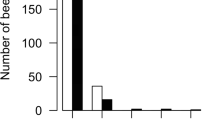
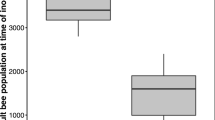
Abstract
The size of animal groups has profound effects on individual and collective behavior, particularly in social insect colonies. Larger colonies are predicted to be more complex with more specialization among members. However, the empirical support of this theoretical expectation is limited. Hygienic behavior of honey bees is a complex cooperative behavior of workers detecting, uncap**, and removing unhealthy brood. It is an important defense against brood diseases, including the ectoparasitic mite Varroa destructor. We support the prediction that hygienic behavior increases with group size using a simulation model. To also test this prediction empirically, we performed five experiments, to compare the hygienic performance of small and large honey bee groups at four different scales, roughly representing four orders of magnitude. Hygienic performance qualitatively increased across the different scales, but different methodologies limit quantitative comparisons across experiments. Within experiments, group size was also positively related to hygienic behavior. The strongest effects of group size were measured in groups that were smaller than what honey bees adopt under natural conditions. The group-size effect on hygienic performance decreased with increasing scale and at the full colony scale, group size was unrelated to hygienic assay scores. Therefore, colony size is unlikely to confound the hygienic evaluation of colonies in apicultural practice although we demonstrate clear effects of group size on hygienic behavior. Direct observations of individual behavior that were performed in two small scale experiments did not support our prediction of increased individual specialization in larger groups. Thus, our study supports the notion of performance benefits of larger groups in the context of social immunity, although the mechanisms of how group size enhances hygienic behavior remain to be investigated further.
Significance Statement
Social insects owe their ecological success partly to their efficient division of labor and behavioral specialization of colony members. Empirical support for the theoretical argument that group performance increases with group size is insufficient. Hygienic behavior is an important defense against brood pests and diseases that threaten honey bee health. Yet, it has not been investigated with respect to group size. Here, we analyze a simulation model, demonstrating theoretically that group size is predicted to increase hygienic behavior. We then provide experimental support for this prediction across a range of group sizes, though results of some experiments are equivocal and sample sizes are limited, constraining our empirical conclusions. In small groups, we find support for the theoretical idea that hygienic performance increases with group size, but our study also indicates that this effect is not very important under apiculturally relevant group size conditions. We find no support for higher individual specialization in larger groups. Furthermore, our study indicates that beekeepers can disregard deviations in colony size when assessing their stock for hygienic behavior.








Similar content being viewed by others
Data availability
All data of this study are included in this published article and its supplementary information files.
References
Arathi HS, Spivak M (2001) Influence of colony genotypic composition on the performance of hygienic behaviour in the honeybee, Apis mellifera L. Anim Behav 62:57–66. https://doi.org/10.1006/anbe.2000.1731
Arathi H, Burns I, Spivak M (2000) Ethology of hygienic behaviour in the honey bee Apis mellifera L. (Hymenoptera: Apidae): behavioural repertoire of hygienic bees. Ethology 106:365–379. https://doi.org/10.1046/j.1439-0310.2000.00556.x
Barrs KR, Ani MO, Eversman KK et al (2021) Time-accuracy trade-off and task partitioning of hygienic behavior among honey bee (Apis mellifera) workers. Behav Ecol Sociobiol 75:12. https://doi.org/10.1007/s00265-020-02940-y
Beshers SN, Fewell JH (2001) Models of division of labor in social insects. Annu Rev Entomol 46:413–440. https://doi.org/10.1146/annurev.ento.46.1.413
Bond CF, Titus LJ (1983) Social facilitation: A meta-analysis of 241 studies. Psychol Bull 94:265. https://doi.org/10.1037/0033-2909.94.2.265
