Abstract
Objectives
To develop and validate a radiomics-based model for predicting SOX9-positive hepatocellular carcinoma (HCC) using preoperative contrast-enhanced computed tomography (CT) images.
Methods
From January 2013 to April 2017, patients with histologically proven HCC who received systemic sorafenib treatment after curative resection were retrospectively enrolled. Radiomic features were extracted from portal venous phase CT images and selected to build a radiomics score using logistic regression analysis. The factors associated with SOX9 expression were selected and combined by univariate and multivariate analyses to establish clinico-liver imaging (CL) model and clinico-liver imaging-radiomics (CLR) model. Diagnostic performance was measured by area under curve (AUC). Overall survival (OS) and recurrence-free survival (RFS) rates were compared using Kaplan-Meier method.
Results
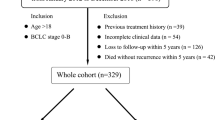
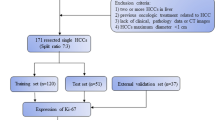
A total of 108 patients (training cohort: n = 80; validation cohort: n = 28) were enrolled. Multivariate analyses revealed that the albumin-bilirubin grade and tumor size were significant independent factors for predicting SOX9-positive HCCs and were included in the CL model. The CLR model integrating the radiomics score with albumin-bilirubin grade and tumor size showed better discriminative performance than the CL model with AUCs of 0.912 and 0.790 in the training and validation cohorts. Survival curves for RFS and OS showed that SOX9 expression was closely related to the prognosis of HCC patients. RFS and OS rates were significantly lower in patients with SOX9-positive than SOX9-negative (51.02% vs. 75.00% at 1-year RFS rates; 76.92% vs. 94.94% at 2-year OS rates).
Conclusion
Radiomics signatures may serve as noninvasive predictors for SOX9 status evaluation in patients with HCC and may aid in constructing individualized treatment strategies.
Graphical abstract






Similar content being viewed by others
Data availability
The datasets of the current study would be available from the corresponding author on reasonable request.
Abbreviations
- HCC:
-
Hepatocellular carcinoma
- CT:
-
Computed tomography
- CECT:
-
Contrast-enhanced computed tomography
- OS:
-
Overall survival
- RFS:
-
Recurrence-free survival
- SOX:
-
Sex-determining region Y box
- AUC:
-
Area under curve
- ROC:
-
Receiver operating characteristics.
- CSCs:
-
Cancer stem cells
- MVI:
-
Microvascular invasion
- ICIs:
-
Immune checkpoint inhibitors
- CAR-T:
-
Chimeric antigen receptor-modified T cell
- TACE:
-
Transcatheter arterial chemoembolization
- MVI:
-
Microvascular invasion
- VOI:
-
Volumes of interest
- ICC:
-
Intraclass correlation coefficient
- LASSO:
-
Least absolute shrinkage and selection operator
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin 2021;71:209-249.
Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc J-F, de Oliveira AC, et al. Sorafenib in Advanced Hepatocellular Carcinoma. New England Journal of Medicine 2008;359:378-390.
Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, Luo R, et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol 2009;10:25-34.
Huang Y, Zhang Z, Zhou Y, Yang J, Hu K, Wang Z. Should we apply sorafenib in hepatocellular carcinoma patients with microvascular invasion after curative hepatectomy? Onco Targets Ther 2019;12:541-548.
Dong W, Yan K, Yu H, Huo L, **an Z, Zhao Y, Li J, et al. Prognostic Nomogram for Sorafenib Benefit in Hepatitis B Virus-Related Hepatocellular Carcinoma After Partial Hepatectomy. Front Oncol 2020;10:605057.
Yang J, Liang H, Hu K, **ong Z, Cao M, Zhong Z, Yao Z, et al. The effects of several postoperative adjuvant therapies for hepatocellular carcinoma patients with microvascular invasion after curative resection: a systematic review and meta-analysis. Cancer Cell Int 2021;21:92.
Zhang W, Zhao G, Wei K, Zhang Q, Ma W, Song T, Wu Q, et al. Adjuvant sorafenib reduced mortality and prolonged overall survival and post-recurrence survival in hepatocellular carcinoma patients after curative resection: a single-center experience. Biosci Trends 2014;8:333-338.
**a F, Wu LL, Lau WY, Huan HB, Wen XD, Ma KS, Li XW, et al. Adjuvant sorafenib after heptectomy for Barcelona Clinic Liver Cancer-stage C hepatocellular carcinoma patients. World J Gastroenterol 2016;22:5384-5392.
