Abstract
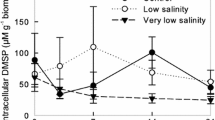
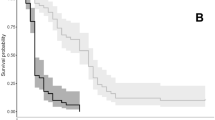
When emersed, intertidal seaweeds experience rapid changes in environmental conditions. On warm sunny days, seaweeds can desiccate soon after emersion. On rainy days or in areas with freshwater inputs, they may experience rapid drops in salinity. To determine whether one low tide’s exposure to environmental stress alters seaweed growth, palatability to herbivores, and dimethylsulfoniopropionate (DMSP) concentrations, we exposed Ulva fenestrata to desiccation and hyposaline conditions for 3 h in summer and winter. Two days later, summer desiccation decreased growth, consumption by Lacuna vincta, and DMSP by 71%, 83%, and 67%, relative to controls. Winter desiccation reduced DMSP by 19% and did not affect growth or L. vincta feeding. Summer hyposaline conditions increased growth by 75% but did not affect L. vincta grazing or DMSP, whereas winter hyposaline conditions reduced growth by 37%. After 2 days of exposure to L. vincta, seaweeds were allowed to recover for 7 days, then DMSP was measured. U. fenestrata desiccated in the summer did not survive. Average DMSP concentrations were 127% higher in grazed than ungrazed seaweed in the summer salinity experiment, providing evidence that grazing induces DMSP. No DMSP induction occurred in winter, possibly because grazing rates were much lower. In a follow-up summer desiccation experiment, maximum quantum yields (MQYs) and DMSP decreased to 12% and 14% of controls within an hour. After 7 days, MQY and DMSP concentrations recovered, but growth was 81–97% lower in seaweeds desiccated for 30–60 min, relative to controls. These results provide the first evidence for grazer-induced DMSP increases.




Similar content being viewed by others
Data availability
Data are available in the figshare repository (https://doi.org/https://doi.org/10.6084/m9.figshare.24769041).
References
Amsler CD (2001) Induced defenses in macroalgae: the herbivore makes a difference. J Phycol 37:353–356. https://doi.org/10.1046/j.1529-8817.2001.037003353.x
Anderson R, Berges J, Harrison P, Watanabe M (2005) Recipes for freshwater and seawater media. In: Anderson R (ed) Algal culturing techniques. Elsevier, Amsterdam, pp 429–532
Bell EC (1995) Environmental and morphological influences on thallus temperature and desiccation of the intertidal alga Mastocarpus papillatus Kützing. J Exp Mar Biol Ecol 191:29–55. https://doi.org/10.1016/0022-0981(95)00037-R
Beer S, Eshel A (1983) Photosynthesis of Ulva sp. I. Effects of desiccation when exposed to air. J Exp Mar Biol Ecol 70:91–97. https://doi.org/10.1016/0022-0981(83)90151-X
Bews E, Booher L, Polizzi T, Long C, Kim JH, Edwards MS (2021) Effects of salinity and nutrients on metabolism and growth of Ulva lactuca: implications for bioremediation of coastal watersheds. Mar Poll Bull 166:112199. https://doi.org/10.1016/j.marpolbul.2021.112199
Bischof K, Rautenberger R (2012) Seaweed responses to environmental stress: reactive oxygen and antioxidative strategies. In: Wiencke C, Bischof K (eds) Seaweed biology. Springer, Berlin, pp 109–132. https://doi.org/10.1007/978-3-642-28451-9_6
Bordeyne F, Migné A, Plus M, Davoult D (2020) Modelling the annual primary production of an intertidal brown algal community based on in situ measurements. Mar Ecol Prog Ser 656:95–107. https://doi.org/10.3354/meps13450
Brinkhuis BH (1977) Seasonal variations in salt-marsh macroalgae photosynthesis. I. Ascophyllum nodosum ecad scorpioides. Mar Biol 44:165–175. https://doi.org/10.1007/BF00386956