Boyce MS, Perrins C (1987) Optimizing great tit clutch size in a fluctuating environment. Ecology 68:142–153. https://doi.org/10.2307/1938814
Calderone NW (2012) Insect pollinated crops, insect pollinators and US agriculture: trend analysis of aggregate data for the period 1992–2009. PLoS ONE 7:e37235. https://doi.org/10.1371/journal.pone.0037235
Delaplane KS, van der Steen J, Guzman-Novoa E (2013) Standard methods for estimating strength parameters of Apis mellifera colonies. J Apicult Res 52:1–12. https://doi.org/10.3896/ibra/1.52.1.03
Dornhaus A, Powell S, Bengston S (2012) Group size and its effects on collective organization. Annu Rev Entomol 57:123–141. https://doi.org/10.1146/annurev-ento-120710-100604
Eckert CD, Winston ML, Ydenberg RC (1994) The relationship between population size, amount of brood, and individual foraging behaviour in the honey bee, Apis mellifera L. Oecologia 97:248–255. https://doi.org/10.1007/BF00323157
Erez T, Bonda E, Kahanov P et al (2022) Multiple benefits of breeding honey bees for hygienic behavior. J Invertebr Pathol 193:107788. https://doi.org/10.1016/j.jip.2022.107788
Ferguson-Gow H, Sumner S, Bourke AFG, Jones KE (2014) Colony size predicts division of labour in attine ants. Proc R Soc Ser B-Bio 281:20141411. https://doi.org/10.1098/rspb.2014.1411
Fuchs S, Moritz RFA (1999) Evolution of extreme polyandry in the honeybee Apis mellifera L. Behav Ecol Sociobiol 45:269–275. https://doi.org/10.1007/s002650050561
Gautrais J, Theraulaz G, Deneubourg J-L, Anderson C (2002) Emergent polyethism as a consequence of increased colony size in insect societies. J Theor Biol 215:363–373. https://doi.org/10.1006/jtbi.2001.2506
Gavrilets S (2010) Rapid transition towards the division of labor via evolution of developmental plasticity. PLoS Comput Biol 6:e1000805. https://doi.org/10.1371/journal.pcbi.1000805
Gempe T, Stach S, Bienefeld K et al (2016) Behavioral and molecular studies of quantitative differences in hygienic behavior in honeybees. BMC Res Notes 9:474. https://doi.org/10.1186/s13104-016-2269-y
Gramacho KP, Spivak M (2003) Differences in olfactory sensitivity and behavioral responses among honey bees bred for hygienic behavior. Behav Ecol Sociobiol 54:472–479. https://doi.org/10.1007/s00265-003-0643-y
Gramacho KP, Gonçalves LS, Rosenkranz P, De Jong D (1999) Influence of body fluid from pin-killed honey bee pupae on hygienic behavior. Apidologie 30:367–374. https://doi.org/10.1051/apido:19990502
Han B, Wei Q, Wu F et al (2021) Tachykinin signaling inhibits task-specific behavioral responsiveness in honeybee workers. eLife 10:e64830. https://doi.org/10.7554/eLife.64830
Harbo JR (1986) Effect of population size on brood production, worker survival and honey gain in colonies of honeybees. J Apicult Res 25:22–29. https://doi.org/10.1080/00218839.1986.11100687
Holbrook CT, Barden PM, Fewell JH (2011) Division of labor increases with colony size in the harvester ant Pogonomyrmex californicus. Behav Ecol 22:960–966. https://doi.org/10.1093/beheco/arr075
Hölldobler B, Wilson EO (1990) The Ants. The Belknap Press of Harvard University Press, Cambridge, Mass
Jack CJ, Ellis JD (2021) Integrated pest management control of Varroa destructor (Acari: Varroidae), the most damaging pest of (Apis mellifera L. (Hymenoptera: Apidae)) colonies. J Insect Sci 21:6. https://doi.org/10.1093/jisesa/ieab058
Jeanne RL, Loope KJ, Bouwma AM et al (2022) Five decades of misunderstanding in the social Hymenoptera: a review and meta-analysis of Michener’s paradox. Biol Rev 97:1559–1611. https://doi.org/10.1111/brv.12854
Jeanson R (2019) Within-individual behavioural variability and division of labour in social insects. J Exp Biol 222:jeb190868. https://doi.org/10.1242/jeb.190868
Jeanson R, Weidenmüller A (2014) Interindividual variability in social insects - proximate causes and ultimate consequences. Biol Rev 89:671–687. https://doi.org/10.1111/brv.12074
Jeanson R, Fewell JH, Gorelick R, Bertram SM (2007) Emergence of increased division of labor as a function of group size. Behav Ecol Sociobiol 62:289–298. https://doi.org/10.1007/s00265-007-0464-5
Karsai I, Wenzel JW (1998) Productivity, individual-level and colony-level flexibility, and organization of work as consequences of colony size. P Natl Acad Sci USA 95:8665–8669. https://doi.org/10.1073/pnas.95.15.8665
