Abstract
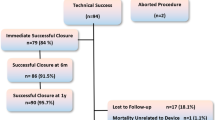
Nonsurgical closure of congenital ventricular septal defects (VSD) has become increasingly acceptable with the availability of different occlusion systems. Transcatheter device treatment is used for perimembranous and muscular defects. Atrio-ventricular block remains the most troublesome complication of device closure. The aim of this study was to describe our experience with closure of VSD using the Amplatzer Duct Occluder II (ADO II) as an “off-label” approach in children and adults. Between 2004 and 2012 transcatheter closure of 31 VSD (20 perimembranous, 10 muscular VSD and 1 ruptured sinus valsalva) with ADO II was undertaken in patients between 3 months and 55 years of age and with a body weight ranging from 4 to 105 kg in our institution. In 29 of 31 procedures, the defect was successfully closed (93.5 %) without any significant complications. No increase of aortic or tricuspid valve regurgitation was found in any after procedure. Small residual shunts were observed immediately after the device implantation, but disappeared during a median follow-up period of 38 months (0.4–63) in 27 of 31 patients. There was no incidence of AV block or other conductance abnormalities during implantation or follow-up. The ADO II device is safe and effective for transcatheter VSD closure, but this is still an “off-label” use. After long-term follow-up in a large number of patients this device may be approved for VSD closure in the future.





Similar content being viewed by others
References
Arora R, Trehan V, Kumar A (2003) Transcatheter closure of congenital ventricular septal defects. Experience with various devices. J Interv Cardiol 16:83–91
Bass JL, Gruenstein D (2012) Transcatheter closure of the perimembranous ventricular septal defect-preclinical trial of a new Amplatzer device. Catheter Cardiovasc Interv 79(7):1153–1160. doi:10.1002/ccd.23367 Epub 2011 Dec 12
Bass JL, Kalra GS, Arora R et al (2003) Initial human experience with the Amplatzer perimembranous ventricular septal occluder device. Cathet Cardiovasc Intervent 58:238–245
Benton JP, Barker KS (1992) Transcatheter closure of ventricular septal defect: anonsurgical approach to the care of the patient with acute ventricular septal rupture. Heart Lung 4:356–364
Bol-Raap G, Weerheim J, Kappetein AP, Witsenburg M, Bogers AJ (2003) Follow-up after surgical closure of congenital ventricular septal defects. Eur J Cardiothorac Surg 24:511–515
Butera G, Chessa M, Piazza L, Negura D, Micheletti A, Carminati M (2006) Percutaneous closure of ventricular septal defects. Expert Rev Cardiovasc Ther. 4(5):671–680 Review
Butera G, Carminati M, Chessa M et al (2006) Percutaneous closure of ventricular septal defects in children aged b 12: early and midterm results. Eur Heart J 27(23):2889–2895
Butera G, Carminati M, Chessa M, Piazza L, Micheletti A, Negura DG, Abella R, Giamberti A, Frigiola A (2007) Transcatheter closure of perimembranous ventricular septal defects: early and long-term results. J Am Coll Cardiol 50(12):1189–1195
Chessa M, Carminati M, Butera G et al (2002) Early and late complications associated with transcatheter occlusion of secundum atrial septal defect. J Am Coll Cardiol 39:1061–1065
Hijazi ZM, Hakim F, Al Fadley F, Abdelhamid J, Cao QL (2000) Transcatheter closure of single muscular ventricular septal defects using the Amplatzer muscular VSD occluder: initial results and technical considerations. Catheter Cardiovasc Intervent 49:167–172
Hijazi ZM, Hakim F, Haweleh AA et al (2002) Catheter closure of perimembranous ventricular septal defects using the new Amplatzer membranous VSD occluder: initial clinical experience. Cathet Cardiovasc Interv 56:508–515
