Abstract
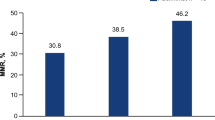
We assessed the efficacy and toxicity of etoposide, methylprednisolone, high-dose cytarabine, and oxaliplatin (ESHAOx) combination chemotherapy in patients with refractory or relapsed Hodgkin’s lymphoma (HL). This was an open-label, non-randomized, multi-center phase II study. The ESHAOx regimen consisted of intravenous (i.v.) etoposide 40 mg/m2 on days 1 to 4, i.v. methylprednisolone 500 mg on days 1 to 5, i.v. cytarabine 2 g/m2 on day 5, and i.v. oxaliplatin 130 mg/m2 on day 1. Cycles (up to six) were repeated every 3 weeks. In an effort to identify prognostic markers, the serum levels of cytokines including tumor necrosis factor-α (TNF-α), C-reactive protein (CRP), and vascular endothelial growth factor (VEGF) were measured at the time of study entry. A total of 37 patients were enrolled, and 36 were available for evaluation of tumor response. The overall response rate was 72.2% (26/36) (complete response, 33.3% [12/36]; partial response, 38.9% [14/36]). The median time to progression was 34.9 months (95% confidence interval, 23.1–46.7 months). The most common grade 3 or 4 hematological adverse events were neutropenia (16/37, 43.2%), followed by thrombocytopenia (10/37, 27.0%). Grade 3 or 4 non-hematological adverse events were nausea (3/37, 8.1%), anorexia (2/37, 5.4%), mucositis (1/37, 2.7%), and skin rash (1/37, 2.7%). There were no treatment-related deaths. High levels of TNF-α and CRP were significantly associated with poorer overall survival (p = 0.00005 for TNF-α, p = 0.0004 for CRP, respectively). The ESHAOx regimen exhibited antitumor activity and an acceptable safety profile in patients with refractory or relapsed HL. Trial Registration: ClinicalTrials.gov. Registered February 21, 2011, https://clinicaltrials.gov/ct2/show/NCT01300156


Similar content being viewed by others
References
Chisesi T, Bellei M, Luminari S, Montanini A, Marcheselli L, Levis A, Gobbi P, Vitolo U, Stelitano C, Pavone V (2011) Long-term follow-up analysis of HD9601 trial comparing ABVD versus Stanford V versus MOPP/EBV/CAD in patients with newly diagnosed advanced-stage Hodgkin's lymphoma: a study from the Intergruppo Italiano Linfomi. J Clin Oncol 29(32):4227–4233
Linch D, Goldstone A, McMillan A, Chopra R, Hudson GV, Winfield D, Hancock B, Moir D, Milligan D (1993) Dose intensification with autologous bone-marrow transplantation in relapsed and resistant Hodgkin's disease: results of a BNLI randomised trial. Lancet 341(8852):1051–1054
Schmitz N, Pfistner B, Sextro M, Sieber M, Carella AM, Haenel M, Boissevain F, Zschaber R, Müller P, Kirchner H (2002) Aggressive conventional chemotherapy compared with high-dose chemotherapy with autologous haemopoietic stem-cell transplantation for relapsed chemosensitive Hodgkin's disease: a randomised trial. Lancet 359(9323):2065–2071
Mendler JH, Friedberg JW (2009) Salvage therapy in Hodgkin's lymphoma. Oncologist 14(4):425–432
Bartlett N, Niedzwiecki D, Johnson J, Friedberg J, Johnson K, Van Besien K, Zelenetz A, Cheson B, Canellos G (2007) Gemcitabine, vinorelbine, and pegylated liposomal doxorubicin (GVD), a salvage regimen in relapsed Hodgkin's lymphoma: CALGB 59804. Ann Oncol 18(6):1071–1079
Crump M, Kuruvilla J, Couban S, MacDonald DA, Kukreti V, Kouroukis CT, Rubinger M, Buckstein R, Imrie KR, Federico M (2014) Randomized comparison of gemcitabine, dexamethasone, and cisplatin versus dexamethasone, cytarabine, and cisplatin chemotherapy before autologous stem-cell transplantation for relapsed and refractory aggressive lymphomas: NCIC-CTG LY. 12. J Clin Oncol 32(31):3490–3496
