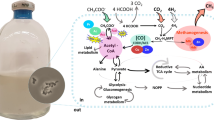
Abstract
Methanotrophs are able to metabolize volatile organic sulfur compounds (VOSCs), excrete organic carbon during CH4 oxidation, and influence microbial community structure and function of the ecosystem. In return, microbial community structure and environmental factors can affect the growth metabolism of methanotrophs. In this study, Methylomonas koyamae and Hyphomicrobium methylovorum were used for model organisms, and methanethiol (MT) was chosen for a typical VOSC to investigate the synergy effects under VOSC stress. The results showed that when Hyphomicrobium methylovorum was co-cultured with Methylomonas koyamae in the medium with CH4 used as the carbon source, the co-culture had better MT tolerance relative to Methylomonas koyamae and oxidized all CH4 within 120 h, even at the initial MT concentration of 2000 mg m−3. The optimal co-culture ratios of Methylomonas koyamae to Hyphomicrobium methylovorum were 4:1–12:1. Although MT could be converted spontaneously to dimethyl disulfide (DMDS), H2S, and CS2 in air, faster losses of MT, DMDS, H2S, and CS2 were observed in each strain mono-culture and the co-culture. Compared with Hyphomicrobium methylovorum, MT was degraded more quickly in the Methylomonas koyamae culture. During the co-culture, the CH4 oxidation process of Methylomonas koyamae could provide carbon and energy sources for the growth of Hyphomicrobium methylovorum, while Hyphomicrobium methylovorum oxidized MT to help Methylomonas koyamae detoxify. These findings are helpful to understand the synergy effects of Methylomonas koyamae and Hyphomicrobium methylovorum under MT stress and enrich the role of methanotrophs in the sulfur biogeochemical cycle.
Key points
• The co-culture of Methylomonas and Hyphomicrobium has better tolerance to CH 3 SH.
• Methylomonas can provide carbon sources for the growth of Hyphomicrobium.
• The co-culture of Methylomonas and Hyphomicrobium enhances the removal of CH 4 and CH 3 SH.
Graphical Abstract








Similar content being viewed by others
Data availability statement
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding author.
References
Benassi R (2004) Ab initio study of the oxidation of CH3SH to CH3SSCH3. Theor Chem Acc 112(2):95–105
Bodelier PLE, Meima-Franke M, Hordijk CA, Steenbergh AK, Hefting M, Bodrossy L, Mv B, Seifert J (2013) Microbial minorities modulate methane consumption through niche partitioning. ISME J 7:2214–2228
Boden R, Kelly DP, Murrell JC, Schäfer H (2010) Oxidation of dimethylsulfide to tetrathionate by Methylophaga thiooxidans sp. nov.: a new link in the sulfur cycle. Environ Microbiol 12(10):2688–2699
Cáceres M, Morales M, Martín RS, Urrutia H, Aroca G (2010) Oxidation of volatile reduced sulphur compounds in biotrickling filter inoculated with Thiobacillus thioparus. Electron J Biotechnol 13(5):1–11
Chen M, Yao X, Ma R, Song Q, Long Y, He R (2017) Methanethiol generation potential from anaerobic degradation of municipal solid waste in landfills. Environ Sci Pollut Res 24(30):23992–24001
Cho K-s, Hirai M, Shoda M (1992) Enhanced removability of odorous sulfur-containing gases by mixed cultures of purified bacteria from peat biofilters. J Ferment Bioeng 73:219–224
de Zwart JMM, Nelisse PN, Kuenen JG (1996) Isolation and characterization of Methylophaga sulfidovorans sp. nov.: an obligately methylotrophic, aerobic, dimethylsulfide oxidizing bacterium from a microbial mat. FEMS Microbiol Ecol 20(4):261–270
