Abstract
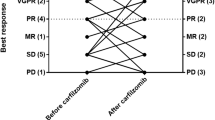
Carfilzomib, lenalidomide, and dexamethasone (KRd) have been approved for the treatment of relapsed and refractory multiple myeloma (RRMM) based on ASPIRE clinical trial. However, its effectiveness and safety profile in real clinical practice should be further assessed. We retrospectively evaluated 130 consecutive RRMM patients treated with KRd between December 2015 and August 2018, in 9 Hematology Departments of Rete Ematologica Pugliese (REP). The overall response rate (ORR) was 79%, with 37% complete response (CR). Treatment with KRd led to an improvement in response regardless of age, refractory disease, and number and type of previous therapies. After a median follow-up of 18 months, median PFS was 24 months and 2y-PFS was 54%. PFS was longer in patients achieving a very good partial response (VGPR) with median PFS of 32.4 months. The relapses after prior autologous transplant (ASCT) positively impact median PFS. Several baseline disease characteristics, such as III ISS scoring or elevated LDH, and prior exposure to lenalidomide were found to negatively impact PFS. Primary refractory or relapsed myeloma patients have been treated with KRd as bridge to ASCT with a great benefit. Thirty-four (83%) reached at least a partial response after KRd and 21 (61%) performed ASCT. In transplanted patients, median PFS was not reached and 2y-PFS was 100%. The treatment discontinuation rate due to adverse events (AEs) was 18%, most commonly for lenalidomide (11%). Overall, in 10% of patients, a KRd dose reduction was necessary at least once (2.5% for carfilzomib and 8% for lenalidomide). The most frequent AE was neutropenia (44%) and anemia (41%). Infections occurred in 14% of patients. Cardiovascular events occurred in 11% of patients. Elderly patients have tolerated therapy very well, without additional side effects compared to younger patients, except for cardiac impairment. Our analysis confirmed that KRd is effective in RRMM patients. It is well tolerated and applicable to the majority of patients outside clinical trials. A longer PFS was shown in patients achieving VGPR, in those lenalidomide naïve and in patients relapsing after previous ASCT. Previous ASCT should not hamper the option for KRd therapy. Accordingly, KRd should be used as bridge regimen to ASCT with remarkable improvement in response and PFS rates. Further clinical studies are needed.



Similar content being viewed by others
References
Kumar SK, Callander NS, Alsina M, Atanackovic D, Biermann JS, Chandler JC, Costello C, Faiman M, Fung HC, Gasparetto C, Godby K, Hofmeister C, Holmberg L, Holstein S, Huff CA, Kassim A, Liedtke M, Martin T, Omel J, Raje N, Reu FJ, Singhal S, Somlo G, Stockerl-Goldstein K, Treon SP, Weber D, Yahalom J, Shead DA Kumar R(2017) Multiple Myeloma, Version 3.2017, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw 15:230–269
Stewart AK, Rajkumar SV, Dimopoulos MA, Masszi T, Špička I, Oriol A, Hájek R, Rosiñol L, Siegel DS, Mihaylov GG, Goranova-Marinova V, Rajnics P, Suvorov A, Niesvizky R, Jakubowiak AJ, San-Miguel JF, Ludwig H, Wang M, Maisnar V, Minarik J, Bensinger WI, Mateos MV, Ben-Yehuda D, Kukreti V, Zojwalla N, Tonda ME, Yang X, **ng B, Moreau P, Palumbo A, Investigators ASPIRE (2015) Carfilzomib, lenalidomide and dexamethasone for relapsed multiple myeloma. N Engl J Med 372:142–152
Dimopoulos MA, Moreau P, Palumbo A, Joshua D, Pour L, Hájek R, Facon T, Ludwig H, Oriol A, Goldschmidt H, Rosiñol L, Straub J, Suvorov A, Araujo C, Rimashevskaya E, Pika T, Gaidano G, Weisel K, Goranova-Marinova V, Schwarer A, Minuk L, Masszi T, Karamanesht I, Offidani M, Hungria V, Spencer A, Orlowski RZ, Gillenwater HH, Mohamed N, Feng S, Chng WJ, Investigators ENDEAVOR (2016) Carfilzomib and dexamethasone versus bortezomib and dexamethasone for patients with relapsed or refractory multiple myeloma (ENDEAVOR): a randomised,phase 3, open-label, multicentre study. Lancet Oncol 17:27–38
Durie BG, Harousseau JL, Miguel JS, Bladé J, Barlogie B, Anderson K, Gertz M, Dimopoulos M, Westin J, Sonneveld P, Ludwig H, Gahrton G, Beksac M, Crowley J, Belch A, Boccadaro M, Cavo M, Turesson I, Joshua D, Vesole D, Kyle R, Alexanian R, Tricot G, Attal M, Merlini G, Powles R, Richardson P, Shimizu K, Tosi P, Morgan G, Rajkumar SV, International Myeloma Working Group (2006) International uniform response criteria for multiple myeloma. Leukemia 20:1467–1473
Kaplan G, Meier P (1958) Non-parametric estimations from incomplete observations. J Am Stat Assoc 53:457–481
Mateos MV, Goldschimidt H, San-Miguel J, Mikhael J, DeCosta L, Zhou L, Obreja M, Blaedel J, Szabo Z, Leleu X (2018) Carfilzomib in relapsed or refractory multiple myeloma patients with early or late relapse following prior therapy: a subgroup analysis of the randomized phase 3 ASPIRE and ENDEAVOR trials. Hematol Oncol 36(2):463–470