Leclercq G, Francis F, Gengler N, Blacquière T (2018) Bioassays to quantify hygienic behavior in honey bee (Apis mellifera L.) colonies: a review. J Apicult Res 57:663–673. https://doi.org/10.1080/00218839.2018.1494916
Lee PC, Winston ML (1985) The effect of swarm size and date of issue on comb construction in newly founded colonies of honeybees (Apis mellifera L.). Can J Zool 63:524–527. https://doi.org/10.1139/z85-077
London KB, Jeanne RL (2003) Effects of colony size and stage of development on defense response by the swarm-founding wasp Polybia occidentalis. Behav Ecol Sociobiol 54:539–546. https://doi.org/10.1007/s00265-003-0662-8
Masterman R, Smith BH, Spivak M (2000) Brood odor discrimination abilities in hygienic honey bees (Apis mellifera L.): Using proboscis extension reflex conditioning. J Insect Behav 13:87–101. https://doi.org/10.1023/A:1007767626594
Michener CD (1964) Reproductive efficiency in relation to colony size in hymenopterous societies. Insect Soc 11:317–341. https://doi.org/10.1007/bf02227433
Mondet F, Kim SH, de Miranda JR et al (2016) Specific cues associated with honey bee social defence against Varroa destructor infested brood. Sci Rep 6:25444. https://doi.org/10.1038/srep25444
Mondet F, Blanchard S, Barthes N et al (2021) Chemical detection triggers honey bee defense against a destructive parasitic threat. Nat Chem Biol 17:524–530. https://doi.org/10.1038/s41589-020-00720-3
Morisita M (1962) Iσ-Index, a measure of dispersion of individuals. Res Popul Ecol 4:1–7. https://doi.org/10.1007/BF02533903
Naug D (2009) Structure and resilience of the social network in an insect colony as a function of colony size. Behav Ecol Sociobiol 63:1023–1028. https://doi.org/10.1007/s00265-009-0721-x
Nazzi F, Della VG, D’Agaro M (2004) A semiochemical from brood cells infested by Varroa destructor triggers hygienic behaviour in Apis mellifera. Apidologie 35:65–70
Newton D, Ostasiewski N (1986) A simplified bioassay for behavioral resistance to American Foulbrood in honey bees (Apis mellifera L). Am Bee J 126:278–281
O’Quigley J (2008) Proportional Hazards Regression (Statistics for Biology and Health), 2008th edn. Springer, New York, p 560
Oldroyd BP, Fewell JH (2007) Genetic diversity promotes homeostasis in insect colonies. Trends Ecol Evol 22:408–413. https://doi.org/10.1016/j.tree.2007.06.001
Oxley PR, Spivak M, Oldroyd BP (2010) Six quantitative trait loci influence task thresholds for hygienic behaviour in honeybees (Apis mellifera). Mole Ecol 19:1452–1461
Pequeno PACL (2024) Resource adaptation drives the size-complexity rule in termites. Proc R Soc Lond B 291:20232363. https://doi.org/10.1098/rspb.2023.2363
Pérez-Sato J-A, Châline N, Martin SJ et al (2009) Multi-level selection for hygienic behaviour in honeybees. Heredity 102:609–615. https://doi.org/10.1038/hdy.2009.20
Posada-Florez F, Lamas ZS, Hawthorne DJ et al (2021) Pupal cannibalism by worker honey bees contributes to the spread of deformed wing virus. Sci Rep 11:1–12. https://doi.org/10.1038/s41598-021-88649-y
Reeve HK, Hölldobler B (2007) The emergence of a superorganism through intergroup competition. P Natl Acad Sci USA 104:9736–9740. https://doi.org/10.1073/pnas.0703466104
Rinderer TE, Harris JW, Hunt GJ, de Guzman LI (2010) Breeding for resistance to Varroa destructor in North America. Apidologie 41:409–424. https://doi.org/10.1051/apido/2010015
Rinkevich FD (2020) Detection of amitraz resistance and reduced treatment efficacy in the Varroa Mite, Varroa destructor, within commercial beekee** operations. PLoS ONE 15:e0227264. https://doi.org/10.1371/journal.pone.0227264
Rothenbuhler WC (1964) Behavior genetics of nest cleaning in honey bees. IV. Responses of F1 and backcross generations to disease-killed brood. Am Zool 4:111–123. https://doi.org/10.1093/icb/4.2.111
Rueppell O, Kaftanouglu O, Page RE (2009) Honey bee (Apis mellifera) workers live longer in small than in large colonies. Exp Gerontol 44:447–452
Rueppell O, Meier S, Deutsch R (2012) Multiple mating but not recombination causes quantitative increase in offspring genetic diversity for varying genetic architectures. PLoS ONE 7:e47220. https://doi.org/10.1016/j.exger.2009.04.003
Seeley TD (1985) Honeybee Ecology: A Study of Adaptation in Social Life. Princeton University Press, Princeton, NJ
