Abstract
Purpose
The purpose of this study was to investigate the effects of the new anti-epileptic drug, lamotrigine, on cerebral blood flow by performing 99mTc-ethylcysteinate dimer (ECD) single-photon emission computed tomography (SPECT) before and after medication in patients with drug-naive idiopathic generalised epilepsy.
Methods
Interictal 99mTc-ECD brain SPECT was performed before drug treatment started and then repeated after lamotrigine medication for 4–5 months in 30 patients with generalised epilepsy (M/F=14/16, 19.3±3.4 years). Seizure types were generalised tonic-clonic seizure in 23 patients and myoclonic seizures in seven. The mean lamotrigine dose used was 214.1±29.1 mg/day. For SPM analysis, all SPECT images were spatially normalised to the standard SPECT template and then smoothed using a 12-mm full-width at half-maximum Gaussian kernel. The paired t test was used to compare pre- and post-lamotrigine SPECT images.
Results
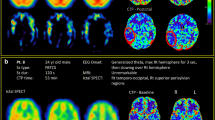
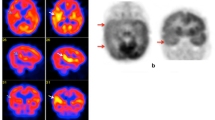
SPM analysis of pre- and post-lamotrigine brain SPECT images showed decreased perfusion in bilateral dorsomedial nuclei of thalami, bilateral uncus, right amygdala, left subcallosal gyrus, right superior and inferior frontal gyri, right precentral gyrus, bilateral superior and inferior temporal gyri and brainstem (pons, medulla) after lamotrigine medication at a false discovery rate-corrected p<0.05. No brain region showed increased perfusion after lamotrigine administration.
Conclusion
Our study demonstrates for the first time the effect of lamotrigine on interictal cerebral perfusion in drug-naive idiopathic generalised epilepsy patients. In summary, lamotrigine medication was found to reduce perfusion in cortico-thalamo-limbic areas, the orbitofrontal cortex, and brainstem.

Similar content being viewed by others
References
Theodore WH. Antiepileptic drugs and cerebral glucose metabolism. Epilepsia 1988;29:S48–S55
Theodore WH, Bromfield E, Onorati L. The effect of carbamazepine on cerebral glucose metabolism. Ann Neurol 1989;25: 516–520
Theodore WH, DiChiro G, Margolin R, Fishbein D, Porter RJ, Brooks RA. Barbiturates reduce human cerebral glucose metabolism. Neurology 1986;36:60–64
Theodore WH, Bairamian D, Newmark ME, DiChiro G, Porter RJ, Larson S, et al. Effect of phenytoin on human cerebral glucose metabolism. J Cereb Blood Flow Metab 1986b;6: 315–320
Leiderman DB, Balish M, Bromfield EB, Theodore WH. Effect of valproate on human cerebral glucose metabolism. Epilepsia 1991;32:417–422
Spanaki MV, Siegel H, Kopylev L, Fazilat S, Dean A, Liow K, et al. The effect of vigabatrin (gamma-vinyl GABA) on cerebral blood flow and metabolism. Neurology 1999;53:1518–1522
Gaillard WD, Zeffiro T, Fazilat S, DeCarli C, Theodore WH. Effect of valproate on cerebral metabolism and blood flow: an 18F-2-deoxyglucose and 15O water positron emission tomography study. Epilepsia 1996;37:515–521
Duncan JS. Idiopathic generalized epilepsies with typical absences. J Neurol 1997;244:403–411
Woermann FG, Free SL, Koepp MJ, Sisodiya SM, Duncan JS. Abnormal cerebral structure in juvenile myoclonic epilepsy demonstrated with voxel-based analysis of MRI. Brain 1999;122:2101–2108
Tae WS, Joo EY, Hong SB. Distribution of cerebral gray and white matters in juvenile myoclonic epilepsy: voxel based morphometry. J Korean Neurol Assoc 2003;21:62–69