Li J, Hou Y, Cai XB, Liu B. Sorafenib after resection improves the outcome of BCLC stage C hepatocellular carcinoma. World J Gastroenterol 2016;22:4034-4040.
Panda M, Tripathi SK, Biswal BK. SOX9: An emerging driving factor from cancer progression to drug resistance. Biochim Biophys Acta Rev Cancer 2021;1875:188517.
Guo W, Keckesova Z, Donaher JL, Shibue T, Tischler V, Reinhardt F, Itzkovitz S, et al. Slug and SOX9 cooperatively determine the mammary stem cell state. Cell 2012;148:1015-1028.
Kadaja M, Keyes BE, Lin M, Pasolli HA, Genander M, Polak L, Stokes N, et al. SOX9: a stem cell transcriptional regulator of secreted niche signaling factors. Genes Dev 2014;28:328-341.
Guo X, **ong L, Sun T, Peng R, Zou L, Zhu H, Zhang J, et al. Expression features of SOX9 associate with tumor progression and poor prognosis of hepatocellular carcinoma. Diagn Pathol 2012;7:44.
Liu C, Liu L, Chen X, Cheng J, Zhang H, Shen J, Shan J, et al. SOX9 regulates self-renewal and tumorigenicity by promoting symmetrical cell division of cancer stem cells in hepatocellular carcinoma. Hepatology 2016;64:117-129.
Leung CO, Mak WN, Kai AK, Chan KS, Lee TK, Ng IO, Lo RC. SOX9 confers stemness properties in hepatocellular carcinoma through Frizzled-7 mediated Wnt/β-catenin signaling. Oncotarget 2016;7:29371-29386.
Wang M, Wang Z, Zhi X, Ding W, **ong J, Tao T, Yang Y, et al. SOX9 enhances sorafenib resistance through upregulating ABCG2 expression in hepatocellular carcinoma. Biomed Pharmacother 2020;129:110315.
Aerts HJ, Velazquez ER, Leijenaar RT, Parmar C, Grossmann P, Carvalho S, Bussink J, et al. Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach. Nat Commun 2014;5:4006.
Gillies RJ, Kinahan PE, Hricak H. Radiomics: Images Are More than Pictures, They Are Data. Radiology 2016;278:563-577.
Lambin P, Rios-Velazquez E, Leijenaar R, Carvalho S, van Stiphout RG, Granton P, Zegers CM, et al. Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer 2012;48:441-446.
Segal E, Sirlin CB, Ooi C, Adler AS, Gollub J, Chen X, Chan BK, et al. Decoding global gene expression programs in liver cancer by noninvasive imaging. Nat Biotechnol 2007;25:675-680.
Rutman AM, Kuo MD. Radiogenomics: creating a link between molecular diagnostics and diagnostic imaging. Eur J Radiol 2009;70:232-241.
Ma X, Wei J, Gu D, Zhu Y, Feng B, Liang M, Wang S, et al. Preoperative radiomics nomogram for microvascular invasion prediction in hepatocellular carcinoma using contrast-enhanced CT. Eur Radiol 2019;29:3595-3605.
Chen S, Feng S, Wei J, Liu F, Li B, Li X, Hou Y, et al. Pretreatment prediction of immunoscore in hepatocellular cancer: a radiomics-based clinical model based on Gd-EOB-DTPA-enhanced MRI imaging. Eur Radiol 2019;29:4177-4187.
EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol 2018;69:182-236.
Heimbach JK, Kulik LM, Finn RS, Sirlin CB, Abecassis MM, Roberts LR, Zhu AX, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology 2018;67:358-380.
**e DY, Ren ZG, Zhou J, Fan J, Gao Q. 2019 Chinese clinical guidelines for the management of hepatocellular carcinoma: updates and insights. Hepatobiliary Surg Nutr 2020;9:452-463.
Bennett AV, Dueck AC, Mitchell SA, Mendoza TR, Reeve BB, Atkinson TM, Castro KM, et al. Mode equivalence and acceptability of tablet computer-, interactive voice response system-, and paper-based administration of the U.S. National Cancer Institute's Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). Health Qual Life Outcomes 2016;14:24.
Palmer DH. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 2008;359:2498; author reply 2498-2499.
Wang W, Gu D, Wei J, Ding Y, Yang L, Zhu K, Luo R, et al. A radiomics-based biomarker for cytokeratin 19 status of hepatocellular carcinoma with gadoxetic acid-enhanced MRI. Eur Radiol 2020;30:3004-3014.
