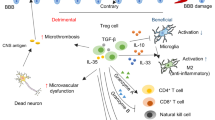
Abstract
Unrestrained excessive inflammatory responses exacerbate ischemic brain injury and impede post-stroke brain recovery. CD4+CD25+Foxp3+ regulatory T (Treg) cells play important immunosuppressive roles to curtail inflammatory responses and regain immune homeostasis after stroke. Accumulating evidence confirms that Treg cells are neuroprotective at the acute stage after stroke and promote brain repair at the chronic phases. The beneficial effects of Treg cells are mediated by diverse mechanisms involving cell–cell interactions and soluble factor release. Multiple types of cells, including both immune cells and non-immune CNS cells, have been identified to be cellular targets of Treg cells. In this review, we summarize recent findings regarding the function of Treg cells in ischemic stroke and the underlying cellular and molecular mechanisms. The protective and reparative properties of Treg cells endorse them as good candidates for immune therapy. Strategies that boost the numbers and functions of Treg cells have been actively develo** in the fields of transplantation and autoimmune diseases. We discuss the approaches for Treg cell expansion that have been tested in stroke models. The application of these approaches to stroke patients may bring new hope for stroke treatments.


Similar content being viewed by others
References
Nishizuka Y, Sakakura T (1969) Thymus and reproduction: sex-linked dysgenesia of the gonad after neonatal thymectomy in mice. Science 166:753–755
Kojima A, Prehn RT (1981) Genetic susceptibility to post-thymectomy autoimmune diseases in mice. Immunogenetics 14:15–27
Hori S, Nomura T, Sakaguchi S (2003) Control of regulatory T cell development by the transcription factor Foxp3. Science 299:1057–1061
Munoz-Rojas AR, Mathis D (2021) Tissue regulatory T cells: regulatory chameleons. Nat Rev Immunol 21:597–611
Anrather J, Iadecola C (2016) Inflammation and stroke: an overview. Neurotherapeutics 13:661–670
Rayasam A, Hsu M, Hernández G, Kijak J, Lindstedt A, Gerhart C, Sandor M, Fabry Z (2017) Contrasting roles of immune cells in tissue injury and repair in stroke: the dark and bright side of immunity in the brain. Neurochem Int 107:104–116
Seifert HA, Collier LA, Chapman CB, Benkovic SA, Willing AE, Pennypacker KR (2014) Pro-inflammatory interferon gamma signaling is directly associated with stroke induced neurodegeneration. J Neuroimmune Pharmacol 9:679–689
Yilmaz G, Arumugam TV, Stokes KY, Granger DN (2006) Role of T lymphocytes and interferon-gamma in ischemic stroke. Circulation 113:2105–2112
Shevach EM (2009) Mechanisms of foxp3+ T regulatory cell-mediated suppression. Immunity 30:636–645
Ralainirina N, Poli A, Michel T, Poos L, Andrès E, Hentges F, Zimmer J (2007) Control of NK cell functions by CD4+CD25+ regulatory T cells. J Leukoc Biol 81:144–153
Ito M, Komai K, Mise-Omata S, Iizuka-Koga M, Noguchi Y, Kondo T, Sakai R, Matsuo K, Nakayama T, Yoshie O, Nakatsukasa H, Chikuma S, Shichita T, Yoshimura A (2019) Brain regulatory T cells suppress astrogliosis and potentiate neurological recovery. Nature 565:246–250
Yan J, Greer JM, Etherington K, Cadigan GP, Cavanagh H, Henderson RD, O’Sullivan JD, Pandian JD, Read SJ, McCombe PA (2009) Immune activation in the peripheral blood of patients with acute ischemic stroke. J Neuroimmunol 206:112–117
Urra X, Cervera A, Villamor N, Planas AM, Chamorro A (2009) Harms and benefits of lymphocyte subpopulations in patients with acute stroke. Neuroscience 158:1174–1183
Mao L, Li P, Zhu W, Cai W, Liu Z, Wang Y, Luo W, Stetler RA, Leak RK, Yu W, Gao Y, Chen J, Chen G, Hu X (2017) Regulatory T cells ameliorate tissue plasminogen activator-induced brain haemorrhage after stroke. Brain 140:1914–1931
Prass K, Meisel C, Höflich C, Braun J, Halle E, Wolf T, Ruscher K, Victorov IV, Priller J, Dirnagl U, Volk HD, Meisel A (2003) Stroke-induced immunodeficiency promotes spontaneous bacterial infections and is mediated by sympathetic activation reversal by poststroke T helper cell type 1-like immunostimulation. J Exp Med 198:725–736
Offner H, Subramanian S, Parker SM, Wang C, Afentoulis ME, Lewis A, Vandenbark AA, Hurn PD (2006) Splenic atrophy in experimental stroke is accompanied by increased regulatory T cells and circulating macrophages. J Immunol 176:6523–6531