Savic I, Osterman Y, Helms G. MRS shows syndrome differentiated metabolite changes in human-generalized epilepsies. Neuroimage 2004;21:163–172
Bercosani N, Andermann F, Arnold DL, Bercosani A. Entorhinal cortex MRI assessment in temporal, extratemporal and idiopathic generalized epilepsy. Epilepsia 2003;44: 1070–1074
Theodore WH, Brooks R, Margolin R, Patronas N, Sato S, Porter RJ, et al. Positron emission tomography in generalized seizures. Neurology 1985;35:684–690
Aghakhani Y, Bagshaw AP, Benar CG, Hawco C, Andermann F, Dubeau F, et al. fMRI activation during spike and wave discharges in idiopathic generalized epilepsy. Brain 2004;127:1127–1144
Pellock JM. Overview of lamotrigine and the new antiepileptic drugs: the challenge. J Child Neurol 1997;12:48–52
Manning JS, Haykal RF, Connor PD, Cunningham PD, Jackson WC, Long S. Sustained remission with lamotrigine augmentation or monotherapy in female resistant depressives with mixed cyclothymic-dysthymic temperament. J Affect Disord 2005;84:259–266
French J A, Kanner AM, Bautista J, Abou-Khalil B, Browne T, Harden CL, et al. Efficacy and tolerability of the new antiepileptic drugs. II: Treatment of refractory epilepsy. Epilepsia 2004;45:410–423
Leach MJ, Marden CM, Miller AA. Pharmacological studies on lamotrigine, a novel potential antiepileptic drug: II. Neurochemical studies on the mechanism of action. Epilepsia 1986;27:490–497
Waldmeier PC, Martin P, Stocklin K, Portet C, Schmutz M. Effect of carbamazepine, oxcarbazepine and lamotrigine on the increase in extracellular glutamate elicited by veratridine in rat cortex and striatum. Naunyn Schmiedebergs Arch Pharmacol 1996;354:164–172
Hassel B, Tauboll E, Gjerstad L. Chronic lamotrigine treatment increases rat hippocampal GABA shunt activity and elevates cerebral taurine levels. Epilepsy Res 2001;43:153–163
Chang L. A method for attenuation correction in computed tomography. IEEE Trans Nucl Sci 1987;25:638–643
Genovese CR, Lazar NA, Nichols T. Thresholding of statistical maps in functional neuroimaging using the false discovery rate. NeuroImage 2002;15:870–878
Cunningham MO, Jones RS. The anticonvulsant, lamotrigine decreases spontaneous glutamate release but increases spontaneous GABA release in the rat entorhinal cortex in vitro. Neuropharmacology 2000;39:2139–2146
Otsuki K, Morimoto K, Sato K, Yamada N, Kuroda S. Effects of lamotrigine and conventional antiepileptic drugs on amygdala- and hippocampal-kindled seizures in rats. Epilepsy Res 1998;31:101–112
Pericic D. Effects of γ-vinyl GABA on the enzymes of GABA system in specific brain regions. Period Biol 1980;82:19–23
Aldenkamp AP, Mulder OG, Overweg J. Cognitive effects of lamotrigine as first line add-on in patients with localized related (partial) epilepsy. J Epilepsy 1997;10:117–121
Bank GK, Beran RG. Neuropsychological assessment in lamotrigine treated epileptic patients. Clin Exp Neurol 1991;28:230–237
Calabrese JR, Vieta E, Shelton MD. Latest maintenance data on lamotrigine in bipolar disorder. Eur Neuropsychopharmacol 2003;13:57–66
Calabrese JR, Suppes T, Bowden CL, Sachs GS, Swann AC, McElroy SL, et al. A double-blind, placebo-controlled, prophylaxis study of lamotrigine in rapid-cycling bipolar disorder. J Clin Psychiatry 2000;61:841–850
Frye MA, Ketter TA, Kimbrell TA, Dunn RT, Speer AM, Osuch EA, et al. A placebo-controlled study of lamotrigine and gabapentin monotherapy in refractory mood disorders. J Clin Psychopharmacol 2000;20:607–614