Dimopoulos M, Wang M, Maisnar V, Minarik J, Bensinger W, Mateos MV, Obreja M, Blaedel J, Moreau P (2018) Response and progression-free survival according to planned treatment duration in patients with relapsed multiple myeloma treated with carfilzomib, lenalidomide, and dexamethasone (KRd) versus lenalidomide and dexamethasone (Rd) in the phase III ASPIRE study. J Hematol Oncol 11(1):49 1-9
Dimopoulos MA, Stewart AK, Masszi T, Špička I, Oriol A, Hájek R, Rosiñol L, Siegel D, Mihaylov GG, Goranova-Marinova V, Rajnics P, Suvorov A, Niesvizky R, Jakubowiak A, San-Miguel J, Ludwig H, Palumbo A, Obreja M, Aggarwal S, Moreau P (2017a) Carfilzomib, lenalidomide, and dexamethasone in patients with relapsed multiple myeloma categorised by age: secondary analysis from the phase 3 ASPIRE study. Br J Haematol 177(3):404–413
Hari P, Mateos MV, Abonour R, Knop S, Bensinger W, Ludwig H, Song K, Hajek R, Moreau P, Siegel DS, Feng S, Obreja M, Aggarwal SK, Iskander K, Goldschmidt H (2017) Efficacy and safety of carfilzomib regimens in multiple myeloma patients relapsing after autologous stem cell transplant: ASPIRE and ENDEAVOR outcomes. Leukemia 31(12):2630–2641
Dimopoulos MA, Stewart AK, Masszi T, Špička I, Oriol A, Hájek R, Rosiñol L, Mihaylov GG, Goranova-Marinova V, Rajnics P, Suvorov A, Niesvizky R, Jakubowiak A, San-Miguel J, Ludwig H, Ro S, Aggarwal S, Moreau P, Palumbo A (2017) Carfilzomib-lenalidomide-dexamethasone vs lenalidomide-dexamethasone in relapsed multiple myeloma by previous treatment. Blood Cancer J 7(4):e554
Danhof S, Schreder M, Rasche L, Strifler S, Einsele H, Knop S (2016) 'Real-life' experience of preapproval carfilzomib-based therapy in myeloma - analysis of cardiac toxicity and predisposing factors. Eur J Haematol 97(1):25–32
Chen CC, Parikh K, Abouzaid S, Purnomo L, McGuiness CB, Hussein M, Wade RL (2017) Real-world treatment patterns, time to next treatment, and economic outcomes in relapsed or refractory multiple myeloma patients treated with pomalidomide or carfilzomib. J Manag Care Spec Pharm 23(2):236–246
Conticello C, Romano A, Del Fabro V, Martino EA, Calafiore V, Sapienza G, Leotta V, Parisi MS, Markovic U, Garibaldi B, Leotta S, Cotzia E, Innao V, Mannina D, Neri S, Musso M, Scalone R, Cangialosi C, Acquaviva F, Cardinale G, Merenda A, Maugeri C, Uccello G, Poidomani M, Longo G, Carlisi M, Tibullo D, Di Raimondo F (2019) Feasibility, tolerability and efficacy of carfilzomib in combination with lenalidomide and dexamethasone in relapsed refractory myeloma patients: a retrospective real-life survey of the Sicilian myeloma network. J Clin Med 19(6):8
Wang M, Bensinger W, Martin T, Alsina M, Siegel DSD, Gabrail P, Hari NY, Singhal S, Vescio RA, Assouline SE, Kunkel AL, Vallone M, Wong A, Niesvizky R (2011) Interim results from PX-171-006, a phase (Ph) II multicenter dose-expansion study of carfilzomib (CFZ), lenalidomide (LEN), and low-dose dexamethasone (loDex) in relapsed and/or refractory multiple myeloma (R/R MM). J Clin Oncol 29(15_suppl, Abstract 8025)
Biran N, Di Capua Siegel DS, Berdeja JG, Raje NS, Cornell RF, Alsina M, Kovacsovics T, Fang B, Kimball A, Landgren CO (2018) Weekly carfilzomib, lenalidomide and dexamethasone (KRD) in relapsed or refractory multiple myeloma (RRMM): a phase 1b study. The European Hematology Association, EHA Library, PS1297
Richez V, Guidez S, Azais I, Durand G, Javaugue V, Brigaud A, Plasse F, Diolez J, Machet A, Moya N, Gruchet C, Bobin A, Levy A, Sabirou F, Bonnin A, Dieval C, Primault S, Barrier J, Fleck E, Daras C, Princet I, Bauwens D, Fouquet G, Bridoux F, Avet Loiseau H, Leleu X (2017) Carfilzomib weekly 20/56mg/m2, lenalidomide and dexamethasone for early relapsed refractory multiple myeloma. Blood 130:1843
Jakubowiak A, Raje N, Vij R, Reece D, Berdeja J, Vesole D, Jagannath S, Cole C, Faham M, Nam J, Stephens L, Severson R, Revethis A, Wolfe B, Rosebeck S, Gurbuxani S, Rosenbaum C, Jasielec J, Dytfeld D (2016) Improved efficacy after incorporating autologous stem cell transplant (ASCT) into KRD treatment with carfilzomib (CFZ), lenalidomide (LEN), and dexamethasone (DEX) in newly diagnosed multiple myeloma. Presented at 21st European Hematology Association Annual Meeting, EHA Library, 10 June 2016
Acknowledgments
The authors would like to thank A. La Torre, M. Lecciso, C. Plati, and F. Clemente who contributed to the collection of clinical data; medical doctors of REP and nurses; and all the patients, families, caregivers, and AIL staff who contributed to this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mele, A., Prete, E., De Risi, C. et al. Carfilzomib, lenalidomide, and dexamethasone in relapsed/refractory multiple myeloma patients: the real-life experience of Rete Ematologica Pugliese (REP). Ann Hematol 100, 429–436 (2021). https://doi.org/10.1007/s00277-020-04329-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-020-04329-3