Santamaria-Cadavid M, Rodriguez-Castro E, Rodriguez-Yanez M, Arias-Rivas S, Lopez-Dequidt I, Perez-Mato M, Rodriguez-Perez M, Lopez-Loureiro I, Hervella P, Campos F, Castillo J, Iglesias-Rey R, Sobrino T (2020) Regulatory T cells participate in the recovery of ischemic stroke patients. BMC Neurol 20:68
Yan J, Read SJ, Henderson RD, Hull R, O’Sullivan JD, McCombe PA, Greer JM (2012) Frequency and function of regulatory T cells after ischaemic stroke in humans. J Neuroimmunol 243:89–94
Pang X, Qian W (2017) Changes in regulatory T-cell levels in acute cerebral ischemia. J Neurol Surg A Cent Eur Neurosurg 78:374–379
Zhang J, Liu G, Chen D, Wang Z, Chen D, Liu Y, Yu W (2020) The combination of CC chemokine receptor type 5(CCR5) and Treg cells predicts prognosis in patients with ischemic stroke. J Neuroimmunol 349:577404
Ruhnau J, Schulze J, von Sarnowski B, Heinrich M, Langner S, Potschke C, Wilden A, Kessler C, Broker BM, Vogelgesang A, Dressel A (2016) Reduced numbers and impaired function of regulatory T cells in peripheral blood of ischemic stroke patients. Mediators Inflamm 2016:2974605
Foy TM, Page DM, Waldschmidt TJ, Schoneveld A, Laman JD, Masters SR, Tygrett L, Ledbetter JA, Aruffo A, Claassen E, Xu JC, Flavell RA, Oehen S, Hedrick SM, Noelle RJ (1995) An essential role for gp39, the ligand for CD40, in thymic selection. J Exp Med 182:1377–1388
Li P, Wang L, Zhou Y, Gan Y, Zhu W, **a Y, Jiang X, Watkins S, Vazquez A, Thomson AW, Chen J, Yu W, Hu X. 2017. C-C chemokine receptor type 5 (CCR5)-mediated docking of transferred Tregs protects against early blood-brain barrier disruption After Stroke. J Am Heart Assoc 6
Lucaciu A, Kuhn H, Trautmann S, Ferreiros N, Steinmetz H, Pfeilschifter J, Brunkhorst R, Pfeilschifter W, Subburayalu J, Vutukuri R. 2020. A sphingosine 1-phosphate gradient is linked to the cerebral recruitment of T helper and regulatory T helper cells during acute ischemic stroke. Int J Mol Sci 21
Lee HT, Liu SP, Lin CH, Lee SW, Hsu CY, Sytwu HK, Hsieh CH, Shyu WC (2017) A crucial role of CXCL14 for promoting regulatory T cells activation in stroke. Theranostics 7:855–875
Shi L, Sun Z, Su W, Xu F, **e D, Zhang Q, Dai X, Iyer K, Hitchens TK, Foley LM, Li S, Stolz DB, Chen K, Ding Y, Thomson AW, Leak RK, Chen J, Hu X (2021) Treg cell-derived osteopontin promotes microglia-mediated white matter repair after ischemic stroke. Immunity 54(1527–42):e8
Guo S, Luo Y (2020) Brain Foxp3(+) regulatory T cells can be expanded by Interleukin-33 in mouse ischemic stroke. Int Immunopharmacol 81:106027
**e D, Miao W, Xu F, Yuan C, Li S, Wang C, Junagade A, Hu X (2022) IL-33/ST2 axis protects against traumatic brain injury through enhancing the function of regulatory T cells. Front Immunol 13:860772
Liesz A, Suri-Payer E, Veltkamp C, Doerr H, Sommer C, Rivest S, Giese T, Veltkamp R (2009) Regulatory T cells are key cerebroprotective immunomodulators in acute experimental stroke. Nat Med 15:192–199
Kleinschnitz C, Kraft P, Dreykluft A, Hagedorn I, Gobel K, Schuhmann MK, Langhauser F, Helluy X, Schwarz T, Bittner S, Mayer CT, Brede M, Varallyay C, Pham M, Bendszus M, Jakob P, Magnus T, Meuth SG, Iwakura Y, Zernecke A, Sparwasser T, Nieswandt B, Stoll G, Wiendl H (2013) Regulatory T cells are strong promoters of acute ischemic stroke in mice by inducing dysfunction of the cerebral microvasculature. Blood 121:679–691
Ren X, Akiyoshi K, Vandenbark AA, Hurn PD, Offner H (2011) CD4+FoxP3+ regulatory T-cells in cerebral ischemic stroke. Metab Brain Dis 26:87–90
Liesz A, Hu X, Kleinschnitz C, Offner H (2015) Functional role of regulatory lymphocytes in stroke: facts and controversies. Stroke 46:1422–1430
Wang J, **e L, Yang C, Ren C, Zhou K, Wang B, Zhang Z, Wang Y, ** K, Yang GY (2015) Activated regulatory T cell regulates neural stem cell proliferation in the subventricular zone of normal and ischemic mouse brain through interleukin 10. Front Cell Neurosci 9:361
Li P, Gan Y, Sun BL, Zhang F, Lu B, Gao Y, Liang W, Thomson AW, Chen J, Hu X (2013) Adoptive regulatory T-cell therapy protects against cerebral ischemia. Ann Neurol 74:458–471