Broadbent AD, Jones GB, Jones RJ (2002) DMSP in corals and benthic algae from the Great Barrier Reef. Estuar Coastal Shelf Sci 55:547–555. https://doi.org/10.1006/ecss.2002.1021
Burdett HL, Aloisi E, Calosi P, Findlay HS, Widdicombe S, Hatton AD, Kamenos NA (2012) The effect of chronic and acute low pH on the intracellular DMSP production and epithelial cell morphology of red coralline algae. Mar Biol Res 8:756–763. https://doi.org/10.1080/17451000.2012.676189
Burdett HL, Hatton AD, Kamenos NA (2015a) Coralline algae as a globally significant pool of marine dimethylated sulfur. Global Biogeochem Cycles 29:1845–1853. https://doi.org/10.1002/2015GB005274
Burdett HL, Hatton AD, Kamenos NA (2015b) Effects of reduced salinity on the photosynthetic characteristics and intracellular DMSP concentrations of the red coralline alga, Lithothamnion glaciale. Mar Biol 162:1077–1085. https://doi.org/10.1007/s00227-015-2650-8
Burnaford JL, Nguyen TVT, Henderson SY, Van Alstyne KL (2021) Linking physiology to ecological function: environmental conditions affect performance and size of the intertidal kelp Hedophyllum sessile (Laminariales, Phaeophyceae). J Phycol 57:128–142. https://doi.org/10.1111/jpy.13071
Cetrulo GL, Hay ME (2000) Activated chemical defenses in tropical versus temperate seaweeds. Mar Ecol Prog Ser 207:243–253. https://doi.org/10.3354/meps207243
Choi TS, Kang EJ, Kim JH, Kim KY (2010) Effect of salinity on growth and nutrient uptake of Ulva pertusa (Chlorophyta) from an eelgrass bed. Algae 25:17–26
Coleman RA, Ramchunder SJ, Moody AJ, Foggo A (2007) An enzyme in snail saliva induces herbivore-resistance in a marine alga. Funct Ecol 21:101–106
Contreras-Porcia L, Lopez-Cristoffanini C, Maynard A, Kumar M (2017) Tolerance pathways to desiccation stress in seaweeds. In: Kumar M, Ralph P (eds) Systems biology of marine ecosystems. Springer, Heidelberg, pp 13–33. https://doi.org/10.1007/978-3-319-62094-7_2
Cronin G, Hay ME (1996) Susceptibility to herbivores depends on recent history of both the plant and animal. Ecology 77:1531–1543. https://doi.org/10.2307/2265549
Davison IR, Pearson GA (1996) Stress tolerance in intertidal seaweeds. J Phycol 32:197–211. https://doi.org/10.1007/978-3-319-62094-7_2
Davison IR, Johnson LE, Brawley SH (1993) Sublethal stress in the intertidal zone: tidal emersion inhibits photosynthesis and retards development in embryos of the brown alga Pelvetia fastigiata. Oecologia 96:483–492. https://doi.org/10.1007/BF00320505
Dring MJ (2005) Stress resistance and disease resistance in seaweeds: the role of reactive oxygen metabolism. Adv Bot Res 43:175–207. https://doi.org/10.1016/S0065-2296(05)43004-9
Gao G, Zhong Z, Zhou X, Xu J (2016) Changes in morphological plasticity of Ulva prolifera under different environmental conditions: a laboratory experiment. Harmful Algae 59:51–58. https://doi.org/10.1016/j.hal.2016.09.004
Gleadow RM, Møller BL (2014) Cyanogenic glycosides: synthesis, physiology, and phenotypic plasticity. Ann Rev Plant Biol 65:155–185. https://doi.org/10.1146/annurev-arplant-050213-040027
Hammerstrom K, Dethier MN, Duggins DO. (1998) Rapid phlorotannin induction and relaxation in five Washington kelps. Mar Ecol Prog Ser 165:293–305. https://www.jstor.org/stable/24827275
Hay ME (1981) The functional morphology of turf forming seaweeds: persistence in stressful marine habitats. Ecology 62:739–750. https://doi.org/10.2307/1937742
Heaven C, Scrosati R (2004) Feeding preference of Littorina snails (Gastropoda) for bleached and photosynthetic tissues of the seaweed Mazzaella parksii (Rhodophyta). Hydrobiologia 513:239–243. https://doi.org/10.1023/B:hydr.0000018298.94207.d2
