Abstract
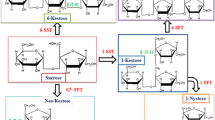
L-Fucose is one of the key metabolites in human–gut microbiome interactions. It is continuously synthesized by humans in the form of fucosylated glycans and fucosyl-oligosaccharides and delivered into the gut throughout their lifetime. Gut microorganisms metabolize L-fucose and produce short-chain fatty acids, which are absorbed by epithelial cells and used as energy sources or signaling molecules. Recent studies have revealed that the carbon flux in L-fucose metabolism by gut microorganisms is distinct from that in other sugar metabolisms because of cofactor imbalance and low efficiencies in energy synthesis of L-fucose metabolism. The large amounts of short-chain fatty acids produced during microbial L-fucose metabolism are used by epithelial cells to recover most of the energy used up during L-fucose synthesis. In this review, we present a detailed overview of microbial L-fucose metabolism and a potential solution for disease treatment and prevention using genetically engineered probiotics that modulate fucose metabolism. Our review contributes to the understanding of human–gut microbiome interactions through L-fucose metabolism.
Key points
• Fucose-metabolizing microorganisms produce large amounts of short-chain fatty acids
• Fucose metabolism differs from other sugar metabolisms by cofactor imbalance
• Modulating fucose metabolism is the key to control host–gut microbiome interactions


Similar content being viewed by others
Data availability
Data sharing is not applicable to this article as no datasets were generated or analyzed for this mini-review.
References
Amin HM, Hashem AM, Ashour MS, Hatti-Kaul R (2013) 1,2 Propanediol utilization by Lactobacillus reuteri DSM 20016, role in bioconversion of glycerol to 1,3 propanediol, 3-hydroxypropionaldehyde and 3-hydroxypropionic acid. J Genet Eng Biotechnol 11:53–59. https://doi.org/10.1016/j.jgeb.2012.12.002
Ashida H, Miyake A, Kiyohara M, Wada J, Yoshida E, Kumagai H, Katayama T, Yamamoto K (2009) Two distinct α-L-fucosidases from Bifidobacterium bifidum are essential for the utilization of fucosylated milk oligosaccharides and glycoconjugates. Glycobiology 19:1010–1017. https://doi.org/10.1093/glycob/cwp082
Autieri SM, Lins JJ, Leatham MP, Laux DC, Conway T, Cohen PS (2007) L-Fucose stimulates utilization of D-ribose by Escherichia coli MG1655 ΔfucAO and E. coli Nissle 1917 ΔfucAO mutants in the mouse intestine and in M9 minimal medium. Infect Immun 75:5465–5475. https://doi.org/10.1128/IAI.00822-07
Bunesova V, Lacroix C, Schwab C (2016) Fucosyllactose and L-fucose utilization of infant Bifidobacterium longum and Bifidobacterium kashiwanohense. BMC Microbiol 16:248. https://doi.org/10.1186/s12866-016-0867-4
Centanni M, Ferguson SA, Sims IM, Biswas A, Tannock GW (2019) Bifidobacterium bifidum ATCC 15696 and Bifidobacterium breve 24b metabolic interaction based on 2′-O-fucosyl-lactose studied in steady-state cultures in a freter-style chemostat. Appl Environ Microbiol 85:e02783–e02718. https://doi.org/10.1128/AEM.02783-18
Cheong YE, Kim J, ** Y-S, Kim KH (2022) Elucidation of the fucose metabolism of probiotic Lactobacillus rhamnosus GG by metabolomic and flux balance analyses. J Biotechnol 360:110–116. https://doi.org/10.1016/j.jbiotec.2022.11.002
Dedon LR, Özcan E, Rani A, Sela DA (2020) Bifidobacterium infantis metabolizes 2′fucosyllactose-derived and free fucose through a common catabolic pathway resulting in 1,2-propanediol secretion. Front Nutr 7:583397. https://doi.org/10.3389/fnut.2020.583397
