Abstract
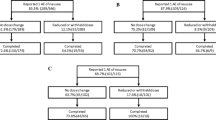
Patients receiving vinca alkaloids for hematological malignancies frequently experience constipation that is unresponsive to laxatives. Research on treatment of vinca alkaloid-induced constipation is limited. This study aimed to determine whether the chloride channel activator lubiprostone ameliorates vinca alkaloid–induced constipation in patients with hematological malignancies. In this retrospective cohort study, vinca alkaloid–induced constipation (grade ≥ 3 using the Common Terminology Criteria for Adverse Events) was investigated in patients treated for hematological malignancies between July 2014 and June 2019 who had already been prescribed osmotic laxatives and additionally received either a stimulant laxative or lubiprostone. Univariate and multivariate analyses were performed to identify the risk factors for persistent constipation after introduction of the second laxative. A propensity score model was used to match 67 patients taking a stimulant laxative and 67 treated with lubiprostone, and the occurrence of intractable constipation was compared between groups. Overall, 203 patients were included, among whom 50 (25%) had constipation. On multivariate analysis, body mass index, opioid use, and addition of lubiprostone were independently associated with constipation. Patients treated with lubiprostone were significantly less likely to experience intractable constipation than did those treated with stimulant laxatives (10% vs. 34%, P = 0.002). Moreover, post-constipation diarrhea was significantly less frequent among patients treated with lubiprostone (42% vs. 63%, P = 0.024). Lubiprostone was more effective than stimulant laxatives at treating vinca alkaloid–induced intractable constipation in patients with hematological malignancies, and its use could enable safe vinca alkaloid chemotherapy.
Similar content being viewed by others
Data availability
The dataset generated and analyzed during the current study is available from the corresponding author on reasonable request.
References
Kantarjian HM, Walters RS, Keating MJ, Smith TL, O'Brien S, Estey EH, Huh YO, Spinolo J, Dicke K, Barlogie B, McCredie KB, Freireich EJ (1990) Results of the vincristine, doxorubicin, and dexamethasone regimen in adults with standard- and high-risk acute lymphocytic leukemia. J Clin Oncol 8:994–1004. https://doi.org/10.1200/JCO.1990.8.6.994
Geisler S, Doan RA, Strickland A, Huang X, Milbrandt J, DiAntonio A (2016) Prevention of vincristine-induced peripheral neuropathy by genetic deletion of SARM1 in mice. Brain 139:3092–3108. https://doi.org/10.1093/brain/aww251
Haim N, Epelbaum R, Ben-Shahar M, Yarnitsky D, Simri W, Robinson E (1994) Full dose vincristine (without 2-mg dose limit) in the treatment of lymphomas. Cancer 73:2515–2519. https://doi.org/10.1002/1097-0142(19940515)73:10<2515::aid-cncr2820731011>3.0.co;2-g
Yamagishi A, Morita T, Miyashita M, Kimura F (2009) Symptom prevalence and longitudinal follow-up in cancer outpatients receiving chemotherapy. J Pain Symptom Manag 37:823–830. https://doi.org/10.1016/j.jpainsymman.2008.04.015
Toghill PJ, Burke JD (1970) Death from paralytic ileus following vincristine therapy. Postgrad Med J 46:330–331. https://doi.org/10.1136/pgmj.46.535.330
Anderson H, Scarffe JH, Lambert M, Smith DB, Chan CC, Chadwick G, McMahon A, Chang J, Crowther D, Swindell R (1987) VAD chemotherapy--toxicity and efficacy--in patients with multiple myeloma and other lymphoid malignancies. Hematol Oncol 5:213–222. https://doi.org/10.1002/hon.2900050308
McQuade RM, Stojanovska V, Abalo R, Bornstein JC, Nurgali K (2016) Chemotherapy-induced constipation and diarrhea: pathophysiology, current and emerging treatments. Front Pharmacol 7:414. https://doi.org/10.3389/fphar.2016.00414