Helmuth BS, Hofmann GE (2001) Microhabitats, thermal heterogeneity, and patterns of physiological stress in the rocky intertidal zone. Biol Bull 201:374–384. https://doi.org/10.2307/1543615
Horwitz R, Norin T, Watson SA, Pistevos JC, Beldade R, Hacquart S, Gattuso JP, Rodolfo-Metalpa R, Vidal-Dupiol J, Killen SS, Mills SC (2020) Near-future ocean warming and acidification alter foraging behaviour, locomotion, and metabolic rate in a keystone marine mollusc. Sci Rep 10:5461. https://doi.org/10.1038/s41598-020-62304-4
Howe GA, Schaller A (2008). Direct defenses in plants and their induction by wounding and insect herbivores. In: Schaller A (ed), Induced plant resistance to herbivory, Springer, Dordrecht, pp 7–29. https://doi.org/10.1007/978-1-4020-8182-8_1
Hunt LJ, Denny MW (2008) Desiccation protection and disruption: a trade-off for an intertidal marine alga. J Phycol 44:1164–1170. https://doi.org/10.1111/j.1529-8817.2008.00578.x
Hurd CL, Harrison PJ, Bischof K, Lobban CS (2014) Seaweed ecology and physiology. Cambridge University Press, Cambridge
Karsten U, Wiencke C, Kirst GO (1991a) Growth pattern and β-dimethylsulphoniopropionate (DMSP) content of green macroalgae at different irradiances. Mar Biol 108:151–155. https://doi.org/10.1007/BF01313483
Karsten U, Wiencke C, Kirst GO (1991b) The effect of salinity changes upon the physiology of eulittoral green macroalgae from Antarctica and southern Chile: II. Intracellular inorganic ions and organic compounds. J Exp Bot 42:1533–1539. https://doi.org/10.1016/S0176-1617(11)81313-1
Karsten U, Kirst GO, Wiencke C (1992) Dimethylsulphoniopropionate (DMSP) accumulation in green macroalgae from polar to temperate regions: interactive effects of light versus salinity and light versus temperature. Polar Biol 12:603–607. https://doi.org/10.1007/BF00236983
Karsten U, Kuck K, Daniel C, Wiencke C, Kirst GO (1994) A method for complete determination of dimethylsulfoniopropionate (DMSP) in marine macroalgae for different geographical regions. Phycologia 33:171–176. https://doi.org/10.2216/i0031-8884-33-3-171.1
Kiene RP, Nowinski B, Esson K, Preston C, Marin R, Birch J, Scholin C, Ryan J, Moran MA (2019) Unprecedented DMSP concentrations in a massive dinoflagellate bloom in Monterey Bay, CA. Geophys Res Lett 46:12279–12288. https://doi.org/10.1029/2019GL085496
Kinnby A, Toth GB, Pavia H (2021) Climate change increases susceptibility to grazers in a foundation seaweed. Front Mar Sci 8:688406. https://doi.org/10.3389/fmars.2021.688406
Kumar M, Kumari P, Reddy CRK, Jha B (2014) Salinity and desiccation induced oxidative stress acclimation in seaweeds. Adv Bot Res 71:91–123. https://doi.org/10.1016/B978-0-12-408062-1.00004-4
Lesser MP (2006) Oxidative stress in marine environments: biochemistry and physiological ecology. Annu Rev Physiol 68:253–278. https://doi.org/10.1146/annurev.physiol.68.040104.110001
Liss PS, Malin G, Turner SM, Holligan PM (1994) Dimethyl sulphide and Phaeocystis: a review. J Mar Syst 5:41–53. https://doi.org/10.1016/0924-7963(94)90015-9
Lu IF, Sung MS, Lee TM (2006) Salinity stress and hydrogen peroxide regulation of antioxidant defense system in Ulva fasciata. Mar Biol 150:1–15. https://doi.org/10.1007/s00227-006-0323-3
Luo MB, Liu F (2011) Salinity-induced oxidative stress and regulation of antioxidant defense system in the marine macroalga Ulva prolifera. J Exp Mar Biol Ecol 409:223–228. https://doi.org/10.1016/j.jembe.2011.08.023
Lyons DA, Scheibling RE, Van Alstyne KL (2010) Spatial and temporal variation in DMSP content in the invasive seaweed Codium fragile ssp. fragile: effects of temperature, light and grazing. Mar Ecol Prog Ser 417:51–61. https://doi.org/10.3354/meps08818