Diene SM, Merhej V, Henry M, El Filali A, Roux V, Robert C, Azza S, Gavory F, Barbe V, La Scola B, Raoult D, Rolain J-M (2013) The rhizome of the multidrug-resistant Enterobacter aerogenes genome reveals how new “killer bugs” are created because of a sympatric lifestyle. Mol Biol Evol 30:369–383. https://doi.org/10.1093/molbev/mss236
Duncan SH, Holtrop G, Lobley GE, Calder AG, Stewart CS, Flint HJ (2004a) Contribution of acetate to butyrate formation by human faecal bacteria. Br J Nutr 91:915–923. https://doi.org/10.1079/BJN20041150
Duncan SH, Louis P, Flint HJ (2004b) Lactate-utilizing bacteria, isolated from human feces, that produce butyrate as a major fermentation product. Appl Environ Microbiol 70:5810–5817. https://doi.org/10.1128/AEM.70.10.5810-5817.2004
Engels C, Ruscheweyh H-J, Beerenwinkel N, Lacroix C, Schwab C (2016) The common gut microbe Eubacterium hallii also contributes to intestinal propionate formation. Front Microbiol 7. https://doi.org/10.3389/fmicb.2016.00713
Garber JM, Hennet T, Szymanski CM (2021) Significance of fucose in intestinal health and disease. Mol Microbiol 115:1086–1093. https://doi.org/10.1111/mmi.14681
Goto Y, Uematsu S, Kiyono H (2016) Epithelial glycosylation in gut homeostasis and inflammation. Nat Immunol 17:1244–1251. https://doi.org/10.1038/ni.3587
Holdener BC, Haltiwanger RS (2019) Protein O-fucosylation: structure and function. Curr Opin Struct Biol 56:78–86. https://doi.org/10.1016/j.sbi.2018.12.005
Hudson AW, Barnes AJ, Bray AS, Ornelles DA, Zafar MA (2022) Klebsiella pneumoniae L-fucose metabolism promotes gastrointestinal colonization and modulates its virulence determinants. Infect Immun 90:e00206–e00222. https://doi.org/10.1128/iai.00206-22
Joseph S, Desai P, Ji Y, Cummings CA, Shih R, Degoricija L, Rico A, Brzoska P, Hamby SE, Masood N, Hariri S, Sonbol H, Chuzhanova N, McClelland M, Furtado MR, Forsythe SJ (2012) Comparative analysis of genome sequences covering the seven Cronobacter species. PLoS One 7:e49455. https://doi.org/10.1371/journal.pone.0049455
Kashyap PC, Marcobal A, Ursell LK, Smits SA, Sonnenburg ED, Costello EK, Higginbottom SK, Domino SE, Holmes SP, Relman DA, Knight R, Gordon JI, Sonnenburg JL (2013) Genetically dictated change in host mucus carbohydrate landscape exerts a diet-dependent effect on the gut microbiota. Proc Natl Acad Sci U S A 110:17059–17064. https://doi.org/10.1073/pnas.1306070110
Kim J, Cheong YE, Jung I, Kim KH (2019) Metabolomic and transcriptomic analyses of Escherichia coli for efficient fermentation of L-fucose. Mar Drugs 17:82. https://doi.org/10.3390/md17020082
Kim J, Cheong YE, Yu S, ** Y-S, Kim KH (2022) Strain engineering and metabolic flux analysis of a probiotic yeast Saccharomyces boulardii for metabolizing l-fucose, a mammalian mucin component. Microb Cell Factories 21:204. https://doi.org/10.1186/s12934-022-01926-x
Kononova S, Litvinova E, Vakhitov T, Skalinskaya M, Sitkin S (2021) Acceptive immunity: the role of fucosylated glycans in human host–microbiome interactions. Int J Mol Sci 22:3854. https://doi.org/10.3390/ijms22083854
Kostopoulos I, Elzinga J, Ottman N, Klievink JT, Blijenberg B, Aalvink S, Boeren S, Mank M, Knol J, de Vos WM, Belzer C (2020) Akkermansia muciniphila uses human milk oligosaccharides to thrive in the early life conditions in vitro. Sci Rep 10:14330. https://doi.org/10.1038/s41598-020-71113-8
Kudelka MR, Stowell SR, Cummings RD, Neish AS (2020) Intestinal epithelial glycosylation in homeostasis and gut microbiota interactions in IBD. Nat Rev Gastroenterol Hepatol 17:597–617. https://doi.org/10.1038/s41575-020-0331-7