Gopal AK, Press OW, Shustov AR, Petersdorf SH, Gooley TA, Daniels JT, Garrison MA, Gjerset GF, Lonergan M, Murphy AE (2010) Efficacy and safety of gemcitabine, carboplatin, dexamethasone, and rituximab in patients with relapsed/refractory lymphoma: a prospective multi-center phase II study by the Puget Sound Oncology Consortium. Leuk Lymphoma 51(8):1523–1529
Santoro A, Magagnoli M, Spina M, Pinotti G, Siracusano L, Michieli M, Nozza A, Sarina B, Morenghi E, Castagna L (2007) Ifosfamide, gemcitabine, and vinorelbine: a new induction regimen for refractory and relapsed Hodgkin’s lymphoma. Haematologica 92(1):35–41
Moskowitz AJ, Hamlin PA Jr, Perales M-A, Gerecitano J, Horwitz SM, Matasar MJ, Noy A, Palomba ML, Portlock CS, Straus DJ (2012) Phase II study of bendamustine in relapsed and refractory Hodgkin lymphoma. J Clin Oncol 31(4):456–460
Fehniger TA, Larson S, Trinkaus K, Siegel MJ, Cashen AF, Blum KA, Fenske TS, Hurd DD, Goy A, Schneider SE (2011) A phase 2 multicenter study of lenalidomide in relapsed or refractory classical Hodgkin lymphoma. Blood 118(19):5119–5125
Johnston PB, Inwards DJ, Colgan JP, Laplant BR, Kabat BF, Habermann TM, Micallef IN, Porrata LF, Ansell SM, Reeder CB (2010) A phase II trial of the oral mTOR inhibitor everolimus in relapsed Hodgkin lymphoma. Am J Hematol 85(5):320–324
Younes A, Bartlett NL, Leonard JP, Kennedy DA, Lynch CM, Sievers EL, Forero-Torres A (2010) Brentuximab vedotin (SGN-35) for relapsed CD30-positive lymphomas. N Engl J Med 363(19):1812–1821
Ansell SM, Lesokhin AM, Borrello I, Halwani A, Scott EC, Gutierrez M, Schuster SJ, Millenson MM, Cattry D, Freeman GJ (2015) PD-1 blockade with nivolumab in relapsed or refractory Hodgkin's lymphoma. N Engl J Med 372(4):311–319
Younes A, Santoro A, Shipp M, Zinzani PL, Timmerman JM, Ansell S, Armand P, Fanale M, Ratanatharathorn V, Kuruvilla J (2016) Nivolumab for classical Hodgkin’s lymphoma after failure of both autologous stem-cell transplantation and brentuximab vedotin: a multicentre, multicohort, single-arm phase 2 trial. Lancet Oncol 17(9):1283–1294
Hartmann JT, Lipp H-P (2003) Toxicity of platinum compounds. Expert Opin Pharmacother 4(6):889–901
Alinari L, Musuraca G, Tani M, Stefoni V, Gabriele A, Marchi E, Fina M, De Vivo A, Pileri S, Baccarani M (2005) Value of oxaliplatin treatment in heavily pretreated patients with non-Hodgkin’s lymphoma. Leuk Lymphoma 46(10):1437–1440
Machover D, Delmas-Marsalet B, Misra S, Gumus Y, Goldschmidt E, Schilf A, Frenoy N, Emile J-F, Debuire B, Guettier C (2001) Dexamethasone, high-dose cytarabine, and oxaliplatin (DHAOx) as salvage treatment for patients with initially refractory or relapsed non-Hodgkin's lymphoma. Ann Oncol 12(10):1439–1443
Sym SJ, Lee DH, Kang HJ, Nam SH, Kim HY, Kim SJ, Eom HS, Kim WS, Suh C (2009) A multicenter phase II trial of etoposide, methylprednisolone, high-dose cytarabine, and oxaliplatin for patients with primary refractory/relapsed aggressive non-Hodgkin’s lymphoma. Cancer Chemother Pharmacol 64(1):27–33
Marri PR, Hodge LS, Maurer MJ, Ziesmer SC, Slager SL, Habermann TM, Link BK, Cerhan JR, Novak AJ, Ansell SM (2013) Prognostic significance of pretreatment serum cytokines in classical Hodgkin lymphoma. Clin Cancer Res
Health UDo, Services H (2009) Common terminology criteria for adverse events (CTCAE) version 4.0. National Institutes of Health, National Cancer Institute 4 (03)
