Abstract
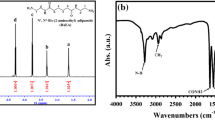
This study examined the kinetic reaction between hydrogenated dimer fatty acid C36 and 4,4′-methylene bis(cyclohexylamine) in the melt phase at 140, 145, 150, 160, and 165 ºC. The acid value, degree of polymerization, number average molecular weight, and weight average molecular weight were calculated at different times. Infrared spectroscopy was used to determine the structure of the resulting polymer. The kinetic degradation was carried out using thermogravimetric analysis, and thermodynamic and equilibrium constants were defined. Additionally, differential scanning calorimetric was used to determine the thermodynamic constants for melting. The kinetic of the polyamidation reaction was discovered to be of overall second order, with an activation energy of 187.45 kJ/g mol up to 80, 84, 92, 93, and 94% conversion at 140, 145, 150, 160, and 165 ºC, respectively. After reaching these conversion percentages, the reaction becomes overall third order.









Similar content being viewed by others
References
Ebewele RO (1996) Polymer science and technology, University of Benin Nigeria. P161
Brydson JA (1999) Plastics Materials, University of North London, p 478–480
Fan XD, Deng Y, Waterhouse J, Pfromm P (1998) Synthesis and characterization of polyamide resins from soy-based dimer acids and different amides. J Appl Polym Sci 68(2):305–314
Chen X, Zhong H, Jia L, Ning J, Tang R, Qiao J, Zhang Z (2002) Polyamides derived from piperazine and used for hot-melt adhesives; Synthesis and properties. Int J Adhes 22:75–79
Chen YH, Chang YC, Rwei SP (2022) High-value copolyamide 6 materials with colorless transparent and low water absorption upgraded from upcycled and biomass comonomers. Polymer 257:125269
Chermisinoff NP (1996) Polymer characterization laboratory techniques and analysis, Westwood, New Jersey, U.S.A,07675, p. 17–23
Deng Y, Fan XD, Waterhouse J (1999) Synthesis and characterization of soy-based copolyamides with different α–amino acids. J Appl Polym Sci 73:1081–1088
Vedanayagam HS, Kale V, Subbarao R (1992) Kinetics of reaction of C36 dimeric fatty acids and ethylenediamine in solution. J Appl Polym Sci 45:2245–2248
Guidici R (1999) Mathematical modeling of an industrial process of nylon-66 polymerization in a two phase flow tubular reactor. Chem Eng Sci 54:3243
Kale V, Vedanayagam HS, Rao MB, Devi KS, Rao SV, Lakshminarayana G (1988) Kinetics of reaction of ethylenediamine and dimeric fatty acids. J Appl Polym Sci 36:1517–1524
Heidarian J, Ghasem NM, Daud W (2003) Kinetic model for polymerization 0f dimer fatty acids with ethylenediamine. University of Malaya pada, vol 45, p 135
Heidarian J, Ghasem NM, Ashri M, Daud W (2004) Study on kinetics of polymerization of dimer fatty acids with ethylenediamine in the presence of catalyst. J Appl Polym Sci 100:85–93
Heidarian J, Ghasem NM, Ashri M, Daud W (2005) Kinetics of polymerization of dimer fatty acid with ethylenediamine after 90% conversion. J Polym Sci 206(6):658–663
Zifeng L, **angyu W, Shiling J, Ling Z, Zepeng W, Li**g H, Hongwei P, Huiliang Z, Lisong D (2022) The construction of super-tough polylactide/crosslinked polyamide blends by dynamic vulcanization. Polymer 202:110007
Zhou YN, Li JJ, Wang TT, Wu YY, Luo ZH (2022) Precision polymer synthesis by controlled radical polymerization: fusing the progress from polymer chemistry and reaction engineering. Prog Mater Sci 130:101555
Tohamy HAS, El-Sakhawy M, Kamel S (2023) Eco-friendly synthesis of carbon quantum dots as an effective adsorbent. J Fluoresc 33:423–435
Zhu C, Tang N, Gan J, Zhang X, Li Y, **n J, Cheng Y (2021) A pH-sensitive semi-interpenetrating polymer network hydrogels constructed by konjac glucomannan and poly (γ-glutamic acid): synthesis, characterization and swelling behavior. Int J Biol Macromol 185:229–239
Zhang X-Z, Zhang J-T, Zhuo R-X, Chu C-C (2002) Synthesis and properties of thermosensitive, crown ether incorporated poly( N-isopropylacrylamide) hydrogel. Polymer 43:4823–4827
Li D, Hu D, Tan J, Zhu Q, Liu Z, Su C, Chen J, Cao Y (2021) Design and synthesis of dimer acid-based waterborne polyurethane as water- and oil-resistant coating for paper substrates. Mater Des 211:110142
Tonbul Y, Yurdakoj K (2001) Thermogravimetric investigation of the Dehydration, Turkish bentonite. Turk J Chem 25:P33-339
Tohamy HAS, El-Sakhawy M, Kamel S (2022) Development of magnetite/graphene oxide hydrogels from agricultural wastes for water treatment. J Renew Mater 10:1889–1909
Tohamy HAS, El-Sakhawy M, Kamel S (2022) Carbon nanotubes from agricultural wastes: effective environmental adsorbent. Egypt J Chem 65:437–446
Tohamy HAS (2022) Reinforced modified carboxymethyl cellulose films with graphene oxide/silver nanoparticles as antimicrobial agents. Egypt J Chem 65:509–518
Tohamy HAS, El-Sakhawy M, Strachota B, Strachota A, Pavlova E, Barbosa SM, Kamel S (2023) Temperature- and pH-responsive super-absorbent hydrogel based on grafted cellulose and capable of heavy metal removal from aqueous solutions. Gels 9:296. https://doi.org/10.3390/gels9040296
Jia F, Mao JL, Yang XY, Ma Y, Yao C (2013) Thermal, physical and mechanical properties of hydrogenated dimer acid-based Nylon 636/Nylon 66 copolymers. Chin Chem Lett 24:654–658
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) funded by the Korean government (MSIT) (No. 2022R1A2C1006743).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
AL Mohammad, H., Kaseem, M. Kinetics of reaction between hydrogenated dimer fatty acid C36 and 4,4′-methylene bis(cyclohexylamine): determination of thermodynamic constants. Polym. Bull. 81, 6587–6601 (2024). https://doi.org/10.1007/s00289-023-05026-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-023-05026-7