 30(3):349–359. https://doi.org/10.1046/j.1365-313X.2002.01289.x
Funding
This work was supported by the National Natural Science Foundation of China (31870592), Jiangsu Institute of Botany Talent Fund (JIBTF202206) and Jiangsu Long-Term Scientific Research Base for Taxodium Rich. Breeding and Cultivation (LYKJ [2021]05).
Author information
Authors and Affiliations
Contributions
HJF contributed conception and design of the study; CTT and JXY performed the experiments; CTT, JXY, ZR, LY, YCG and YYL carried out the statistical analysis; CTT wrote the manuscript. All authors contributed to the manuscript’s revision, read, and approved the submitted version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Handling Editor: Heather Nonhebel.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, T., Jia, X., Zhang, R. et al. Somatic Embryogenesis in Taxodium Hybrid ‘zhongshanshan’, an Opportunity to Propagate an Ecologically Friendly Species. J Plant Growth Regul 43, 1447–1457 (2024). https://doi.org/10.1007/s00344-023-11196-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-023-11196-5