Šimkovic M, Träuble B (2019) Robustness of statistical methods when measure is affected by ceiling and/or floor effect. PLoS ONE 14:e0220889. https://doi.org/10.1371/journal.pone.0220889
Smith ML, Koenig PA, Peters JM (2017) The cues of colony size: how honey bees sense that their colony is large enough to begin to invest in reproduction. J Exp Biol 220:1597–1605. https://doi.org/10.1242/jeb.150342
Spivak M, Danka RG (2021) Perspectives on hygienic behavior in Apis mellifera and other social insects. Apidologie 52:1–16. https://doi.org/10.1007/s13592-020-00784-z
Spivak M, Downey DL (1998) Field assays for hygienic behavior in honey bees (Hymenoptera: Apidae). J Econ Entomol 91:64–70. https://doi.org/10.1093/jee/91.1.64
Spivak M, Reuter GS (2001) Resistance to American foulbrood disease by honey bee colonies Apis mellifera bred for hygienic behavior. Apidologie 32:555–565. https://doi.org/10.1051/apido:2001103
Swanson JAI, Torto B, Kells SA et al (2009) Odorants that induce hygienic behavior in honeybees: Identification of volatile compounds in chalkbrood-infected honeybee larvae. J Chem Ecol 35:1108–1116. https://doi.org/10.1007/s10886-009-9683-8
Thomas ML, Elgar MA (2003) Colony size affects division of labour in the ponerine ant Rhytidoponera metallica. Naturwissenschaften 90:88–92. https://doi.org/10.1007/s00114-002-0396-x
Tsuji K, Tsuji N (1998) Indices of reproductive skew depend on average reproductive success. Evol Ecol 12:141–152. https://doi.org/10.1023/a:1006575411224
Ulrich Y, Saragosti J, Tokita CK et al (2018) Fitness benefits and emergent division of labour at the onset of group living. Nature 560:635–638. https://doi.org/10.1038/s41586-018-0422-6
Wagoner KM, Millar JG, Schal C, Rueppell O (2020) Cuticular pheromones stimulate hygienic behavior in the honey bee (Apis mellifera). Sci Rep 10:7132. https://doi.org/10.1038/s41598-020-64144-8
Wagoner KM, Spivak M, Millar JG et al (2021) Hygiene-eliciting brood semiochemicals as a tool for assaying honey bee (Hymenoptera: Apidae) colony resistance to Varroa (Mesostigmata: Varroidae). J Insect Sci 21:4. https://doi.org/10.1093/jisesa/ieab064
Wilson EO (1971) The Insect Societies. The Belknap Press of Harvard University Press, Cambridge, Mass
Wilson EO (1990) Success and dominance in ecosystems. the case of the social insects = excellence in ecology Vol. 2. In: Kinne O (ed) 15 figs. Published by Ecology Institute, Oldendorf/Luhe, Federal Republic of Germany, p 104
Winston ML (1987) The Biology of the Honey Bee. Harvard University Press, Cambridge, p 296
Winston ML, Dropkin JA, Taylor OR (1981) Demography and life history characteristics of two honey bee races (Apis mellifera). Oecologia 48:407–413. https://doi.org/10.1007/BF00346502
Yao M, Rosenfeld J, Attridge S et al (2009) The ancient chemistry of avoiding risks of predation and disease. Evol Biol 36:267–281. https://doi.org/10.1007/s11692-009-9069-4
Acknowledgements
The authors would like to thank Jennifer Keller, Moshe Nagari, Shaun Pitts, Jackson Keever, and Paz Kahanov for practical help with the experiments. Financial support was provided by a grant of the Binational Agricultural Research and Development Fund (IS-5078-18) to V.S. and O.R., grants by the US Army Research Office (W911NF2210195) and the National Science and Engineering Research Council of Canada (RGPIN-2022-03629) to O.R., and the Alberta Beekeepers Commission.
Author information
Authors and Affiliations
Contributions
P.S.: Conceptualization, Experimentation, Data collection, Formal analysis, Writing, J.M.: Computer modeling, J.J.H.: Experimentation, S.F.: Experimentation, K.M.W.: Conceptualization, Experimentation, Editing, V.S.: Conceptualization, Funding acquisition, Editing, O.R.: Conceptualization, Funding acquisition, Formal analysis, Writing.
Corresponding author
Ethics declarations
Conflict of interest
The funders had not influence on the design or interpretation of the study and the authors declare no competing interests.
Additional information
Communicated by B. Baer
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Snyder, P., Martin, J., Herman, J.J. et al. The impact of honey bee (Apis mellifera) group size on hygienic behavior performance. Behav Ecol Sociobiol 78, 52 (2024). https://doi.org/10.1007/s00265-024-03471-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-024-03471-6