Mantri VA, Singh RP, Bijo AJ, Kumari P, Reddy CRK, Jha B (2011) Differential response of varying salinity and temperature on zoospore induction, regeneration and daily growth rate in Ulva fasciata (Chlorophyta, Ulvales). J Appl Phycol 23:243–250. https://doi.org/10.1007/s10811-010-9544-4
Molis M, Körner J, Ko YW, Kim JH, Wahl M (2006) Inducible responses in the brown seaweed Ecklonia cava: the role of grazer identity and season. J Ecol 94:243–249. https://doi.org/10.1111/j.1365-2745.2005.01058.x
Molis M, Körner J, Ko YW, Kim JH (2008) Specificity of inducible seaweed anti-herbivory defences depends on identity of macroalgae and herbivores. Mar Ecol Prog Ser 354:97–105. https://doi.org/10.3354/MEPS07255
Nelson TA, Nelson AV, Tjoelker M (2003) Seasonal and spatial patterns of “green tides” (ulvoid algal blooms) and related water quality parameters in the coastal waters of Washington State, USA. Bot Mar 46:263–275. https://doi.org/10.1515/BOT.2003.024
Nelson TA, Haberlin K, Nelson AV, Ribarich H, Hotchkiss R, Van Alstyne KL, Buckingham L, Simunds DJ (2008) Ecological and physiological controls of species composition in green macroalgal blooms. Ecology 89:1287–1298. https://doi.org/10.1890/07-0494.1
Nelson TA, Olson J, Imhoff L, Nelson AV (2010) Aerial exposure and desiccation tolerances are correlated to species composition in “green tides” of the Salish Sea (northeastern Pacific). Bot Mar 53:103–111. https://doi.org/10.1515/BOT.2010.020
Otte ML, Morris JT (1994) Dimethylsulphoniopropionate (DMSP) in Spartina alterniflora Loisel. Aquat Bot 48:239–259. https://doi.org/10.1016/0304-3770(94)90018-3
Padilla DK (1989) Algal structure defenses: form and calcification in resistance to tropical limpets. Ecology 70:835–884. https://doi.org/10.2307/1941352
Padilla DK (1998) Inducible phenotypic plasticity of the radula in Lacuna (Gastropoda: Littorinidae). Veliger 41:201–204
Padilla DK (2001) Food and environmental cues trigger an inducible offence. Evol Ecol Res 3:1–13
Padilla DK, Savedo MM (2013) A systematic review of phenotypic plasticity in marine invertebrate and plant systems. Adv Mar Biol 65:67–94. https://doi.org/10.1016/B978-0-12-410498-3.00002-1
Paine ER, Brewer EA, Schmid M, Diaz-Pulido G, Boyd PW, Hurd CL (2023) Strong seasonal patterns of DOC release by a temperate seaweed community: implications for the coastal ocean carbon cycle. J Phycol 59:723–750. https://doi.org/10.1111/jpy.13352
Paul VJ, Van Alstyne KL (1992) Activation of chemical defenses in the tropical green algae Halimeda spp. J Exp Mar Biol Ecol 160:191–203. https://doi.org/10.1016/0022-0981(92)90237-5
Paul VJ, Arthur KE, Ritson-Williams R, Ross C, Sharp K (2007) Chemical defenses: from compounds to communities. Biol Bull 213:226–251. https://doi.org/10.2307/25066642
Pavia H, Toth GB (2000) Inducible chemical resistance to herbivory in the brown seaweed Ascophyllum nodosum. Ecology 81:3212–3225. https://doi.org/10.1890/0012-9658(2000)081[3212:ICRTHI]2.0.CO;2
Pohnert G (2004) Chemical defense strategies of marine organisms. Top Curr Chem 239:179–219. https://doi.org/10.1007/b95453
Raffaelli D, Hawkins S (1996) The shore environment: major gradients. In: Raffaelli D, Hawkins S (eds) Intertidal ecology. Springer, Dordrecht, pp 1–35
Renaud PE, Hay ME, Schmitt TM (1990) Interactions of plant stress and herbivory: intraspecific variation in the susceptibility of a palatable versus an unpalatable seaweed to sea urchin grazing. Oecologia 82:217–226. https://doi.org/10.1007/BF00323538
Román M, Román S, Vázquez E, Troncoso J, Olabarria C (2020) Heatwaves during low tide are critical for the physiological performance of intertidal macroalgae under global warming scenarios. Sci Rep 10:21408. https://doi.org/10.1038/s41598-020-78526-5