Lee S, Inzerillo S, Lee GY, Bosire EM, Mahato SK, Song J (2022) Glycan-mediated molecular interactions in bacterial pathogenesis. Trends Microbiol 30:254–267. https://doi.org/10.1016/j.tim.2021.06.011
Li M, Bai Y, Zhou J, Huang W, Yan J, Tao J, Fan Q, Liu Y, Mei D, Yan Q, Yuan J, Malard P, Wang Z, Gu J, Tanigchi N, Li W (2019) Core fucosylation of maternal milk N-glycan evokes B cell activation by selectively promoting the L-fucose metabolism of gut Bifidobacterium spp and Lactobacillus spp. mBio 10:e00128–e00119. https://doi.org/10.1128/mBio.00128-19
Liu J-J, Kwak S, Pathanibul P, Lee JW, Yu S, Yun EJ, Lim H, Kim KH, ** Y-S (2018) Biosynthesis of a functional human milk oligosaccharide, 2′-fucosyllactose, and L-fucose using engineered Saccharomyces cerevisiae. ACS Synth Biol 7:2529–2536. https://doi.org/10.1021/acssynbio.8b00134
Louis P, Flint HJ (2017) Formation of propionate and butyrate by the human colonic microbiota. Environ Microbiol 19:29–41. https://doi.org/10.1111/1462-2920.13589
Luijkx YMCA, Bleumink NMC, Jiang J, Overkleeft HS, Wösten MMSM, Strijbis K, Wennekes T (2020) Bacteroides fragilis fucosidases facilitate growth and invasion of Campylobacter jejuni in the presence of mucins. Cell Microbiol 22:e13252. https://doi.org/10.1111/cmi.13252
Millard P, Pérochon J, Létisse F (2021) Functional analysis of deoxyhexose sugar utilization in Escherichia coli reveals fermentative metabolism under aerobic conditions. Appl Environ Microbiol 87:e00719–e00721. https://doi.org/10.1128/AEM.00719-21
Moore RE, Xu LL, Townsend SD (2021) Prospecting human milk oligosaccharides as a defense against viral infections. ACS Infect Dis 7:254–263. https://doi.org/10.1021/acsinfecdis.0c00807
Moya-Gonzálvez EM, Peña-Gil N, Rubio-del-Campo A, Coll-Marqués JM, Gozalbo-Rovira R, Monedero V, Rodríguez-Díaz J, Yebra MJ (2022) Infant gut microbial metagenome mining of α-L-fucosidases with activity on fucosylated human milk oligosaccharides and glycoconjugates. Microbiol Spectr 10:e01775–e01722. https://doi.org/10.1128/spectrum.01775-22
Pacheco AR, Curtis MM, Ritchie JM, Munera D, Waldor MK, Moreira CG, Sperandio V (2012) Fucose sensing regulates bacterial intestinal colonization. Nature 492:113–117. https://doi.org/10.1038/nature11623
Parada Venegas D, De la Fuente MK, Landskron G, González MJ, Quera R, Dijkstra G, Harmsen HJM, Faber KN, Hermoso MA (2019) Short chain fatty acids (SCFAs)-mediated gut epithelial and immune regulation and its relevance for inflammatory bowel diseases. Front Immunol 10. https://doi.org/10.3389/fimmu.2019.00277
Park Y-T, Kim T, Ham J, Choi J, Lee H-S, Yeon YJ, Choi SI, Kim N, Kim Y-R, Seok Y-J (2022) Physiological activity of E. coli engineered to produce butyric acid. Microb Biotechnol 15:832–843. https://doi.org/10.1111/1751-7915.13795
Pérez-Reytor D, Puebla C, Karahanian E, García K (2021) Use of short-chain fatty acids for the recovery of the intestinal epithelial barrier affected by bacterial toxins. Front Physiol 12. https://doi.org/10.3389/fphys.2021.650313
Qing J, Hu X, Li C, Song W, Tirichen H, Yaigoub H, Li Y (2022) Fucose as a potential therapeutic molecule against the immune-mediated inflammation in IgA nepharopathy: an unrevealed link. Front Immunol 13. https://doi.org/10.3389/fimmu.2022.929138
Rodríguez-Díaz J, Rubio-del-Campo A, Yebra MJ (2012) Lactobacillus casei ferments the N-acetylglucosamine moiety of fucosyl-α-1,3-N-acetylglucosamine and excretes L-fucose. Appl Environ Microbiol 78:4613–4619. https://doi.org/10.1128/AEM.00474-12