Yasu T, Ohno N, Kawamata T, Kurokawa Y (2016) Vincristine-induced paralytic ileus during induction therapy of treatment protocols for acute lymphoblastic leukemia in adult patients. Int J Clin Pharmacol Ther 54:471–473. https://doi.org/10.5414/CP202584
Hayashi H, Suzuki A, Ohata K, Ishihara M, Kubota Y, Kobayashi R, Shibata Y, Nakamura H, Nakamura N, Kitagawa J, Tsurumi H, Shimizu M, Itoh Y (2017) Control of constipation in patients receiving CHOP or CHOP-like chemotherapy regimens for non-Hodgkin’s lymphoma. Biol Pharm Bull 40:698–702. https://doi.org/10.1248/bpb.b16-01001
Mearin F, Lacy BE, Chang L, Chey WD, Lembo AJ, Simren M, Spiller R (2016) Bowel disorders. Gastroenterology 150:1393–1407. https://doi.org/10.1053/j.gastro.2016.02.031
Legha SS (1986) Vincristine neurotoxicity. Pathophysiology and management. Med Toxicol 1:421–427. https://doi.org/10.1007/bf03259853
Basch E, Iasonos A, McDonough T, Barz A, Culkin A, Kris MG, Scher HI, Schrag D (2006) Patient versus clinician symptom reporting using the National Cancer Institute common terminology criteria for adverse events: results of a questionnaire-based study. Lancet Oncol l7:903–909. https://doi.org/10.1016/S1470-2045(06)70910-X
Ikehara O (1992) Vincristine-induced paralytic ileus: role of fiberoptic colonoscopy and prostaglandin F2 alpha. Am J Gastroenterol 87:207–210
Johanson JF, Ueno R (2007) Lubiprostone, a locally acting chloride channel activator, in adult patients with chronic constipation: a double-blind, placebo-controlled, dose-ranging study to evaluate efficacy and safety. Aliment Pharmacol Ther 25:1351–1361. https://doi.org/10.1111/j.1365-2036.2007.03320.x
Noergaard M, Traerup Andersen J, Jimenez-Solem E, Bring Christensen M (2019) Long term treatment with stimulant laxatives - clinical evidence for effectiveness and safety? Scand J Gastroenterol 54:27–34. https://doi.org/10.1080/00365521.2018.1563806
Kurniawan I, Simadibrata M (2011) Management of chronic constipation in the elderly. Acta Med Indones 43:195–205
Abernethy AP, Wheeler JL, Zafar SY (2009) Detailing of gastrointestinal symptoms in cancer patients with advanced disease: new methodologies, new insights, and a proposed approach. Curr Opin Support Palliat Care 3:41–49. https://doi.org/10.1097/SPC.0b013e32832531ce
Abramowitz L, Beziaud N, Labreze L, Giardina V, Caussé C, Chuberre B, Allaert FA, Perrot S (2013) Prevalence and impact of constipation and bowel dysfunction induced by strong opioids: a cross-sectional survey of 520 patients with cancer pain: DYONISOS study. J Med Econ 16:1423–1433. https://doi.org/10.3111/13696998.2013.851082
Johanson JF, Sonnenberg A, Koch TR (1989) Clinical epidemiology of chronic constipation. J Clin Gastroenterol 11:525–536. https://doi.org/10.1097/00004836-198910000-00008
Şendir M, Büyükıylmaz F, Aştı T, Gürpınar Ş, Yazgan İ (2012) Postoperative constipation risk assessment in Turkish orthopedic patients. Gastroenterol Nurs 35:106–113. https://doi.org/10.1097/SGA.0b013e31824d2945
Dore MP, Pes GM, Bibbò S, Tedde P, Bassotti G (2018) Constipation in the elderly from northern Sardinia is positively associated with depression, malnutrition and female gender. Scand J Gastroenterol 53:797–802. https://doi.org/10.1080/00365521.2018.1473485
Huang L, Jiang H, Zhu M, Wang B, Tong M, Li H, Lin MB, Li L (2017) Prevalence and risk factors of chronic constipation among women aged 50 years and older in Shanghai, China. Med Sci Monit 23:2660–2667. https://doi.org/10.12659/msm.904040
Wald A (2016) Constipation: advances in diagnosis and treatment. JAMA 315:185–191. https://doi.org/10.1001/jama.2015.16994
Donald IP, Smith RG, Cruikshank JG, Elton RA, Stoddart ME (1985) A study of constipation in the elderly living at home. Gerontology 31:112–118. https://doi.org/10.1159/000212689