Eyice Ö, Myronova N, Pol A, Carrión O, Todd JD, Smith TJ, Gurman SJ, Cuthbertson A, Mazard S, Mennink-Kersten MASH, Bugg TDH, Andersson KK, Johnston AWB, Op den Camp HJM, Schäfer H (2018) Bacterial SBP56 identified as a Cu-dependent methanethiol oxidase widely distributed in the biosphere. ISME J 12(1):145–160
Fernandez HT, Rico IR, Prida JJdl, Langenhove HV (2013) Dimethyl sulfide bioflltration using immobilized Hyphomicrobium VS and Thiobadllus thioparus TK-m in sugarcane bagasse. Environ Technol 34(2):257–262
Fernández HTT, Rico ILR, de la Prida JJ, Van Langenhove HR (2013) Dimethyl sulfide biofiltration using immobilized Hyphomicrobium VS and Thiobacillus thioparus TK-m in sugarcane bagasse. Environ Technol 34(2):257–262
Gęsicka A, Oleskowicz-Popiel P, Łężyk M (2021) Recent trends in methane to bioproduct conversion by methanotrophs. Biotechnol Adv 53:107861
Gu W, Sun W, Lu Y, Li X, Xu P, **e K, Sun L, Wu H (2018) Effect of Thiobacillus thioparus 1904 and sulphur addition on odour emission during aerobic composting. Bioresour Technol 249:254–260
Gwak J-H, Awala SI, Nguyen N-L, Yu W-J, Yang H-Y, von Bergen M, Jehmlich N, Kits KD, Loy A, Dunfield PF, Dahl C, Hyun J-H, Rhee S-K (2022) Sulfur and methane oxidation by a single microorganism. Proc Natl Acad Sci USA 119(32):e2114799119
He R, **a F, Bai Y, Wang J, Shen D (2012) Mechanism of H2S removal during landfill stabilization in waste biocover soil, an alterative landfill cover. J Hazard Mater 217–218:67–75
He R, Yao X, Chen M, Ma R, Li H, Wang C, Ding S (2018) Conversion of sulfur compounds and microbial community in anaerobic treatment of fish and pork waste. Waste Manage 76:383–393
He R, Su Y, Leewis MC, Chu Y, Wang J, Ma R, Wu D, Zhan L, Herriott IC, Leigh MB (2020) Low O2 level enhances CH4-derived carbon flow into microbial communities in landfill cover soils. Environ Pollut 258:113676
He R, Wang J, Pohlman JW, Jia Z, Chu Y, Wooller MJ, Leigh MB (2022) Metabolic flexibility of aerobic methanotrophs under anoxic conditions in Arctic lake sediments. ISME J 16:78–90
Jia T, Sun S, Chen K, Zhang L, Peng Y (2020) Simultaneous methanethiol and dimethyl sulfide removal in a single-stage biotrickling filter packed with polyurethane foam: performance, parameters and microbial community analysis. Chemosphere 244:125460
Kalyuzhnaya MG, Yang S, Rozova ON, Smalley NE, Clubb J, Lamb AD, Gowda GAN, Raftery D, Fu Y, Fo B, Vuilleumier S, Beck DAC, Trotsenko YA, Khmelenina VN, Lidstrom ME (2013) Highly efficient methane biocatalysis revealed in a methanotrophic bacterium. Nat Commun 4:2785
Kim Hg, Doronina NV, Trotsenko YA, Kim SW (2007) Methylophaga aminisulfidivorans sp. nov., a restricted facultatively methylotrophic marine bacterium. Int J Syst Evol Microbiol 57(9):2096–2101
Krober E, Schafer H (2019) Identification of proteins and genes expressed by Methylophaga thiooxydans during growth on dimethylsulfide and their presence in other members of the genus. Front Microbiol 10:1132
Ksibi M (2006) Chemical oxidation with hydrogen peroxide for domestic wastewater treatment. Chem Eng J 119(2–3):161–165
Lee C-L, Brimblecombe P (2016) Anthropogenic contributions to global carbonyl sulfide, carbon disulfide and organosulfides fluxes. Earth-Sci Rev 160:1–18
Lee SG, Goo JH, Kim HG, Oh J-I, Kim YM, Kim SW (2004) Optimization of methanol biosynthesis from methane using Methylosinus trichosporium OB3b. Biotechnol Lett 26:947–950
Lee J-H, Kim TG, Cho K-S (2012) Isolation and characterization of a facultative methanotroph degrading malodor-causing volatile sulfur compounds. J Hazard Mater 235–236:224–229
Li Y, Gong X, Zhao Z, Shen Q, Zhang L (2022) Distribution and release of volatile organic sulfur compounds in Yangcheng Lake. Water 14:1199