Ross C, Van Alstyne KL (2007) Intraspecific variation in stress-induced hydrogen peroxide scavenging by the ulvoid macroalga Ulva lactuca. J Phycol 43:466–474. https://doi.org/10.1111/j.1529-8817.2007.00346.x
Roussel S, Poitevin P, Day R, Le Grand F, Stiger-Pouvreau V, Leblanc C, Huchette S (2020) Haliotis tuberculata, a generalist marine herbivore that prefers a mixed diet, but with consistent individual foraging activity. Ethology 126:716–726. https://doi.org/10.1111/eth.13020
Schagerl M, Möstl M (2011) Drought stress, rain and recovery of the intertidal seaweed Fucus spiralis. Mar Biol 158:2471–2479. https://doi.org/10.1007/s00227-011-1748-x
Schonbeck M, Norton TA (1978) Factors controlling the upper limits of fucoid algae on the shore. J Exp Mar Biol Ecol 31:303–313. https://doi.org/10.1016/0022-0981(78)90065-5
Simonson EJ, Scheibling RE, Metaxas A (2015) Kelp in hot water: I. Warming seawater temperature induces weakening and loss of kelp tissue. Mar Ecol Prog Ser 537:89–104. https://doi.org/10.3354/meps11438
Sotka EE, Giddens H (2009) Seawater temperature alters feeding discrimination by cold-temperate but not subtropical individuals of an ectothermic herbivore. Biol Bull 216:75–84. https://doi.org/10.1086/BBLv216n1p75
Stillman JH (2019) Heat waves, the new normal: summertime temperature extremes will impact animals, ecosystems, and human communities. Physiology 34:86–100. https://doi.org/10.1152/physiol.00040.2018
Sunda WK, Kieber DJ, Kiene RP, Huntsman S (2002) An antioxidant function for DMSP and DMS in marine algae. Nature 418:317–320. https://doi.org/10.1038/nature00851
Thompson RC, Roberts MF, Norton TA, Hawkins SJ (2000) Feast or famine for intertidal grazing molluscs: a mis-match between seasonal variations in grazing intensity and the abundance of microbial resources. Hydrobiologia 440:357–367. https://doi.org/10.1023/A:1004116505004
Van Alstyne KL (1988) Herbivore grazing increases polyphenolic defenses in the intertidal brown alga Fucus distichus. Ecology 69:655–663. https://doi.org/10.2307/1941014
Van Alstyne KL (2016) Seasonal changes in isotopic signatures and nitrogen content of the bloom-forming seaweed Ulva lactuca in a northeastern Pacific embayment. Mar Poll Bull 103:186–194. https://doi.org/10.1016/j.marpolbul.2015.12.020
Van Alstyne KL (2018) Seawater nitrogen concentration and light independently alter performance, growth, and resource allocation in the bloom-forming seaweeds Ulva lactuca and Ulvaria obscura (Chlorophyta). Harmful Algae 78:27–35. https://doi.org/10.1016/j.hal.2018.07.005
Van Alstyne KL, Houser LT (2003) Dimethylsulfide release during macroinvertebrate grazing and its role as an activated chemical defense. Mar Ecol Prog Ser 250:175–181. https://doi.org/10.3354/meps250175
Van Alstyne KL, Wolfe GV, Freidenburg TL, Neill A, Hicken C (2001) Activated defense systems in marine macroalgae: evidence for an ecological role for DMSP cleavage. Mar Ecol Prog Ser 213:53–65. https://doi.org/10.3354/MEPS213053
Van Alstyne KL, Pelletreau KN, Rosario K (2003) The effects of salinity on dimethylsulfoniopropionate production in the green alga Ulva fenestrata Postels et Ruprecht (Chlorophyta). Bot Mar 46:350–356. https://doi.org/10.1515/BOT.2003.033
Van Alstyne KL, Koellermeier L, Nelson T (2007) Spatial variation in dimethylsulfoniopropionate (DMSP) production in Ulva lactuca (Chlorophyta) from the Northeast Pacific. Mar Biol 150:1127–1135. https://doi.org/10.1007/s00227-006-0448-4
Van Alstyne KL, Pelletreau KN, Kirby A (2009) Nutritional preferences override chemical defenses in determining food choice by a generalist herbivore, Littorina sitkana. J Exp Mar Biol Ecol 379:85–91. https://doi.org/10.1016/j.jembe.2009.08.002
