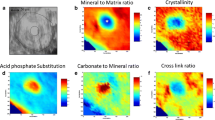
Abstract
Enzymatic cross-linking of the bone collagen is important to resist to crack growth and to increased flexural strength. In the present study, we proposed a new method for assessment of enzymatic cross-link based on Fourier transform infrared (FTIR) microspectroscopy that takes into account secondary structure of type I collagen. Briefly, femurs were collected from sham or ovariectomized mice and subjected either to high-performance liquid chromatography—mass spectrometry or embedded in polymethylmethacrylate, cut and analyzed by FTIR microspectroscopy. FTIR acquisition was recorded before and after ultraviolet (UV) exposure or acid treatment. In addition, femurs from a second animal study were used to compare gene expression of Plod2 and Lox enzymes and enzymatic cross-links determined by FTIR microspectroscopy. We evidenced here that intensities and areas of subbands located at ~1660, ~1680, and ~1690 cm−1 were positively and significantly associated with the concentration of pyridinoline (PYD), deoxypyridinoline, or immature dihydroxylysinonorleucine/hydroxylysinonorleucine cross-links. Seventy-two hours exposure to UV light significantly reduced by ~86% and ~89% the intensity and area of the ~1660 cm−1 subband. Similarly, 24 h of acid treatment significantly reduced by 78% and 76% the intensity and area of the ~1690 cm−1 subband. Plod2 and Lox expression were also positively associated to the signal of the ~1660 and ~1690 cm−1 subbands. In conclusion, our study provided a new method for decomposing the amide I envelope of bone section that positively correlates with PYD and immature collagen cross-links. This method allows for investigation of tissue distribution of enzymatic cross-links in bone section.





Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Granke M, Does MD, Nyman JS (2015) The role of water compartments in the material properties of cortical bone. Calcif Tissue Int 97:292–307
Yamauchi M, Sricholpech M (2012) Lysine post-translational modifications of collagen. Essays Biochem 52:113–133
McNerny EM, Gong B, Morris MD, Kohn DH (2015) Bone fracture toughness and strength correlate with collagen cross-link maturity in a dose-controlled lathyrism mouse model. J Bone Miner Res 30:455–464
Eyre DR, Paz MA, Gallop PM (1984) Cross-linking in collagen and elastin. Annu Rev Biochem 53:717–748
Uzawa K, Grzesik WJ, Nishiura T, Kuznetsov SA, Robey PG, Brenner DA, Yamauchi M (1999) Differential expression of human lysyl hydroxylase genes, lysine hydroxylation, and cross-linking of type I collagen during osteoblastic differentiation in vitro. J Bone Miner Res 14:1272–1280
Saito T, Terajima M, Taga Y, Hayashi F, Oshima S, Kasamatsu A, Okubo Y, Ito C, Toshimori K, Sunohara M, Tanzawa H, Uzawa K, Yamauchi M (2022) Decrease of lysyl hydroxylase 2 activity causes abnormal collagen molecular phenotypes, defective mineralization and compromised mechanical properties of bone. Bone 154:116242
Trackman PC (2016) Enzymatic and non-enzymatic functions of the lysyl oxidase family in bone. Matrix Biol 52–54:7–18
Yamauchi M, Katz EP, Otsubo K, Teraoka K, Mechanic GL (1989) Cross-linking and stereospecific structure of collagen in mineralized and nonmineralized skeletal tissues. Connect Tissue Res 21:159–167
Bailey AJ, Peach CM, Fowler LJ (1970) Chemistry of the collagen cross-links. Isolation and characterization of two intermediate intermolecular cross-links in collagen. Biochem J 117:819–831
Bielajew BJ, Hu JC, Athanasiou KA (2020) Collagen: quantification, biomechanics, and role of minor subtypes in cartilage. Nat Rev Mater 5:730–747
Wang ZX, Lloyd AA, Burket JC, Gourion-Arsiquaud S, Donnelly E (2016) Altered distributions of bone tissue mineral and collagen properties in women with fragility fractures. Bone 84:237–244
Paschalis EP, Verdelis K, Doty SB, Boskey AL, Mendelsohn R, Yamauchi M (2001) Spectroscopic characterization of collagen cross-links in bone. J Bone Miner Res 16:1821–1828
Paschalis EP, Gamsjaeger S, Tatakis DN, Hassler N, Robins SP, Klaushofer K (2015) Fourier transform Infrared spectroscopic characterization of mineralizing type I collagen enzymatic trivalent cross-links. Calcif Tissue Int 96:18–29
Wolfel EM, Schmidt FN, Vom Scheidt A, Siebels AK, Wulff B, Mushumba H, Ondruschka B, Puschel K, Scheijen J, Schalkwijk CG, Vettorazzi E, Jahn-Rickert K, Gludovatz B, Schaible E, Amling M, Rauner M, Hofbauer LC, Zimmermann EA, Busse B (2022) Dimorphic mechanisms of fragility in diabetes mellitus: the role of reduced collagen fibril deformation. J Bone Miner Res 37:2259–2276
Schmidt FN, Zimmermann EA, Campbell GM, Sroga GE, Puschel K, Amling M, Tang SY, Vashishth D, Busse B (2017) Assessment of collagen quality associated with non-enzymatic cross-links in human bone using Fourier-transform infrared imaging. Bone 97:243–251