Li P, Mao L, Liu X, Gan Y, Zheng J, Thomson AW, Gao Y, Chen J, Hu X (2014) Essential role of program death 1-ligand 1 in regulatory T-cell-afforded protection against blood-brain barrier damage after stroke. Stroke 45:857–864
Hu X, Leak RK, Thomson AW, Yu F, **a Y, Wechsler LR, Chen J (2018) Promises and limitations of immune cell-based therapies in neurological disorders. Nat Rev Neurol 14:559–568
Collison LW, Workman CJ, Kuo TT, Boyd K, Wang Y, Vignali KM, Cross R, Sehy D, Blumberg RS, Vignali DA (2007) The inhibitory cytokine IL-35 contributes to regulatory T-cell function. Nature 450:566–569
Chen ML, Pittet MJ, Gorelik L, Flavell RA, Weissleder R, von Boehmer H, Khazaie K (2005) Regulatory T cells suppress tumor-specific CD8 T cell cytotoxicity through TGF-beta signals in vivo. Proc Natl Acad Sci U S A 102:419–424
Benakis C, Brea D, Caballero S, Faraco G, Moore J, Murphy M, Sita G, Racchumi G, Ling L, Pamer EG, Iadecola C, Anrather J (2016) Commensal microbiota affects ischemic stroke outcome by regulating intestinal gammadelta T cells. Nat Med 22:516–523
**e L, Choudhury GR, Winters A, Yang SH, ** K (2015) Cerebral regulatory T cells restrain microglia/macrophage-mediated inflammatory responses via IL-10. Eur J Immunol 45:180–191
Kim M, Kim SD, Kim KI, Jeon EH, Kim MG, Lim YR, Lkhagva-Yondon E, Oh Y, Na K, Chung YC, ** BK, Song YS, Jeon MS (2021) Dynamics of T lymphocyte between the periphery and the brain from the acute to the chronic phase following ischemic stroke in mice. Exp Neurobiol 30:155–169
Sanjabi S, Oh SA, Li MO. 2017. Regulation of the immune response by TGF-β: from conception to autoimmunity and infection. Cold Spring Harb Perspect Biol 9
Dolati S, Ahmadi M, Khalili M, Taheraghdam AA, Siahmansouri H, Babaloo Z, Aghebati-Maleki L, Jadidi-Niaragh F, Younesi V, Yousefi M (2018) Peripheral Th17/Treg imbalance in elderly patients with ischemic stroke. Neurol Sci 39:647–654
Wang M, Wang Y, He J, Wei S, Zhang N, Liu F, Liu X, Kang Y, Yao X (2013) Albumin induces neuroprotection against ischemic stroke by altering Toll-like receptor 4 and regulatory T cells in mice. CNS Neurol Disord Drug Targets 12:220–227
Liu K, Huang A, Nie J, Tan J, **ng S, Qu Y, Jiang K (2021) IL-35 regulates the function of immune cells in tumor microenvironment. Front Immunol 12:683332
Xu C, Zhu H, Shen R, Feng Q, Zhou H, Zhao Z (2018) IL-35 is a protective immunomodulator in brain ischemic injury in mice. Neurochem Res 43:1454–1463
**ao T, Qu H, Zeng Z, Li C, Wan J. 2022. Interleukin-35 from interleukin-4-stimulated macrophages alleviates oxygen glucose deprivation/re-oxygenation-induced neuronal cell death via the Wnt/β-catenin signaling pathway. Neurotox Res
Chen W, ** W, Hardegen N, Lei KJ, Li L, Marinos N, McGrady G, Wahl SM (2003) Conversion of peripheral CD4+CD25- naive T cells to CD4+CD25+ regulatory T cells by TGF-beta induction of transcription factor Foxp3. J Exp Med 198:1875–1886
Zheng SG, Wang JH, Gray JD, Soucier H, Horwitz DA (2004) Natural and induced CD4+CD25+ cells educate CD4+CD25- cells to develop suppressive activity: the role of IL-2, TGF-beta, and IL-10. J Immunol 172:5213–5221
Bodhankar S, Chen Y, Lapato A, Dotson AL, Wang J, Vandenbark AA, Saugstad JA, Offner H (2015) PD-L1 monoclonal antibody treats ischemic stroke by controlling central nervous system inflammation. Stroke 46:2926–2934
Bodhankar S, Chen Y, Vandenbark AA, Murphy SJ, Offner H (2013) PD-L1 enhances CNS inflammation and infarct volume following experimental stroke in mice in opposition to PD-1. J Neuroinflammation 10:111
Topalian SL, Drake CG, Pardoll DM (2012) Targeting the PD-1/B7-H1(PD-L1) pathway to activate anti-tumor immunity. Curr Opin Immunol 24:207–212
Wing K, Onishi Y, Prieto-Martin P, Yamaguchi T, Miyara M, Fehervari Z, Nomura T, Sakaguchi S (2008) CTLA-4 control over Foxp3+ regulatory T cell function. Science 322:271–275
Qureshi OS, Zheng Y, Nakamura K, Attridge K, Manzotti C, Schmidt EM, Baker J, Jeffery LE, Kaur S, Briggs Z, Hou TZ, Futter CE, Anderson G, Walker LS, Sansom DM (2011) Trans-endocytosis of CD80 and CD86: a molecular basis for the cell-extrinsic function of CTLA-4. Science 332:600–603