Ma R-C, Chu Y-X, Wang J, Wang C, Leigh MB, Chen Y, He R (2021) Stable-isotopic and metagenomic analyses reveal metabolic and microbial link of aerobic methane oxidation coupled to denitrification at different O2 levels. Sci Total Environ 764:142901
Manheim DC, Yeşiller N, Hanson JL (2021) Climate change effects of gases from municipal solid waste landfills. Jpn Geotech Soc Spec Publ 9(4):142–147
Oshkin IY, Beck DAC, Lamb AE, Tchesnokova V, Benuska G, McTaggart TL, Kalyuzhnaya MG, Dedysh SN, Lidstrom ME, Chistoserdova L (2014) Methane-fed microbial microcosms show differential community dynamics and pinpoint taxa involved in communal response. ISME J 9:1119–1129
Ozbay G, Jones MB, Gadde M, Isah S, Attarwala T (2021) Design and operation of effective landfills with minimal effects on the environment and human health. J Environ Public Health 2021:6921607
Pokorna D, Zabranska J (2015) Sulfur-oxidizing bacteria in environmental technology. Biotechnol Adv 33(6):1246–1259
Prenafeta-Boldú FX, Rojo N, Gallastegui G, Guivernau M, Viñas M, Elias A (2014) Role of Thiobacillus thioparus in the biodegradation of carbon disulfide in a biofilter packed with a recycled organic pelletized material. Biodegradation 25:557–568
Rahalkar MC (2006) Aerobic methanotrophic bacterial communities in sediments of Lake Constance.
Rojo N, Gallastegi G, Barona A, Gurtubay L, Ibarra-Berastegi G, Elias A (2010) Biotechnology as an alternative for carbon disulfide treatment in air pollution control. Environ Rev 18:321–332
Schiffman SS, Bennett JL, Raymer JH (2001) Quantification of odors and odorants from swine operations in North Carolina. Agric for Meteorol 108(3):213–240
Schmitz RA, Peeters SH, Versantvoort W, Picone N, Pol A, Jetten MSM, Op den Camp HJM (2021) Verrucomicrobial methanotrophs: ecophysiology of metabolically versatile acidophiles. FEMS Microbiol Rev 45(5):007
Schmitz RA, Mohammadi SS, van Erven T, Berben T, Jetten MSM, Pol A, Op den Camp HJM (2022) Methanethiol consumption and hydrogen sulfide production by the thermoacidophilic methanotroph Methylacidiphilum fumariolicum SolV. Front Microbiol 13:857442
Schmitz RA, Peeters SH, Mohammadi SS, Berben T, van Erven T, Iosif CA, van Alen T, Versantvoort W, Jetten MSM, Op den Camp HJM, Pol A (2022b) Methanotrophs are vigorous H2S oxidizers using a sulfide:quinone oxidoreductase and a ba3-type terminal oxidase. bioRxiv:505896. https://doi.org/10.1101/2022.08.31.505896
Semrau JD, DiSpirito AA, Yoon S (2010) Methanotrophs and copper. FEMS Microbiol Rev 34(4):496–531
Shihab MS, Mhemid RKS, Saeed L, Ismail H, Alp K (2022) Reducing volatile organic compound emissions using biotrickling filters and bioscrubber systems. J Ecol Eng 23(10):255–268
Shmareva MN, Doronina NV, Tarlachkov SV, Vasilenko OV, Trotsenko YA (2018) Methylophaga muralis Bur 1, a haloalkaliphilic methylotroph isolated from the Khilganta soda lake (Southern Transbaikalia, Buryat Republic). Microbiology 87:33–46
Siddharth T, Sridhar P, Vinila VS, Tyagi RD (2021) Environmental applications of microbial extracellular polymeric substance (EPS): a review. J Environ Manage 287:112307
Sonawane JM, Rai AK, Sharma M, Tripathi M, Prasad R (2022) Microbial biofilms: recent advances and progress in environmental bioremediation. Sci Total Environ 824:153843
Su Y, Zhang X, Wei X, Kong J, **a F, Li W, He R (2014) Evaluation of simultaneous biodegradation of methane and toluene in landfill covers. J Hazard Mater 274:367–375
Suylen G, Large PJ, Dijken J, Kuenen JG (1987) Methyl mercaptan oxidase, a key enzyme in the metabolism of methylated sulphur compounds by Hyphomicrobium EG. J Gen Microbiol 133(11):2989–2997