Farlay D, Duclos ME, Gineyts E, Bertholon C, Viguet-Carrin S, Nallala J, Sockalingum GD, Bertrand D, Roger T, Hartmann DJ, Chapurlat R, Boivin G (2011) The ratio 1660/1690 cm(-1) measured by infrared microspectroscopy is not specific of enzymatic collagen cross-links in bone tissue. PLoS ONE 6:e28736
Belbachir K, Noreen R, Gouspillou G, Petibois C (2009) Collagen types analysis and differentiation by FTIR spectroscopy. Anal Bioanal Chem 395:829–837
Barth A (2007) Infrared spectroscopy of proteins. Biochim Biophys Acta 1767:1073–1101
Siegel RC, Fu JC (1976) Collagen cross-linking. Purification and substrate specificity of lysyl oxidase. J Biol Chem 251:5779–5785
van der Slot AJ, Zuurmond AM, Bardoel AF, Wijmenga C, Pruijs HE, Sillence DO, Brinckmann J, Abraham DJ, Black CM, Verzijl N, DeGroot J, Hanemaaijer R, TeKoppele JM, Huizinga TW, Bank RA (2003) Identification of PLOD2 as telopeptide lysyl hydroxylase, an important enzyme in fibrosis. J Biol Chem 278:40967–40972
Kelly NH, Schimenti JC, Patrick Ross F, van der Meulen MC (2014) A method for isolating high quality RNA from mouse cortical and cancellous bone. Bone 68:1–5
Bassan P, Sachdeva A, Kohler A, Hughes C, Henderson A, Boyle J, Shanks JH, Brown M, Clarke NW, Gardner P (2012) FTIR microscopy of biological cells and tissue: data analysis using resonant Mie scattering (RMieS) EMSC algorithm. Analyst 137:1370–1377
Bailey AJ (1968) Intermediate labile intermolecular crosslinks in collagen fibres. Biochim Biophys Acta 160:447–453
Gineyts E, Borel O, Chapurlat R, Garnero P (2010) Quantification of immature and mature collagen crosslinks by liquid chromatography-electrospray ionization mass spectrometry in connective tissues. J Chromatogr B Analyt Technol Biomed Life Sci 878:1449–1454
Gourion-Arsiquaud S, Faibish D, Myers E, Spevak L, Compston J, Hodsman A, Shane E, Recker RR, Boskey ER, Boskey AL (2009) Use of FTIR spectroscopic imaging to identify parameters associated with fragility fracture. J Bone Miner Res 24:1565–1571
Paschalis EP, Tatakis DN, Robins S, Fratzl P, Manjubala I, Zoehrer R, Gamsjaeger S, Buchinger B, Roschger A, Phipps R, Boskey AL, Dall’Ara E, Varga P, Zysset P, Klaushofer K, Roschger P (2011) Lathyrism-induced alterations in collagen cross-links influence the mechanical properties of bone material without affecting the mineral. Bone 49:1232–1241
Smolina M, Goormaghtigh E (2018) Gene expression data and FTIR spectra provide a similar phenotypic description of breast cancer cell lines in 2D and 3D cultures. Analyst 143:2520–2530
Berteau JP, Gineyts E, Pithioux M, Baron C, Boivin G, Lasaygues P, Chabrand P, Follet H (2015) Ratio between mature and immature enzymatic cross-links correlates with post-yield cortical bone behavior: An insight into greenstick fractures of the child fibula. Bone 79:190–195
Depalle B, Duarte AG, Fiedler IAK, Pujo-Menjouet L, Buehler MJ, Berteau JP (2018) The different distribution of enzymatic collagen cross-links found in adult and children bone result in different mechanical behavior of collagen. Bone 110:107–114
Acevedo C, Bale H, Gludovatz B, Wat A, Tang SY, Wang M, Busse B, Zimmermann EA, Schaible E, Allen MR, Burr DB, Ritchie RO (2015) Alendronate treatment alters bone tissues at multiple structural levels in healthy canine cortical bone. Bone 81:352–363
Gobron B, Couchot M, Irwin N, Legrand E, Bouvard B, Mabilleau G (2023) Development of a first-in-class unimolecular dual GIP/GLP-2 analogue, GL-0001, for the treatment of bone fragility. J Bone Miner Res 38:733–748
Acknowledgements
We are thankful to Prof Peter Gardner (University of Manchester) for supplying the Mie scattering correction routine for Matlab. We are also grateful to the institutional animal lab SCAHU, SFR ICAT 4208, Univ Angers, for their help with animal care.
Funding
This project was funded by an institutional grant from the University of Angers.
Author information
Authors and Affiliations
Contributions
AM: Investigation, Formal analysis and GM: Conceptualization, Investigation, Formal analysis, Writing—Review & Editing, Supervision, Funding acquisition, Data curation.
Corresponding author
Ethics declarations
Competing interest
Aleksandra Mieczkowska and Guillaume Mabilleau have no competing interests to declare that are relevant to the content of this article.
Ethical Approval
All procedures were carried out in accordance with the European Union Directive 2010/63/EU for animal experiments and were approved by the regional ethical committee for animal use (authorization CEEA-PdL06-01740.01).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mieczkowska, A., Mabilleau, G. Validation of Fourier Transform Infrared Microspectroscopy for the Evaluation of Enzymatic Cross-Linking of Bone Collagen. Calcif Tissue Int 113, 344–353 (2023). https://doi.org/10.1007/s00223-023-01105-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-023-01105-z