Ma J, Shen L, Bao L, Yuan H, Wang Y, Liu H, Wang Q (2021) A novel prognosis prediction model, including cytotoxic T lymphocyte-associated antigen-4, ischemia-modified albumin, lipoprotein-associated phospholipase A2, glial fibrillary acidic protein, and homocysteine, for ischemic stroke in the Chinese hypertensive population. J Clin Lab Anal 35:e23756
Noh MY, Lee WM, Lee SJ, Kim HY, Kim SH, Kim YS (2018) Regulatory T cells increase after treatment with poly (ADP-ribose) polymerase-1 inhibitor in ischemic stroke patients. Int Immunopharmacol 60:104–110
Garín MI, Chu CC, Golshayan D, Cernuda-Morollón E, Wait R, Lechler RI (2007) Galectin-1: a key effector of regulation mediated by CD4+CD25+ T cells. Blood 109:2058–2065
Nakamura K, Kitani A, Strober W (2001) Cell contact-dependent immunosuppression by CD4(+)CD25(+) regulatory T cells is mediated by cell surface-bound transforming growth factor beta. J Exp Med 194:629–644
Cheng YH, Jiang YF, Qin C, Shang K, Yuan Y, Wei XJ, Xu Z, Luo X, Wang W, Qu WS (2022) Galectin-1 contributes to vascular remodeling and blood flow recovery after cerebral ischemia in mice. Transl Stroke Res 13:160–170
Wang J, **a J, Zhang F, Shi Y, Wu Y, Pu H, Liou AK, Leak RK, Yu X, Chen L, Chen J (2015) Galectin-1-secreting neural stem cells elicit long-term neuroprotection against ischemic brain injury. Sci Rep 5:9621
Ishibashi S, Kuroiwa T, Sakaguchi M, Sun L, Kadoya T, Okano H, Mizusawa H (2007) Galectin-1 regulates neurogenesis in the subventricular zone and promotes functional recovery after stroke. Exp Neurol 207:302–313
Qu WS, Wang YH, Wang JP, Tang YX, Zhang Q, Tian DS, Yu ZY, **e MJ, Wang W (2010) Galectin-1 enhances astrocytic BDNF production and improves functional outcome in rats following ischemia. Neurochem Res 35:1716–1724
Wang Y, Liu G, Hong D, Chen F, Ji X, Cao G (2016) White matter injury in ischemic stroke. Prog Neurobiol 141:45–60
Dombrowski Y, O’Hagan T, Dittmer M, Penalva R, Mayoral SR, Bankhead P, Fleville S, Eleftheriadis G, Zhao C, Naughton M, Hassan R, Moffat J, Falconer J, Boyd A, Hamilton P, Allen IV, Kissenpfennig A, Moynagh PN, Evergren E, Perbal B, Williams AC, Ingram RJ, Chan JR, Franklin RJM, Fitzgerald DC (2017) Regulatory T cells promote myelin regeneration in the central nervous system. Nat Neurosci 20:674–680
de la Vega GN, Penalva R, Dittmer M, Naughton M, Falconer J, Moffat J, de la Fuente AG, Hombrebueno JR, Lin Z, Perbal B, Ingram RJ, Evergren E, Fitzgerald DC (2020) Dynamic CCN3 expression in the murine CNS does not confer essential roles in myelination or remyelination. Proc Natl Acad Sci U S A 117:18018–18028
Hsieh J, Aimone JB, Kaspar BK, Kuwabara T, Nakashima K, Gage FH (2004) IGF-I instructs multipotent adult neural progenitor cells to become oligodendrocytes. J Cell Biol 164:111–122
Glezer I, Rivest S (2010) Oncostatin M is a novel glucocorticoid-dependent neuroinflammatory factor that enhances oligodendrocyte precursor cell activity in demyelinated sites. Brain Behav Immun 24:695–704
Hu X (2020) Microglia/macrophage polarization: fantasy or evidence of functional diversity? J Cereb Blood Flow Metab 40:S134–S136
Lyu J, **e D, Bhatia TN, Leak RK, Hu X, Jiang X (2021) Microglial/macrophage polarization and function in brain injury and repair after stroke. CNS Neurosci Ther 27:515–527
Reynolds AD, Banerjee R, Liu J, Gendelman HE, Mosley RL (2007) Neuroprotective activities of CD4+CD25+ regulatory T cells in an animal model of Parkinson’s disease. J Leukoc Biol 82:1083–1094
Reynolds AD, Stone DK, Mosley RL, Gendelman HE (2009) Nitrated {alpha}-synuclein-induced alterations in microglial immunity are regulated by CD4+ T cell subsets. J Immunol 182:4137–4149
Beers DR, Henkel JS, Zhao W, Wang J, Huang A, Wen S, Liao B, Appel SH (2011) Endogenous regulatory T lymphocytes ameliorate amyotrophic lateral sclerosis in mice and correlate with disease progression in patients with amyotrophic lateral sclerosis. Brain 134:1293–1314
Zhao W, Beers DR, Liao B, Henkel JS, Appel SH (2012) Regulatory T lymphocytes from ALS mice suppress microglia and effector T lymphocytes through different cytokine-mediated mechanisms. Neurobiol Dis 48:418–428
Moorman HR, Poschel D, Klement JD, Lu C, Redd PS, Liu K. 2020. Osteopontin: a key regulator of tumor progression and immunomodulation. Cancers (Basel) 12