Sun R, Limkin EJ, Vakalopoulou M, Dercle L, Champiat S, Han SR, Verlingue L, et al. A radiomics approach to assess tumour-infiltrating CD8 cells and response to anti-PD-1 or anti-PD-L1 immunotherapy: an imaging biomarker, retrospective multicohort study. Lancet Oncol 2018;19:1180-1191.
Gönczy P. Mechanisms of asymmetric cell division: flies and worms pave the way. Nat Rev Mol Cell Biol 2008;9:355-366.
Wang W, Li Y, Li X, Liu B, Han S, Li X, Zhang B, et al. Circular RNA circ-FOXP1 induced by SOX9 promotes hepatocellular carcinoma progression via sponging miR-875-3p and miR-421. Biomed Pharmacother 2020;121:109517.
Johnson PJ, Berhane S, Kagebayashi C, Satomura S, Teng M, Reeves HL, O'Beirne J, et al. Assessment of liver function in patients with hepatocellular carcinoma: a new evidence-based approach-the ALBI grade. J Clin Oncol 2015;33:550-558.
Kuo YH, Wang JH, Hung CH, Rau KM, Wu IP, Chen CH, Kee KM, et al. Albumin-Bilirubin grade predicts prognosis of HCC patients with sorafenib use. J Gastroenterol Hepatol 2017;32:1975-1981.
Abdel-Rahman O. Impact of baseline characteristics on outcomes of advanced HCC patients treated with sorafenib: a secondary analysis of a phase III study. J Cancer Res Clin Oncol 2018;144:901-908.
Wang C, Deng J, Deng H, Kang Z, Huang Z, Ding Z, Dong L, et al. A Novel SOX9/lncRNA H19 Axis Contributes to Hepatocyte Death and Liver Fibrosis. Toxicol Sci 2020;177:214-225.
Español-Suñer R, Carpentier R, Van Hul N, Legry V, Achouri Y, Cordi S, Jacquemin P, et al. Liver progenitor cells yield functional hepatocytes in response to chronic liver injury in mice. Gastroenterology 2012;143:1564-1575.e1567.
Pritchett J, Harvey E, Athwal V, Berry A, Rowe C, Oakley F, Moles A, et al. Osteopontin is a novel downstream target of SOX9 with diagnostic implications for progression of liver fibrosis in humans. Hepatology 2012;56:1108-1116.
Toyoda H, Lai PB, O'Beirne J, Chong CC, Berhane S, Reeves H, Manas D, et al. Long-term impact of liver function on curative therapy for hepatocellular carcinoma: application of the ALBI grade. Br J Cancer 2016;114:744-750.
Tabrizian P, Jibara G, Shrager B, Schwartz M, Roayaie S. Recurrence of hepatocellular cancer after resection: patterns, treatments, and prognosis. Ann Surg 2015;261:947-955.
Kawai T, Yasuchika K, Ishii T, Miyauchi Y, Kojima H, Yamaoka R, Katayama H, et al. SOX9 is a novel cancer stem cell marker surrogated by osteopontin in human hepatocellular carcinoma. Sci Rep 2016;6:30489.
Richtig G, Aigelsreiter A, Schwarzenbacher D, Ress AL, Adiprasito JB, Stiegelbauer V, Hoefler G, et al. SOX9 is a proliferation and stem cell factor in hepatocellular carcinoma and possess widespread prognostic significance in different cancer types. PLoS One 2017;12:e0187814.
Funding
This work was supported by the Science and Technology Support Program of Sichuan Province (Grant No. 2021YFS0021); China Postdoctoral Science Foundation (Grant No. 2021M692289); Post-Doctor Research Project, West China Hospital, Sichuan University (Grant No. 2020HXBH130); and National Natural Science Foundation of China (Grant No. 81971571).
Author information
Authors and Affiliations
Contributions
Conceptualization: FC and YW. Data curation: FC, YW, QX and ML. Formal analysis: FC, YW, QL, TZ and LW. Funding acquisition: YW and BS. Methodology: FC, YW and QL. Project administration: BS. Supervision: BS. Validation: FC, YW, TZ, LW and FY. Visualization: FC and QL. Writing-original draft: FC. Writing-review & editing: YW and QL.
Corresponding authors
Ethics declarations
Conflict of interest
All authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Che, F., Wei, Y., Xu, Q. et al. Noninvasive identification of SOX9 status using radiomics signatures may help construct personalized treatment strategy in hepatocellular carcinoma. Abdom Radiol (2024). https://doi.org/10.1007/s00261-024-04190-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00261-024-04190-2