Sadeghzadeh Z, Khosravi A, Jazi MS, Asadi J (2020) Upregulation of fucosyltransferase 3, 8 and protein O-fucosyltransferase 1, 2 genes in esophageal cancer stem-like cells (CSLCs). Glycoconj J 37:319–327. https://doi.org/10.1007/s10719-020-09917-z
Salvi PS, Cowles RA (2021) Butyrate and the intestinal epithelium: modulation of proliferation and inflammation in homeostasis and disease. Cells 10:1775. https://doi.org/10.3390/cells10071775
Sela DA, Garrido D, Lerno L, Wu S, Tan K, Eom H-J, Joachimiak A, Lebrilla CB, Mills DA (2012) Bifidobacterium longum subsp. infantis ATCC 15697 α-fucosidases are active on fucosylated human milk oligosaccharides. Appl Environ Microbiol 78:795–803. https://doi.org/10.1128/AEM.06762-11
Shan M, Yang D, Dou H, Zhang L (2019) Fucosylation in cancer biology and its clinical applications. In: Zhang L (ed) Progress in Molecular Biology and Translational Science. Academic Press, Cambridge, pp 93–119
Silva YP, Bernardi A, Frozza RL (2020) The role of short-chain fatty acids from gut microbiota in gut-brain communication. Front Endocrinol 11. https://doi.org/10.3389/fendo.2020.00025
Stahl M, Friis LM, Nothaft H, Liu X, Li J, Szymanski CM, Stintzi A (2011) L-Fucose utilization provides Campylobacter jejuni with a competitive advantage. Proc Natl Acad Sci U S A 108:7194–7199. https://doi.org/10.1073/pnas.1014125108
Urashima T, Katayama T, Sakanaka M, Fukuda K, Messer M (2022) Evolution of milk oligosaccharides: origin and selectivity of the ratio of milk oligosaccharides to lactose among mammals. Biochim Biophys Acta Gen Subj 1866:130012. https://doi.org/10.1016/j.bbagen.2021.130012
Vandenplas Y, Berger B, Carnielli VP, Ksiazyk J, Lagström H, Sanchez Luna M, Migacheva N, Mosselmans J-M, Picaud J-C, Possner M, Singhal A, Wabitsch M (2018) Human milk oligosaccharides: 2′-fucosyllactose (2′-FL) and Lacto-N-neotetraose (LNnT) in infant formula. Nutrients 10:1161. https://doi.org/10.3390/nu10091161
Wan L, Zhu Y, Zhang W, Mu W (2020) α-L-Fucosidases and their applications for the production of fucosylated human milk oligosaccharides. Appl Microbiol Biotechnol 104:5619–5631. https://doi.org/10.1007/s00253-020-10635-7
You J, Lin S, Jiang T (2019) Origins and evolution of the α-L-fucosidases: from bacteria to metazoans. Front Microbiol 10. https://doi.org/10.3389/fmicb.2019.01756
Yun EJ, Liu J-J, Lee JW, Kwak S, Yu S, Kim KH, ** Y-S (2019) Biosynthetic routes for producing various fucosyl-oligosaccharides. ACS Synth Biol 8:415–424. https://doi.org/10.1021/acssynbio.8b00436
Funding
This work was supported by the Korea Institute of Planning and Evaluation for Technology in Food, Agriculture, Forestry, and Fisheries funded by the Ministry of Agriculture, Food and Rural Affairs (32136-05-1-SB010) and Ministry of Trade, Industry and Energy (20018132). KHK also received grant support from the Korea University and Food Safety Hall for the Institute of Biomedical Science and Food Safety, Korea University.
Author information
Authors and Affiliations
Contributions
JK searched and organized the literature and drafted the manuscript. KHK and YSJ reviewed the integrity and accuracy of the manuscript. All authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval
Not applicable
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, J., **, YS. & Kim, K.H. L-Fucose is involved in human–gut microbiome interactions. Appl Microbiol Biotechnol 107, 3869–3875 (2023). https://doi.org/10.1007/s00253-023-12527-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-023-12527-y