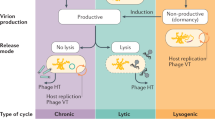
Abstract
Marine bacteriophages have been well characterized in terms of decay rates, population dynamics in relation to their hosts, and their impacts on biogeochemical cycles in the global ocean. Knowledge in soil bacteriophage ecology lags considerably behind, with few studies documenting population dynamics with hosts and even fewer reporting phage decay rates. By using sterile soil or aquatic microcosms inoculated with single bacteriophage isolates, phage decay rates (loss of infectivity over time) were determined, independent of host interactions, for 5 model phage isolates. Decay rates varied by phage from 0.11–2.07% h−1 in soils to 0.07–0.28% h−1 in aquatic microcosms. For phages incubated in both soil and aquatic microcosms, the observed decay rate was consistently higher in soil microcosms than in aquatic microcosms by at least a factor of two. However, when decay rates for soil phage isolates in the present study were compared to those reported for marine and freshwater phage isolates from previous studies, the decay constants for soil phages were, on average, 4 times lower than those for aquatic phages. Slower rates of phage decay in soils indicate a lower turnover rate, which may have subsequent and potentially far-reaching impacts on virus-mediated mortality and bacterial activity. The wide range of decay rates observed in the present study and the lack of information on this critical aspect of virus-host dynamics in soil emphasizes the need for continued research in this field.


Similar content being viewed by others
Data Availability
Upon acceptance, raw data regarding infectious particle counts (pfu gdw−1, pfu ml−1) for all experiments will be provided via figshare (10.6084/m9.figshare.21897168).
Code Availability
Not applicable.
References
Bergh Ø, BØrsheim KY, Bratbak G, Heldal M, (1989) High abundance of viruses found in aquatic environments. Nature 340:467–468. https://doi.org/10.1038/340467a0
Borsheim KY (1993) Native marine bacteriophages. FEMS Microbiol Lett 102:141–159. https://doi.org/10.1111/j.1574-6968.1993.tb05805.x
Thingstad T, Lignell R (1997) Theoretical models for the control of bacterial growth rate, abundance, diversity and carbon demand. Aquat Microb Ecol 13:19–27. https://doi.org/10.3354/ame013019
Fuhrman JA, Schwalbach M (2003) Viral influence on aquatic bacterial communities. Biol Bull 204:192–195. https://doi.org/10.2307/1543557
Fuhrman JA (1999) Marine viruses and their biogeochemical and ecological effects. Nature 399:541–548. https://doi.org/10.1038/21119
Bonilla-Findji O, Malits A, Lefèvre D et al (2008) Viral effects on bacterial respiration, production and growth efficiency: Consistent trends in the Southern Ocean and the Mediterranean Sea. Deep Sea Res Part II 55:790–800. https://doi.org/10.1016/j.dsr2.2007.12.004
Kimura M, Jia Z-J, Nakayama N, Asakawa S (2008) Ecology of viruses in soils: Past, present and future perspectives. Soil Sci Plant Nutr 54:1–32. https://doi.org/10.1111/j.1747-0765.2007.00197.x
Williamson KE, Fuhrmann JJ, Wommack KE, Radosevich M (2017) Viruses in soil ecosystems: an unknown quantity within an unexplored territory. Annual Review of Virology 4:201–219. https://doi.org/10.1146/annurev-virology-101416-041639
Ashelford KE, Day MJ, Fry JC (2003) Elevated abundance of bacteriophage infecting bacteria in soil. Appl Environ Microbiol 69:285–289. https://doi.org/10.1128/AEM.69.1.285-289.2003
Williamson KE, Radosevich M, Wommack KE (2005) Abundance and diversity of viruses in six delaware soils. Appl Environ Microbiol 71:3119–3125. https://doi.org/10.1128/AEM.71.6.3119-3125.2005
Heldal M, Bratbak G (1991) Production and decay of viruses in aquatic environments. Mar Ecol Prog Ser 72:205–212. https://doi.org/10.3354/meps072205
Hennes KP, Suttle CA (1995) Direct counts of viruses in natural waters and laboratory cultures by epifluorescence microscopy. Limnol Oceanogr 40:1050–1055. https://doi.org/10.4319/lo.1995.40.6.1050
Wommack KE, Colwell RR (2000) Virioplankton: Viruses in aquatic ecosystems. Microbiol Mol Biol Rev 64:69–114. https://doi.org/10.1128/MMBR.64.1.69-114.2000
Mathias CB, Kirschner A, Velimirov B (1995) Seasonal variations of virus abundance and viral control of the bacterial production in a backwater system of the danube river. Appl Environ Microbiol 61:3734–3740. https://doi.org/10.1128/aem.61.10.3734-3740.1995
Peduzzi P, Schiemer F (2004) Bacteria and viruses in the water column of tropical freshwater reservoirs. Environ Microbiol 6:707–715. https://doi.org/10.1111/j.1462-2920.2004.00602.x
Thomas R, Berdjeb L, Sime-Ngando T, Jacquet S (2011) Viral abundance, production, decay rates and life strategies (lysogeny versus lysis) in Lake Bourget (France). Environ Microbiol 13:616–630. https://doi.org/10.1111/j.1462-2920.2010.02364.x
Wommack KE, Hill RT, Muller TA, Colwell RR (1996) Effects of sunlight on bacteriophage viability and structure. Appl Environ Microbiol 62:1336–1341. https://doi.org/10.1128/aem.62.4.1336-1341.1996
Gerba CP (1984) Applied and theoretical aspects of virus adsorption to surfaces. Adv Appl Microbiol 30:133–168
Yeager JG, O’Brien RT (1979) Enterovirus inactivation in soil. Appl Environ Microbiol 38:694–701. https://doi.org/10.1128/aem.38.4.694-701.1979
