Abstract
Objectives
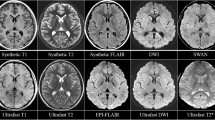
Wave-CAIPI (Controlled Aliasing in Parallel Imaging) enables dramatic reduction in acquisition time of 3D MRI sequences such as 3D susceptibility-weighted imaging (SWI) but has not been clinically evaluated at 1.5 T. We sought to compare highly accelerated Wave-CAIPI SWI (Wave-SWI) with two alternative standard sequences, conventional three-dimensional SWI and two-dimensional T2*-weighted Gradient-Echo (T2*w-GRE), in patients undergoing routine brain MRI at 1.5 T.
Methods
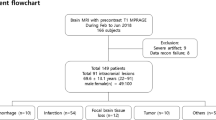
In this study, 172 patients undergoing 1.5 T brain MRI were scanned with a more commonly used susceptibility sequence (standard SWI or T2*w-GRE) and a highly accelerated Wave-SWI sequence. Two radiologists blinded to the acquisition technique scored each sequence for visualization of pathology, motion and signal dropout artifacts, image noise, visualization of normal anatomy (vessels and basal ganglia mineralization), and overall diagnostic quality. Superiority testing was performed to compare Wave-SWI to T2*w-GRE, and non-inferiority testing with 15% margin was performed to compare Wave-SWI to standard SWI.
Results
Wave-SWI performed superior in terms of visualization of pathology, signal dropout artifacts, visualization of normal anatomy, and overall image quality when compared to T2*w-GRE (all p < 0.001). Wave-SWI was non-inferior to standard SWI for visualization of normal anatomy and pathology, signal dropout artifacts, and overall image quality (all p < 0.001). Wave-SWI was superior to standard SWI for motion artifact (p < 0.001), while both conventional susceptibility sequences were superior to Wave-SWI for image noise (p < 0.001).
Conclusions
Wave-SWI can be performed in a 1.5 T clinical setting with robust performance and preservation of diagnostic quality.
Key Points
• Wave-SWI accelerated the acquisition of 3D high-resolution susceptibility images in 70% of the acquisition time of the conventional T2*GRE.
• Wave-SWI performed superior to T2*w-GRE for visualization of pathology, signal dropout artifacts, and overall diagnostic image quality.
• Wave-SWI was noninferior to standard SWI for visualization of normal anatomy and pathology, signal dropout artifacts, and overall diagnostic image quality.




Similar content being viewed by others
Abbreviations
- CAIPI:
-
Controlled Aliasing In Parallel Imaging
- SWI:
-
Susceptibility-Weighted Imaging
- T2*w-GRE:
-
T2*-Weighted Gradient-Echo
- Wave-CAIPI:
-
Wave-Controlled Aliasing In Parallel Imaging
References
Haacke EM, Mittal S, Wu Z et al (2009) Susceptibility-weighted imaging: technical aspects and clinical applications, part 1. AJNR Am J Neuroradiol 30:19–30
Mittal S, Wu Z, Neelavalli J, Haacke EM (2009) Susceptibility-weighted imaging: technical aspects and clinical applications, part 2. AJNR Am J Neuroradiol 30:232–252
Tong KA, Ashwal S, Holshouser BA et al (2003) Hemorrhagic shearing lesions in children and adolescents with posttraumatic diffuse axonal injury: improved detection and initial results. Radiology 227:332–339
Wycliffe ND, Choe J, Holshouser B et al (2004) Reliability in detection of hemorrhage in acute stroke by a new three-dimensional gradient recalled echo susceptibility-weighted imaging technique compared to computed tomography: a retrospective study. J Magn Reson Imaging 20:372–377
Nandigam RNK, Viswanathan A, Delgado P et al (2009) MR imaging detection of cerebral microbleeds: effect of susceptibility-weighted imaging, section thickness, and field strength. AJNR Am J Neuroradiol 30:338–343
Bilgic B, Gagoski BA, Cauley SF et al (2015) Wave-CAIPI for highly accelerated 3D imaging. Magn Reson Med 73:2152–2162
Conklin J, Longo MGF, Cauley SF et al (2019) Validation of highly accelerated wave-CAIPI SWI compared with conventional SWI and T2*-weighted gradient recalled-echo for routine clinical brain MRI at 3 T. AJNR Am J Neuroradiol 40:2073–2080