Tilmes S, Muller R, Salawitch RJ, Schmidt U, Webster CR, Oelhaf H, Camy-Peyret C, Russell JM (2008) Chemical ozone loss in the Arctic winter 1991–1992. Copernicus GmbH 8(7):1897–1910
van Spanning RJM, Guan Q, Melkonian C, Gallant JL, Polerecky L, Flot JF, Brandt BW, Braster M, Iturbe Espinoza P, Aerts JW, Meima-Franke M, Piersma SR, Bunduc CM, Ummels R, Pain A, Fleming EJ, van der Wel NN, Gherman VD, Sarbu SM, Bodelier PLE, Bitter W (2022) Methanotrophy by a Mycobacterium species that dominates a cave microbial ecosystem. Nat Microbiol 7:2089–2100
Wang J, **a F, Bai Y, Fang C, Shen D, He R (2011) Methane oxidation in landfill waste biocover soil: kinetics and sensitivity to ambient conditions. Waste Manage 31(5):864–870
Wang J, Chu Y, Yao X, He R (2021) Enhanced degradation of methanethiol in enrichment cultures in the presence of methane. Biochem Eng J 168:107934
Wang J, Wei Z, Chu Y-X, Tian G-M, He R (2022) Eutrophic levels and algae growth increase emissions of methane and volatile sulfur compounds from lakes. Environ Pollut 306:119435
Watson SB, Jüttner F (2017) Malodorous volatile organic sulfur compounds: sources, sinks and significance in inland waters. Crit Rev Microbiol 43(2):210–237
Wei Z, Sun J, JC H, Huang Q, Chen Z, Ye Q, Luo Y (2014) Dimethyl sulfide removal using biofilter immobilized Thiobacillus sp. ST22. J Environ Eng 140(11):06014004
Wei X, Su Y, Zhang H, Chen M, He R (2015) Responses of methanotrophic activity, community and EPS production to CH4 and O2 concentrations in waste biocover soils. Waste Manage 42:118–127
Yang H, Jung H, Oh K-C, Jeon J-M, Cho K-S (2021) Characterization of the bacterial community associated with methane and odor in a pilot-scale landfill biocover under moderately thermophilic conditions. J Microbiol Biotechnol 31(6):803–814
Yao X, Ma R, Li H, Wang C, Zhang C, Yin S, Wu D, He X, Wang J, Zhan L, He R (2019) Assessment of the major odor contributors and health risks of volatile compounds in three disposal technologies for municipal solid waste. Waste Manage 91:128–138
Zhang L, Hirai M, Shoda M (1991) Removal characteristics of dimethyl sulfide, methanethiol and hydrogen sulfide by Hyphomicrobium sp. 155 isolated from peat biofilter. J Ferment Bioeng 72:392–396
Zhou J, Zheng G, Wong JWC, Zhou L (2013) Degradation of inhibitory substances in sludge by Galactomyces sp. Z3 and the role of its extracellular polymeric substances in improving bioleaching. Bioresour Technol 132:217–223
Zhu J, Xu X, Yuan M, Wu H, Ma Z, Wu W (2017) Optimum O2:CH4 ratio promotes the synergy between aerobic methanotrophs and denitrifiers to enhance nitrogen removal. Front Microbiol 8:1112
Funding
This work was financially supported by the Natural Science Foundation of Zhejiang Province with Grant No. LZ20E080002, the National Natural Science Foundation of China with Grant Nos. 91851109 and 41911530193, and the Central Guide Local Science and Technology Development of **njiang Uygur Autonomous Region with Grant No. ZYYD2023B16.
Author information
Authors and Affiliations
Contributions
XZ performed the experiments and wrote the manuscript. HJL, LJ, and JW performed the experiments and analyzed the data. RH conceived the project and wrote the manuscript.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, X., Li, HJ., Jiang, L. et al. Synergy effects of Methylomonas koyamae and Hyphomicrobium methylovorum under methanethiol stress. Appl Microbiol Biotechnol 107, 3099–3111 (2023). https://doi.org/10.1007/s00253-023-12472-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-023-12472-w