He X, Lin S, Yang L, Tan P, Ma P, Qiu P, Zheng C, Zhang X, Kang W, Lin W (2021) Programmed death protein 1 is essential for maintaining the anti-inflammatory function of infiltrating regulatory T cells in a murine spinal cord injury model. J Neuroimmunol 354:577546
Araque A, Carmignoto G, Haydon PG (2001) Dynamic signaling between astrocytes and neurons. Annu Rev Physiol 63:795–813
Haydon PG (2001) GLIA: listening and talking to the synapse. Nat Rev Neurosci 2:185–193
Faulkner JR, Herrmann JE, Woo MJ, Tansey KE, Doan NB, Sofroniew MV (2004) Reactive astrocytes protect tissue and preserve function after spinal cord injury. J Neurosci 24:2143–2155
Li L, Lundkvist A, Andersson D, Wilhelmsson U, Nagai N, Pardo AC, Nodin C, Stahlberg A, Aprico K, Larsson K, Yabe T, Moons L, Fotheringham A, Davies I, Carmeliet P, Schwartz JP, Pekna M, Kubista M, Blomstrand F, Maragakis N, Nilsson M, Pekny M (2008) Protective role of reactive astrocytes in brain ischemia. J Cereb Blood Flow Metab 28:468–481
Wanner IB, Deik A, Torres M, Rosendahl A, Neary JT, Lemmon VP, Bixby JL (2008) A new in vitro model of the glial scar inhibits axon growth. Glia 56:1691–1709
Ohtake Y, Li S (2015) Molecular mechanisms of scar-sourced axon growth inhibitors. Brain Res 1619:22–35
Zador Z, Stiver S, Wang V, Manley GT. 2009. Role of aquaporin-4 in cerebral edema and stroke. Handb Exp Pharmacol: 159–70
Brambilla R, Bracchi-Ricard V, Hu WH, Frydel B, Bramwell A, Karmally S, Green EJ, Bethea JR (2005) Inhibition of astroglial nuclear factor kappaB reduces inflammation and improves functional recovery after spinal cord injury. J Exp Med 202:145–156
Argaw AT, Gurfein BT, Zhang Y, Zameer A, John GR (2009) VEGF-mediated disruption of endothelial CLN-5 promotes blood-brain barrier breakdown. Proc Natl Acad Sci U S A 106:1977–1982
Liddelow SA, Barres BA (2017) Reactive astrocytes: production, function, and therapeutic potential. Immunity 46:957–967
Zaiss DM, van Loosdregt J, Gorlani A, Bekker CP, Gröne A, Sibilia M, van Bergen en Henegouwen PM, Roovers RC, Coffer PJ, Sijts AJ. (2013) Amphiregulin enhances regulatory T cell-suppressive function via the epidermal growth factor receptor. Immunity 38:275–284
Arpaia N, Green JA, Moltedo B, Arvey A, Hemmers S, Yuan S, Treuting PM, Rudensky AY (2015) A distinct function of regulatory T cells in tissue protection. Cell 162:1078–1089
Mayo L, Cunha AP, Madi A, Beynon V, Yang Z, Alvarez JI, Prat A, Sobel RA, Kobzik L, Lassmann H, Quintana FJ, Weiner HL (2016) IL-10-dependent Tr1 cells attenuate astrocyte activation and ameliorate chronic central nervous system inflammation. Brain 139:1939–1957
Chan A, Yan J, Csurhes P, Greer J, McCombe P (2015) Circulating brain derived neurotrophic factor (BDNF) and frequency of BDNF positive T cells in peripheral blood in human ischemic stroke: effect on outcome. J Neuroimmunol 286:42–47
Raffin C, Vo LT, Bluestone JA (2020) T(reg) cell-based therapies: challenges and perspectives. Nat Rev Immunol 20:158–172
Yin J, Liao SX, He Y, Wang S, **a GH, Liu FT, Zhu JJ, You C, Chen Q, Zhou L, Pan SY, Zhou HW. 2015. Dysbiosis of gut microbiota with reduced trimethylamine-N-oxide level in patients with large-artery atherosclerotic stroke or transient ischemic attack. J Am Heart Assoc 4
Yamashiro K, Tanaka R, Urabe T, Ueno Y, Yamashiro Y, Nomoto K, Takahashi T, Tsuji H, Asahara T, Hattori N (2017) Gut dysbiosis is associated with metabolism and systemic inflammation in patients with ischemic stroke. PLoS ONE 12:e0171521
Haak BW, Westendorp WF, van Engelen TSR, Brands X, Brouwer MC, Vermeij JD, Hugenholtz F, Verhoeven A, Derks RJ, Giera M, Nederkoorn PJ, de Vos WM, van de Beek D, Wiersinga WJ (2021) Disruptions of anaerobic gut bacteria are associated with stroke and post-stroke infection: a prospective case-control study. Transl Stroke Res 12:581–592
Houlden A, Goldrick M, Brough D, Vizi ES, Lenart N, Martinecz B, Roberts IS, Denes A (2016) Brain injury induces specific changes in the caecal microbiota of mice via altered autonomic activity and mucoprotein production. Brain Behav Immun 57:10–20
Singh V, Roth S, Llovera G, Sadler R, Garzetti D, Stecher B, Dichgans M, Liesz A (2016) Microbiota dysbiosis controls the neuroinflammatory response after stroke. J Neurosci 36:7428–7440