IMV Medical Information Division (2018) Benchmark Reports, MRI units
Canadian Agency for Drugs and Technologies in Health (CADTH): The Canadian Medical Imaging Inventory (2017). Available via https://www.cadth.ca/canadian-medical-imaging-inventory. Accessed 29 Nov 2019
Magnetic resonance imaging (MRI) equipment, operations and planning in the NHS: Report from the Clinical Imaging Board (2017). Available via https://www.rcr.ac.uk/sites/default/files/cib_mri_equipment_report.pdf. Accessed 29 Nov 2019
Canadian Agency for Drugs and Technologies in Health (CADTH): Magnetic resonance imaging for patients with implantable cardiac devices: a review of safety and guidelines. Available via https://www.ncbi.nlm.nih.gov/books/NBK545579/. Accessed 29 Nov 2019
Bernstein MA, Huston J, Ward HA (2006) Imaging artifacts at 3.0T. J Magn Reson Imaging 24:735–746
Conklin J, Cauley S, Setsompop K, et al (2018) Optimization and clinical evaluation of Wave-CAIPI susceptibility-weighted imaging (SWI) for detection of intracranial hemorrhage. In: Proceedings of the Radiological Society of North America. Chicago, IL, pp SSE24-04
Ahn S, Park SH, Lee KH (2013) How to demonstrate similarity by using noninferiority and equivalence statistical testing in radiology research. Radiology 267:328–338
Lakens D, Scheel AM, Isager PM (2018) Equivalence testing for psychological research : a tutorial. Adv Methods Pract Psychol Sci 1:259–269
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–174
Lee SJ, Park SH, Kim AY et al (2011) A prospective comparison of standard-dose CT enterography and 50% reduced-dose CT enterography with and without noise reduction for evaluating Crohn disease. AJR Am J Roentgenol 197:50–57
Fagundes J, Longo MG, Huang SY et al (2017) Diagnostic performance of a 10-minute gadolinium-enhanced brain MRI protocol compared with the standard clinical protocol for detection of intracranial enhancing lesions. AJNR Am J Neuroradiol 38:1689–1694
Acknowledgements
The authors are grateful to Wei Liu, Sinyeob Ahn, and Thomas Beck from Siemens Healthcare for supporting the development of the prototype sequence used in this study.
Funding
This study has received funding by John Conklin and Susie Huang. This work was conducted with support from Harvard Catalyst | The Harvard Clinical and Translational Science Center (National Center for Advancing Translational Sciences, National Institutes of Health Award P41 EB015896, R01 EB020613, and UL 1TR002541) and financial contributions from Harvard University and its affiliated academic healthcare centers.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is John Conklin and Susie Huang. The content is solely the responsibility of the authors and does not necessarily represent the official views of Harvard Catalyst, Harvard University and its affiliated academic healthcare centers, or the National Institutes of Health.
Conflict of interest
Wei Liu, Sinyeob Ahn, and Thomas Beck have connections with Siemens Healthcare and Shenzhen Magnetic Resonance Ltd. The remaining authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
M. Gabriela Figueiro Longo, one of our authors, has significant statistical expertise and provided statistical advice for this manuscript.
Informed consent
Written consent was not required by the IRB since no significant time (less than 2 minutes) was added to each exam.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• Prospective
• Comparative
• Performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Conklin, J., Figueiro Longo, M.G., Tabari, A. et al. Clinical validation of Wave-CAIPI susceptibility-weighted imaging for routine brain MRI at 1.5 T. Eur Radiol 32, 7128–7135 (2022). https://doi.org/10.1007/s00330-022-08871-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-022-08871-8