Hobbins SM, Izukawa T, Radford DJ, Williams WG, Trusler GA (1979) Conduction disturbances after surgical correction of ventricular septal defect by the atrial approach. Br Heart J 41:289–293
Holzer R, Balzer D, Qi-Ling C, Lock K, Hijazi ZM, Amplatzer Muscular Ventricular Septal Defect Investigators (2004) Device closure of muscular ventricular septal defects using the Amplatzer muscular ventricular septal defect occluder. J Am Coll Cardiol 43:1257–1263
Holzer R, de Giovanni J, Walsh KP et al (2006) Transcatheter closure of perimem-branous ventricular septal defects using the amplatzer membranous VSD occluder: immediate and midterm results of an international registry. Catheter Cardiovasc Interv 68:620–628
Janorkar S, Goh T, Wilkinson J (1999) Transcatheter closure of ventricular septal defects using the Rashkind device: initial experience. Catheter Cardiovasc Interv 1:43–48
Kalra GS, Verma PK, Dhall A et al (1999) Transcatheter device closure of ventricular septal defects: Immediate results and intermediate-term follow-up. Am Heart J 2(Pt 1):339–344
Kidd L, Discroll DJ, Gersony WH et al (1993) Second natural history study of congenital heart defects: results of treatment of patients with ventricular septal defects. Circulation 87(Suppl I):138–151
Kirklin JW, Barrat-Boyes BG (1993) Ventricular septal defect. In: Kouchoukos NT, Blacksgowe EH, Doty DB, Hawley FL, Karp RB (eds) Cardiac surgery, 3rd edn. Churchill Livingstone, New York, pp 850–910
Koneti NR, Sreeram N, Penumatsa RR, Arramraj SK, Karunakar V, Trieschmann U (2012) Transcatheter retrograde closure of perimembranous ventricular septal defects in children with the Amplatzer duct occluder II device. J Am Coll Cardiol. 60(23):2421–2422. doi:10.1016/j.jacc.2012.08.1004 Epub 2012 Nov 7
Latiff HA, Alwi M, Kandhavel G et al (1999) Transcatheter closure of multiple muscular ventricular septal defects using Gianturco coils. Ann Thorac Surg 4:1400–1401
Lock JE, Block PC, McKay RG et al (1988) Transcatheter closure of ventricular septal defects. Circulation 2:361–368
Mavroudis C, Backer CL, Jacobs JP (2003) Ventricular septal defect. Pediatric cardiac surgery, 3rd edn. Mosby, Philadelphia, pp 298–320
Pass RH, Hijazi Z, Hsu DT, Lewis V, Hellenbrand WE (2004) Multicenter USA Amplatzer patent ductus arteriosus occlusion device trial. J Am Coll Cardiol 44:513–519
Pedra AC, Pedra SRF, Esteves CA et al (2004) Percutaneous closure of peri-membranous ventricular septal defects with the Amplatzer device: technical and morphological considerations. Cathet Cardiovasc Interv 61:403–410
Predescu D, Chaturvedi RR, Friedberg MK, Benson LN, Ozawa A, Lee KJ (2008) Complete heart block associated with device closure of perimembranous ventricular septal defects. J Thorac Cardiovasc Surg 136:1223–1228
Ramakrishnan S, Saxena A, Choudhary SK (2011) Residual VSD closure with an ADO II device in an infant. Congenit Heart Dis 6(1):60–63. doi:10.1111/j.1747-0803.2010.00468
Rigby ML, Redington AN (1994) Primary transcatheter umbrella closure of peri-membranous ventricular septal defect. Br Heart J 4:368–371
Zhao PJ, Yu ZQ, Gao W, Li F, Fu LJ, Liu TL, Li Y, Zhang YQ, Huang MR, Guo Y (2012) Efficacy of the transcatheter closure of perimembranous and muscular ventricular septal defects with the Amplatzer duct occluder II. Zhonghua **n Xue Guan Bing Za Zhi 40(10):817–820
Conflict of interest
Peter Ewert and Felix Berger are consultants for St. Jude Medical. All other authors have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kanaan, M., Ewert, P., Berger, F. et al. Follow-Up of Patients with Interventional Closure of Ventricular Septal Defects with Amplatzer Duct Occluder II. Pediatr Cardiol 36, 379–385 (2015). https://doi.org/10.1007/s00246-014-1017-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-014-1017-0