Sadler R, Singh V, Benakis C, Garzetti D, Brea D, Stecher B, Anrather J, Liesz A (2017) Microbiota differences between commercial breeders impacts the post-stroke immune response. Brain Behav Immun 66:23–30
Benakis C, Poon C, Lane D, Brea D, Sita G, Moore J, Murphy M, Racchumi G, Iadecola C, Anrather J (2020) Distinct commensal bacterial signature in the gut is associated with acute and long-term protection from ischemic stroke. Stroke 51:1844–1854
Sadighi Akha AA (2018) Aging and the immune system: an overview. J Immunol Methods 463:21–26
Finger CE, Moreno-Gonzalez I, Gutierrez A, Moruno-Manchon JF, McCullough LD (2022) Age-related immune alterations and cerebrovascular inflammation. Mol Psychiatry 27:803–818
Raynor J, Lages CS, Shehata H, Hildeman DA, Chougnet CA (2012) Homeostasis and function of regulatory T cells in aging. Curr Opin Immunol 24:482–487
Hwang KA, Kim HR, Kang I (2009) Aging and human CD4(+) regulatory T cells. Mech Ageing Dev 130:509–517
Rosenkranz D, Weyer S, Tolosa E, Gaenslen A, Berg D, Leyhe T, Gasser T, Stoltze L (2007) Higher frequency of regulatory T cells in the elderly and increased suppressive activity in neurodegeneration. J Neuroimmunol 188:117–127
Garg SK, Delaney C, Toubai T, Ghosh A, Reddy P, Banerjee R, Yung R (2014) Aging is associated with increased regulatory T-cell function. Aging Cell 13:441–448
Zhao L, Sun L, Wang H, Ma H, Liu G, Zhao Y (2007) Changes of CD4+CD25+Foxp3+ regulatory T cells in aged Balb/c mice. J Leukoc Biol 81:1386–1394
Carpentier M, Chappert P, Kuhn C, Lalfer M, Flament H, Burlen-Defranoux O, Lantz O, Bandeira A, Malissen B, Davoust J, Gross DA (2013) Extrathymic induction of Foxp3(+) regulatory T cells declines with age in a T-cell intrinsic manner. Eur J Immunol 43:2598–2604
Chougnet CA, Tripathi P, Lages CS, Raynor J, Sholl A, Fink P, Plas DR, Hildeman DA (2011) A major role for Bim in regulatory T cell homeostasis. J Immunol 186:156–163
Rocamora-Reverte L, Melzer FL, Wurzner R, Weinberger B (2020) The complex role of regulatory T cells in immunity and aging. Front Immunol 11:616949
van der Geest KS, Abdulahad WH, Tete SM, Lorencetti PG, Horst G, Bos NA, Kroesen BJ, Brouwer E, Boots AM (2014) Aging disturbs the balance between effector and regulatory CD4+ T cells. Exp Gerontol 60:190–196
Nishioka T, Shimizu J, Iida R, Yamazaki S, Sakaguchi S (2006) CD4+CD25+Foxp3+ T cells and CD4+CD25-Foxp3+ T cells in aged mice. J Immunol 176:6586–6593
Jagger A, Shimojima Y, Goronzy JJ, Weyand CM (2014) Regulatory T cells and the immune aging process: a mini-review. Gerontology 60:130–137
Kuswanto W, Burzyn D, Panduro M, Wang KK, Jang YC, Wagers AJ, Benoist C, Mathis D (2016) Poor repair of skeletal muscle in aging mice reflects a defect in local, interleukin-33-dependent accumulation of regulatory T cells. Immunity 44:355–367
Afshan G, Afzal N, Qureshi S (2012) CD4+CD25(hi) regulatory T cells in healthy males and females mediate gender difference in the prevalence of autoimmune diseases. Clin Lab 58:567–571
Beckmann L, Obst S, Labusek N, Abberger H, Koster C, Klein-Hitpass L, Schumann S, Kleinschnitz C, Hermann DM, Felderhoff-Muser U, Bendix I, Hansen W, Herz J (2022) Regulatory T cells contribute to sexual dimorphism in neonatal hypoxic-ischemic brain injury. Stroke 53:381–390
Ahnstedt H, Patrizz A, Chauhan A, Roy-O’Reilly M, Furr JW, Spychala MS, D’Aigle J, Blixt FW, Zhu L, Bravo Alegria J, McCullough LD (2020) Sex differences in T cell immune responses, gut permeability and outcome after ischemic stroke in aged mice. Brain Behav Immun 87:556–567
Reddy J, Waldner H, Zhang X, Illes Z, Wucherpfennig KW, Sobel RA, Kuchroo VK (2005) Cutting edge: CD4+CD25+ regulatory T cells contribute to gender differences in susceptibility to experimental autoimmune encephalomyelitis. J Immunol 175:5591–5595
Gandhi VD, Cephus JY, Norlander AE, Chowdhury NU, Zhang J, Ceneviva ZJ, Tannous E, Polosukhin VV, Putz ND, Wickersham N, Singh A, Ware LB, Bastarache JA, Shaver CM, Chu HW, Peebles RS, Jr., Newcomb DC. 2022. Androgen receptor signaling promotes Treg suppressive function during allergic airway inflammation. J Clin Invest 132