Cheson BD, Pfistner B, Juweid ME, Gascoyne RD, Specht L, Horning SJ, Coiffier B, Fisher RI, Hagenbeek A, Zucca E (2007) Revised response criteria for malignant lymphoma. J Clin Oncol 25(5):579–586
Simon R (1989) Optimal two-stage designs for phase II clinical trials. Control Clin Trials 10(1):1–10
Contal C, O'Quigley J (1999) An application of changepoint methods in studying the effect of age on survival in breast cancer. Comput Stat Data Anal 30(3):253–270
Aparicio J, Segura A, Garcera S, Oltra A, Santaballa A, Yuste A, Pastor M (1999) ESHAP is an active regimen for relapsing Hodgkin’s disease. Ann Oncol 10(5):593–595
Labrador J, Cabrero-Calvo M, Pérez-López E, Mateos MV, Vázquez L, Caballero MD, García-Sanz R (2014) ESHAP as salvage therapy for relapsed or refractory Hodgkin’s lymphoma. Ann Hematol 93(10):1745–1753
Oak E, Bartlett NL (2016) A safety evaluation of brentuximab vedotin for the treatment of Hodgkin lymphoma. Expert Opin Drug Saf 15(6):875–882
Tixier F, Ranchon F, Iltis A, Vantard N, Schwiertz V, Bachy E, Bouafia-Sauvy F, Sarkozy C, Tournamille J, Gyan E (2017) Comparative toxicities of 3 platinum-containing chemotherapy regimens in relapsed/refractory lymphoma patients. Hematol Oncol 35(4):584–590
Rautert R, Schinköthe T, Franklin J, Weihrauch M, Böll B, Pogge E, Bredenfeld H, Engert A, Diehl V, Re D (2008) Elevated pretreatment interleukin-10 serum level is an International Prognostic Score (IPS)-independent risk factor for early treatment failure in advanced stage Hodgkin lymphoma. Leuk Lymphoma 49(11):2091–2098
Seymour J, Talpaz M, Hagemeister F, Cabanillas F, Kurzrock R (1997) Clinical correlates of elevated serum levels of interleukin-6 in patients with untreated Hodgkin’s disease. Am J Med 102(1):21–28
Skinnider BF, Mak TW (2002) The role of cytokines in classical Hodgkin lymphoma. Blood 99(12):4283–4297
Acknowledgments
We thank Professor Sun-Young Kim (M.P.H, Ph.D, Biostatistics, Environmental Statistics and Epidemiology, National Cancer Center, South Korea) for statistical support and Dr. JongHeon Jung (M.D., Center for Hematologic Malignancy, National Cancer Center, South Korea) for the valuable review of the manuscript.
Funding
This study was supported by the Grants of the National Cancer Center, Goyang, Korea (NCC 1410210). The role of the funding was the design of the study and collection, analysis, and interpretation of data and in writing. In this study, oxaliplatin was provided by Boryung Pharmaceutical (South Korea).
Author information
Authors and Affiliations
Contributions
Conception and design: Y Won, H Lee, H Eom, K Kim. Provision of study materials and patients: H Lee, H Eom, JS Kim, C Suh, DH Yoon, JY Hong, HJ Kang, JH Lee, WS Kim, SJ Kim, W Lee, MH Chang, YR Do, JH Yi, I Kim, J Won, K Kim, SY Oh, J Jo. Collection and assembly of data: Y Won, H Lee, H Eom. Data analysis and interpretation: Y Won, H Lee, H Eom. Manuscript writing: Y Won, H Lee, H Eom. Final approval of manuscript: All authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval and consent to participate
All patients provided written informed consent before enrollment and the study was approved by our Institutional Review Board.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Won, YW., Lee, H., Eom, HS. et al. A phase II study of etoposide, methylprednisolone, high-dose cytarabine, and oxaliplatin (ESHAOx) for patients with refractory or relapsed Hodgkin’s lymphoma. Ann Hematol 99, 255–264 (2020). https://doi.org/10.1007/s00277-019-03891-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-019-03891-9