Cardin F, Minicuci N, Droghi AT, Inelmen EM, Sergi G, Terranova O (2010) Constipation in the acutely hospitalized older patients. Arch Gerontol Geriatr 50:277–281. https://doi.org/10.1016/j.archger.2009.04.007
Ishihara M, Ikesue H, Matsunaga H, Suemaru K, Kitaichi K, Suetsugu K, Oishi R, Sendo T, Araki H, Itoh Y, Japanese Study Group for the Relief of Opioid-induced Gastrointestinal Dysfunction (2012) A multi-institutional study analyzing effect of prophylactic medication for prevention of opioid-induced gastrointestinal dysfunction. Clin J Pain 28:373–381. https://doi.org/10.1097/AJP.0b013e318237d626
Austin PC (2011) Optimal caliper widths for propensity-score matching when estimating differences in means and differences in proportions in observational studies. Pharm Stat 10:150–161. https://doi.org/10.1002/pst.433
Austin PC (2009) Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat Med 28:3083–3107. https://doi.org/10.1002/sim.3697
Wickham RJ (2017) Managing constipation in adults with cancer. J Adv Pract Oncol 8:149–161
Glia A, Lindberg G (1997) Quality of life in patients with different types of functional constipation. Scand J Gastroenterol 32:1083–1089. https://doi.org/10.3109/00365529709002985
Koloski NA, Jones M, Wai R, Gill RS, Byles J, Talley NJ (2013) Impact of persistent constipation on health-related quality of life and mortality in older community-dwelling women. Am J Gastroenterol 108:1152–1158. https://doi.org/10.1038/ajg.2013.137
Sumida K, Molnar MZ, Potukuchi PK, Thomas F, Lu JL, Yamagata K, Kalantar-Zadeh K, Kovesdy CP (2018) Constipation and risk of death and cardiovascular events. Atherosclerosis 281:114–120. https://doi.org/10.1016/j.atherosclerosis.2018.12.021
Geisler S, Huang SX, Strickland A, Doan RA, Summers DW, Mao X, Park J, DiAntonio A, Milbrandt J (2019) Gene therapy targeting SARM1 blocks pathological axon degeneration in mice. J Exp Med 216:294–303. https://doi.org/10.1084/jem.20181040
Sharma RK (1988) Vincristine and gastrointestinal transit. Gastroenterology 95:1435–1436
Esmadi M, Ahmad D, Hewlett A (2019) Efficacy of naldemedine for the treatment of opioid-induced constipation: a meta-analysis. J Gastrointest Liver Dis 28:41–46. https://doi.org/10.15403/jgld.2014.1121.281.any
Twycross R, Sykes N, Mihalyo M, Wilcock A (2012) Stimulant laxatives and opioid-induced constipation. J Pain Symptom Manag 43:306–313. https://doi.org/10.1016/j.jpainsymman.2011.12.002
Dosh SA (2002) Evaluation and treatment of constipation. J Fam Pract 51:555–559
Author information
Authors and Affiliations
Contributions
Material preparation, data collection, and analysis were performed by Kei Kawada, Tsuyoshi Ohta, and Hitoshi Fukuda. The first draft of the manuscript was written by Kei Kawada, and all the authors commented on previous versions of the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
The Kochi Health Sciences Center Institutional Review Board approved the study. The procedures used in this study adhere to the tenets of the Declaration of Helsinki. Informed consent was waived in view of the retrospective nature of the study. Confidentiality and anonymity of patient data was maintained throughout.
Consent to participate
The requirement for informed consent was waived in view of the retrospective nature of the study.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kawada, K., Ohta, T., Fukuda, H. et al. Effect of lubiprostone on vinca alkaloid–induced constipation in patients with hematological malignancies: a propensity score–matched analysis. Ann Hematol 99, 2429–2436 (2020). https://doi.org/10.1007/s00277-020-04222-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-020-04222-z