Aristimuno C, Teijeiro R, Valor L, Alonso B, Tejera-Alhambra M, de Andres C, Minarro DO, Lopez-Lazareno N, Faure F, Sanchez-Ramon S (2012) Sex-hormone receptors pattern on regulatory T-cells: clinical implications for multiple sclerosis. Clin Exp Med 12:247–255
Goodman WA, Bedoyan SM, Havran HL, Richardson B, Cameron MJ, Pizarro TT (2020) Impaired estrogen signaling underlies regulatory T cell loss-of-function in the chronically inflamed intestine. Proc Natl Acad Sci U S A 117:17166–17176
Pernis AB (2007) Estrogen and CD4+ T cells. Curr Opin Rheumatol 19:414–420
Tian W, Jiang SY, Jiang X, Tamosiuniene R, Kim D, Guan T, Arsalane S, Pasupneti S, Voelkel NF, Tang Q, Nicolls MR (2021) The role of regulatory T cells in pulmonary arterial hypertension. Front Immunol 12:684657
Gourdy P, Araujo LM, Zhu R, Garmy-Susini B, Diem S, Laurell H, Leite-de-Moraes M, Dy M, Arnal JF, Bayard F, Herbelin A (2005) Relevance of sexual dimorphism to regulatory T cells: estradiol promotes IFN-gamma production by invariant natural killer T cells. Blood 105:2415–2420
Arenas-Ramirez N, Woytschak J, Boyman O (2015) Interleukin-2: biology, design and application. Trends Immunol 36:763–777
Hernandez R, Poder J, LaPorte KM, Malek TR. 2022. Engineering IL-2 for immunotherapy of autoimmunity and cancer. Nat Rev Immunol In press
Raffin C, Vo LT, Bluestone JA (2020) Treg cell-based therapies: challenges and perspectives. Nat Rev Immunol 20:158–172
Ellis GI, Sheppard NC, Riley JL (2021) Genetic engineering of T cells for immunotherapy. Nat Rev Genet 22:427–447
Fraser H, Safinia N, Grageda N, Thirkell S, Lowe K, Fry LJ, Scotta C, Hope A, Fisher C, Hilton R, Game D, Harden P, Bushell A, Wood K, Lechler RI, Lombardi G (2018) A rapamycin-based GMP-compatible process for the isolation and expansion of regulatory T cells for clinical trials. Mol Ther Methods Clin Dev 8:198–209
MacDonald KG, Hoeppli RE, Huang Q, Gillies J, Luciani DS, Orban PC, Broady R, Levings MK (2016) Alloantigen-specific regulatory T cells generated with a chimeric antigen receptor. J Clin Invest 126:1413–1424
Boardman DA, Philippeos C, Fruhwirth GO, Ibrahim MA, Hannen RF, Cooper D, Marelli-Berg FM, Watt FM, Lechler RI, Maher J, Smyth LA, Lombardi G (2017) Expression of a chimeric antigen receptor specific for donor HLA class I enhances the potency of human regulatory T cells in preventing human skin transplant rejection. Am J Transplant 17:931–943
Tenspolde M, Zimmermann K, Weber LC, Hapke M, Lieber M, Dywicki J, Frenzel A, Hust M, Galla M, Buitrago-Molina LE, Manns MP, Jaeckel E, Hardtke-Wolenski M (2019) Regulatory T cells engineered with a novel insulin-specific chimeric antigen receptor as a candidate immunotherapy for type 1 diabetes. J Autoimmun 103:102289
Chmielewski M, Abken H (2015) TRUCKs: the fourth generation of CARs. Expert Opin Biol Ther 15:1145–1154
Hester J, Schiopu A, Nadig SN, Wood KJ (2012) Low-dose rapamycin treatment increases the ability of human regulatory T cells to inhibit transplant arteriosclerosis in vivo. Am J Transplant 12:2008–2016
Wagner JC, Ronin E, Ho P, Peng Y, Tang Q. 2022. Anti-HLA-A2-CAR Tregs prolong vascularized mouse heterotopic heart allograft survival. Am J Transplant In press
Ratnasothy K, Jacob J, Tung S, Boardman D, Lechler RI, Sanchez-Fueyo A, Martinez-Llordella M, Lombardi G (2019) IL-2 therapy preferentially expands adoptively transferred donor-specific Tregs improving skin allograft survival. Am J Transplant 19:2092–2100
Zhang H, **a Y, Ye Q, Yu F, Zhu W, Li P, Wei Z, Yang Y, Shi Y, Thomson AW, Chen J, Hu X (2018) In vivo expansion of regulatory T cells with IL-2/IL-2 antibody complex protects against transient ischemic stroke. J Neurosci 38:10168–10179
Yshii L, Pasciuto E, Bielefeld P, Mascali L, Lemaitre P, Marino M, Dooley J, Kouser L, Verschoren S, Lagou V, Kemps H, Gervois P, de Boer A, Burton OT, Wahis J, Verhaert J, Tareen SHK, Roca CP, Singh K, Whyte CE, Kerstens A, Callaerts-Vegh Z, Poovathingal S, Prezzemolo T, Wierda K, Dashwood A, **e J, Van Wonterghem E, Creemers E, Aloulou M, Gsell W, Abiega O, Munck S, Vandenbroucke RE, Bronckaers A, Lemmens R, De Strooper B, Van Den Bosch L, Himmelreich U, Fitzsimons CP, Holt MG, Liston A (2022) Astrocyte-targeted gene delivery of interleukin 2 specifically increases brain-resident regulatory T cell numbers and protects against pathological neuroinflammation. Nat Immunol 23:878–891