Braga MF, Aroniadou-Anderjaska V, Post RM, Li H. Lamotrigine reduces spontaneous and evoked GABAA receptor-mediated synaptic transmission in the basolateral amygdala: implications for its effects in seizure and affective disorders. Neuropharmacology 2002;42:522–529
Steriade M. Llinas RR. The functional states of the thalamus and the associated neuronal interplay. Physiol Rev 1988;68:649–742
Pellegrini A, Dossi RC, Dal Pos F, Ermani M, Zanotto L, Testa G. Ethosuximide alters intrathalamic and thalamocortical synchronizing mechanisms: a possible explanation of its antiabsence effect. Brain Res 1989;497:344–360
Bourgeois B, Beaumanoir A, Blajev B, de la Cruz N, Despland PA, Egli M, et al. Monotherapy with valproate in primary generalized epilepsies. Epilepsia 1987;28:S8–S11
Li X, Teneback CC, Nahas Z, Kozel FA, Large C, Cohn J, et al. Interleaved transcranial magnetic stimulation/functional MRI confirms that lamotrigine inhibits cortical excitability in healthy young men. Neuropsychopharmacology 2004;29:1395–1407
Tergau F, Wischer S, Somal HS, Nitsche MA, Mercer AJ, Paulus W, et al. Relationship between lamotrigine oral dose, serum level and its inhibitory effect on CNS: insights from transcranial magnetic stimulation. Epilepsy Res 2003;56:67–77
Ballmaier M, Toga AW, Blanton RE, Sowell ER, Lavretsky H, Peterson J, et al. Anterior cingulate, gyrus rectus, and orbitofrontal abnormalities in elderly depressed patients: an MRI-based parcellation of the prefrontal cortex. Am J Psychiatry 2004;161:99–108
Hahn CG, Gyulai L, Baldassano CF, Lenox RH. The current understanding of lamotrigine as a mood stabilizer. J Clin Psychiatry 2004;65:791–804
Browning RA. Role of the brain-stem reticular formation in tonic-clonic seizures: lesion and pharmacological studies. Fed Proc 1985;44:2425–2431
Browning RA, Nelson DK. Variation in threshold and pattern of electroshock induced seizures in rats depending on site of stimulation. Life Sci 1985;37:2205–2211
Phillips B, Drake ME, Pakalnis A, Bogner J. Brainstem auditory evoked responses in partial and generalized seizures. Clin Electroencephalogr 1990;21:135–139
Lu KT, Gean PW. Endogenous serotonin inhibits epileptiform activity in rat hippocampal CA1 neurons via 5-hydroxytryptamine1A receptor activation. Neuroscience 1998;86:729–737
Wada Y, Nakamura M, Hasegawa H, Yamaguchi N. Role of serotonin receptor subtype in seizures kindled from the feline hippocampus. Neurosci Lett 1992;141:21–24
Lanfumey L, Hamon M. Central 5-HT(1A) receptors: regional distribution and functional characteristics. Nucl Med Biol 2000;27:429–435
Southam E, Kirkby D, Higgins GA, Hagan RM. Lamotrigine inhibits monoamine uptake in vitro and modulates 5-hydroxytryptamine uptake in rats. Eur J Pharmacol 1998;358:19–24
Ahmad S, Fowler LJ, Whitton PS. Effect of acute and chronic lamotrigine on basal and stimulated extracellular 5-hydroxytryptamine and dopamine in the hippocampus of the freely moving rat. Br J Pharmacol 2004;142:136–142
Acknowledgement
This study was supported by a grant (no. A050462) of the Good Health R&D Project, Ministry of Health and Welfare, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Joo, E.Y., Hong, S.B., Tae, W.S. et al. Effect of lamotrigine on cerebral blood flow in patients with idiopathic generalised epilepsy. Eur J Nucl Med Mol Imaging 33, 724–729 (2006). https://doi.org/10.1007/s00259-005-0029-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-005-0029-7