Hurst CJ, Gerba CP, Cech I (1980) Effects of environmental variables and soil characteristics on virus survival in soil. Appl Environ Microbiol 40:1067–1079. https://doi.org/10.1128/aem.40.6.1067-1079.1980
Yates MV, Gerba CP, Kelley LM (1985) Virus persistence in groundwater. Appl Environ Microbiol 49:778–781. https://doi.org/10.1128/aem.49.4.778-781.1985
Williams ST, Mortimer AM, Manchester LN (1987) Phage ecology. Wiley, New York
Booncharoen N, Mongkolsuk S, Sirikanchana K (2018) Comparative persistence of human sewage-specific enterococcal bacteriophages in freshwater and seawater. Appl Microbiol Biotechnol 102:6235–6246. https://doi.org/10.1007/s00253-018-9079-1
Suttle CA, Chen F (1992) Mechanisms and rates of decay of marine viruses in seawater. Appl Environ Microbiol 58:3721–3729. https://doi.org/10.1128/aem.58.11.3721-3729.1992
Goyal SM, Gerba CP (1979) Comparative adsorption of human enteroviruses, simian rotavirus, and selected bacteriophages to soils. Appl Environ Microbiol 38:241–247. https://doi.org/10.1128/aem.38.2.241-247.1979
Gill J, Abedon ST (2003) Bacteriophage ecology and plants. APSnet Features. https://doi.org/10.1094/APSnetFeature-2003-1103
Vettori C, Gallori E, Stotzky G (2000) Clay minerals protect bacteriophage PBS1 of Bacillus subtilis against inactivation and loss of transducing ability by UV radiation. Environ Microbiol 1:347
Noble RT, Fuhrman JA (2000) Rapid virus production and removal as measured with fluorescently labeled viruses as tracers. Appl Environ Microbiol 66:3790–3797. https://doi.org/10.1128/AEM.66.9.3790-3797.2000
Schwarz KR, Sidhu JPS, Toze S et al (2019) Decay rates of Escherichia coli, Enterococcus spp., F-specific bacteriophage MS2, somatic coliphage and human adenovirus in facultative pond sludge. Water Res 154:62–71. https://doi.org/10.1016/j.watres.2019.01.027
Pantastico-Caldas M, Duncan KE, Istock CA, Bell JA (1992) Population dynamics of bacteriophage and bacillus subtilis in soil. Ecology 73:1888–1902. https://doi.org/10.2307/1940040
Manchester L (1986) Modelling the interaction of streptomycetes and their phage. University of Liverpool
Williams FP, Stetler RE, Safferman RS (2001) Chapter 7: Method for Recovering Viruses from Sludges, Soils, Sediments and Other Solids. USEPA Manual of Methods for Virology. U.S. Environmental Protection Agency, Washington, D.C., pp 202–217
Roberts BN, Bailey RH, McLaughlin MR, Brooks JP (2016) Decay rates of zoonotic pathogens and viral surrogates in soils amended with biosolids and manures and comparison of qPCR and culture derived rates. Sci Total Environ 573:671–679. https://doi.org/10.1016/j.scitotenv.2016.08.088
Kropinski AM, Mazzocco A, Waddell TE et al (2009) Enumeration of bacteriophages by double agar overlay plaque assay. In: Clokie MRJ, Kropinski AM (eds) Bacteriophages. Humana Press, Totowa, NJ, pp 69–76
Berns AE, Philipp H, Narres H-D et al (2008) Effect of gamma-sterilization and autoclaving on soil organic matter structure as studied by solid state NMR, UV and fluorescence spectroscopy. Eur J Soil Sci 59:540–550. https://doi.org/10.1111/j.1365-2389.2008.01016.x
Ogunseitan OA, Sayler GS, Miller RV (1990) Dynamic interactions of pseudomonas aeruginosa and bacteriophages in lake water. Microb Ecol 19:171–185
Gersberg RM, Lyon SR, Brenner R, Elkins BV (1987) Fate of viruses in artificial wetlands. Appl Environ Microbiol 53:731–736. https://doi.org/10.1128/aem.53.4.731-736.1987
Noble RT, Fuhrman JA (1997) Virus decay and its causes in coastal waters. Appl Environ Microbiol 63:77–83. https://doi.org/10.1128/aem.63.1.77-83.1997
Miller ES, Kutter E, Mosig G et al (2003) Bacteriophage T4 Genome. Microbiol Mol Biol Rev 67:86–156. https://doi.org/10.1128/mmbr.67.1.86-156.2003
Acknowledgements
This work was supported by a Biology Research and Opportunity Endowment award to KEW and a Mary E. Ferguson Memorial Research Grant award and Charles Center award to AGD. The authors wish to thank Rebecca Quinn, Erin Goodstein, Jacob Warner, and Krista Wink for their help in testing and develo** the microcosm design and extraction procedures.
Funding
This work was completed with funding from the Roy R. Charles Center at the College of William and Mary and a Mary E. Ferguson Research Award to AGD, and a William & Mary Biology Research and Opportunity Endowment award to KEW.
Author information
Authors and Affiliations
Contributions
AGD helped conceive and execute all experiments and wrote the manuscript; SAB executed experiments with B. subtilis; MMZ isolated the phages used in experiments and refined extraction protocols and microcosm design; KEW conceived all experiments, assisted with data processing, and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
The authors consent to publish this work in Current Microbiology.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
DiPietro, A.G., Bryant, S.A., Zanger, M.M. et al. Understanding Viral Impacts in Soil Microbial Ecology Through the Persistence and Decay of Infectious Bacteriophages. Curr Microbiol 80, 276 (2023). https://doi.org/10.1007/s00284-023-03386-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-023-03386-x