Peterson LB, Bell CJM, Howlett SK, Pekalski ML, Brady K, Hinton H, Sauter D, Todd JA, Umana P, Ast O, Waldhauer I, Freimoser-Grundschober A, Moessner E, Klein C, Hosse RJ, Wicker LS (2018) A long-lived IL-2 mutein that selectively activates and expands regulatory T cells as a therapy for autoimmune disease. J Autoimmun 95:1–14
Khoryati L, Pham MN, Sherve M, Kumari S, Cook K, Pearson J, Bogdani M, Campbell DJ, Gavin MA. 2020. An IL-2 mutein engineered to promote expansion of regulatory T cells arrests ongoing autoimmunity in mice. Sci Immunol 5
Zhang SR, Piepke M, Chu HX, Broughton BR, Shim R, Wong CH, Lee S, Evans MA, Vinh A, Sakkal S, Arumugam TV, Magnus T, Huber S, Gelderblom M, Drummond GR, Sobey CG, Kim HA. 2018. IL-33 modulates inflammatory brain injury but exacerbates systemic immunosuppression following ischemic stroke. JCI Insight 3
**ao W, Guo S, Chen L, Luo Y (2019) The role of Interleukin-33 in the modulation of splenic T-cell immune responses after experimental ischemic stroke. J Neuroimmunol 333:576970
Na SY, Mracsko E, Liesz A, Hunig T, Veltkamp R (2015) Amplification of regulatory T cells using a CD28 superagonist reduces brain damage after ischemic stroke in mice. Stroke 46:212–220
Schuhmann MK, Kraft P, Stoll G, Lorenz K, Meuth SG, Wiendl H, Nieswandt B, Sparwasser T, Beyersdorf N, Kerkau T, Kleinschnitz C (2015) CD28 superagonist-mediated boost of regulatory T cells increases thrombo-inflammation and ischemic neurodegeneration during the acute phase of experimental stroke. J Cereb Blood Flow Metab 35:6–10
Wang H, Wang Z, Wu Q, Yuan Y, Cao W, Zhang X (2021) Regulatory T cells in ischemic stroke. CNS Neurosci Ther 27:643–651
Komatsu N, Okamoto K, Sawa S, Nakashima T, Oh-hora M, Kodama T, Tanaka S, Bluestone JA, Takayanagi H (2014) Pathogenic conversion of Foxp3+ T cells into TH17 cells in autoimmune arthritis. Nat Med 20:62–68
Hu Y, Zheng Y, Wu Y, Ni B, Shi S (2014) Imbalance between IL-17A-producing cells and regulatory T cells during ischemic stroke. Mediators Inflamm 2014:813045
Li Q, Wang Y, Yu F, Wang YM, Zhang C, Hu C, Wu Z, Xu X, Hu S (2013) Peripheral Th17/Treg imbalance in patients with atherosclerotic cerebral infarction. Int J Clin Exp Pathol 6:1015–1027
Borlongan MC, Kingsbury C, Salazar FE, Toledo ARL, Monroy GR, Sadanandan N, Cozene B, Gonzales-Portillo B, Saft M, Wang ZJ, Moscatello A, Lee JY (2021) IL-2/IL-2R antibody complex enhances Treg-induced neuroprotection by dampening TNF-alpha inflammation in an in vitro stroke model. Neuromolecular Med 23:540–548
Liu X, Hu R, Pei L, Si P, Wang C, Tian X, Wang X, Liu H, Wang B, **a Z, Xu Y, Song B (2020) Regulatory T cell is critical for interleukin-33-mediated neuroprotection against stroke. Exp Neurol 328:113233
**e L, Sun F, Wang J, Mao X, **e L, Yang SH, Su DM, Simpkins JW, Greenberg DA, ** K (2014) mTOR signaling inhibition modulates macrophage/microglia-mediated neuroinflammation and secondary injury via regulatory T cells after focal ischemia. J Immunol 192:6009–6019
Malone K, Diaz Diaz AC, Shearer JA, Moore AC, Waeber C (2021) The effect of fingolimod on regulatory T cells in a mouse model of brain ischaemia. J Neuroinflammation 18:37
Yang H, Zhang A, Zhang Y, Ma S, Wang C (2016) Resveratrol pretreatment protected against cerebral ischemia/reperfusion injury in rats via expansion of T regulatory cells. J Stroke Cerebrovasc Dis 25:1914–1921
Funding
**aoming Hu is supported by a VA grant (I01 BX003651) and a NIH grant (R01NS094573). AWT is supported by NIH grants AI R01 118777, U01 AI136779, and U19 AI 131453. Rimi Hazra is supported by the pilot grants from the Vascular Medicine Institute, the Hemophilia Center of Western Pennsylvania, Vitalant and Winters Foundation of Pittsburgh.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is a contribution to the special issue on: Immunopathology of Stroke – Guest Editors: Arthur Liesz & Tim Magnus
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, M., Thomson, A.W., Yu, F. et al. Regulatory T lymphocytes as a therapy for ischemic stroke. Semin Immunopathol 45, 329–346 (2023). https://doi.org/10.1007/s00281-022-00975-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00281-022-00975-z