Van Alstyne KL, Gifford S-A, Dohman J, Savedo M (2015) Effects of environmental changes, tissue types, and reproduction on emissions of dimethyl sulfide from seaweeds that form green tides. Environ Chem 13:220–230. https://doi.org/10.1071/EN15037
Van Alstyne KL, Sutton L, Gifford S-A (2020) Inducible versus constitutive antioxidant defenses along an environmental stress gradient. Mar Ecol Prog Ser 640:107–115. https://doi.org/10.3354/meps13263
Van Alstyne KL, Butler JM, Smith N (2023) Eavesdrop** seaweeds: evidence for dimethylsulfoniopropionate (DMSP) induction by airborne dimethyl sulfide (DMS) in the green alga Ulva fenestrata. Sci Rep 13:4298. https://doi.org/10.1038/s41598-023-30881-9
Van Alstyne KL, Haug C, Lee H-Y, McKeen G, Schwarck N, Strom M (2024) Water quality data collected at the Shannon Point Marine Center, Anacortes, Washington, USA: 1974–2023. https://doi.org/10.6084/m9.figshare.25206959
van Hees DH, Van Alstyne KL (2013) Effects of emersion, temperature, dopamine, and hypoxia on the accumulation of extracellular oxidants surrounding the bloom-forming seaweeds Ulva lactuca and Ulvaria obscura. J Exp Mar Biol Ecol 448:207–213. https://doi.org/10.1016/j.jembe.2013.07.013
Wolfe GV, Sherr EB, Sherr BF (1994) Release and consumption of DMSP from Emiliania huxleyi during grazing by Oxyrrhis marina. Mar Ecol Prog Ser 11:111–119. https://doi.org/10.3354/meps11111
Wolfe GV, Levasseur M, Cantin G, Michaud S (2000) DMSP and DMS dynamics and microzooplankton grazing in the Labrador Sea: application of the dilution technique. Deep Sea Res Part I 47:2243–2264. https://doi.org/10.1016/S0967-0637(00)00028-5
Wolfe GV, Strom SL, Holmes JL, Radzio T, Olson MB (2002) Dimethylsulfoniopropionate cleavage by marine phytoplankton in response to mechanical, chemical, or dark stress. J Phycol 38:948–960. https://doi.org/10.1046/j.1529-8817.2002.t01-1-01100.x
Acknowledgements
We thank S.-A. Gifford, C. Kohnert, and T. Keates for assistance with laboratory experiments, B. Bingham for advice on statistical analyses, and the Shannon Point Marine Center for providing temperature data. We also thank two anonymous reviewers for their comments on the manuscript. This study was supported by National Science Foundation grants OCE-041372 to the Shannon Point Marine Center and IOS-1118944 to KLV.
Funding
This study was supported by National Science Foundation grants OCE-041372 to the Shannon Point Marine Center and IOS-1118944 to KLV.
Author information
Authors and Affiliations
Contributions
Both authors contributed to the study conception and design. NB conducted the experiments and collected the data with help from KLVA. KLVA analyzed the data and wrote the first draft of the manuscript. NB reviewed and edited the draft and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Compliance with ethical standards
The authors have no relevant financial or non-financial interests to disclose. All applicable international, national and/or institutional guidelines for sampling, care and experimental use of organisms for the study have been followed and all necessary permits for collecting organisms were obtained.
Additional information
Responsible Editor: K. Bischof.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Van Alstyne, K.L., Borgen, N. Seasonal effects of short-term stress on susceptibility to herbivores and DMSP induction in the intertidal green alga Ulva fenestrata. Mar Biol 171, 86 (2024). https://doi.org/10.1007/s00227-024-04413-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-024-04413-6