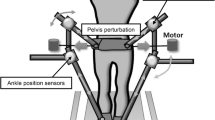
Abstract
The purpose of this study was to determine whether the gradual versus abrupt adaptation to lateral pelvis assistance force improves weight shift toward the paretic side and enhance forced use of the paretic leg during walking. Sixteen individuals who had sustained a hemispheric stroke participated in two experimental sessions, which consisted of (1) treadmill walking with the application of lateral pelvis assistance force (gradual vs. abrupt condition) and (2) overground walking. In the “gradual” condition, during treadmill walking, the assistance force was gradually increased from 0 to 100% of the predetermined force step by step. In the abrupt condition, the force was applied at 100% of the predetermined force throughout treadmill walking. Participants exhibited significant improvements in hip abductor and adductor, ankle dorsiflexor, and knee extensor muscle activities, weight shift toward the paretic side, and overground walking speed in the gradual condition (P < 0.05), but showed no significant changes in the abrupt condition (P > 0.20). Changes in weight shift toward the paretic side were statistically different between conditions (P < 0.001), although changes in muscle activities were not (P > 0.11). In the gradual condition, the error amplitude was proportional to the improvement in weight shift during the late post-adaptation (R2 = 0.32, P = 0.03), but not in the abrupt condition (R2 = 0.001, P = 0.93). In conclusion, the “gradual adaptation” inducing “small errors” during constraint-induced walking may improve weight shift and enhance forced use of the paretic leg in individuals post-stroke. Applying gradual pelvis assistance force during walking may be used as an intervention strategy to improve walking in individuals post-stroke.






Similar content being viewed by others
Availability of data and material
Data can be provided at request.
Code availability
Customized LabVIEW and MATLAB codes will not be shared for intellectual property protection.
References
Alcantara CC, Charalambous CC, Morton SM, Russo TL, Reisman DS (2018) Different error size during locomotor adaptation affects transfer to overground walking poststroke. Neurorehabil Neural Repair 32:1020–1030. https://doi.org/10.1177/1545968318809921
Berniker M, Kording K (2008) Estimating the sources of motor errors for adaptation and generalization. Nat Neurosci 11:1454–1461. https://doi.org/10.1038/nn.2229
Bohannon RW, Andrews AW, Smith MB (1988) Rehabilitation goals of patients with hemiplegia. Int J Rehabil Res 11:3
Chu VW, Hornby TG, Schmit BD (2015) Perception of lower extremity loads in stroke survivors. Clin Neurophysiol 126:372–381. https://doi.org/10.1016/j.clinph.2014.06.047
Diedrichsen J, White O, Newman D, Lally N (2010) Use-dependent and error-based learning of motor behaviors. J Neurosci 30:5159–5166
Fine MS, Thoroughman KA (2007) Trial-by-trial transformation of error into sensorimotor adaptation changes with environmental dynamics. J Neurophysiol 98:1392–1404
Floor-Westerdijk MJ, Schepers HM, Veltink PH, van Asseldonk EH, Buurke JH (2012) Use of inertial sensors for ambulatory assessment of center-of-mass displacements during walking. IEEE Trans Biomed Eng 59:2080–2084. https://doi.org/10.1109/TBME.2012.2197211
Folstein MF, Robins LN, Helzer JE (1983) The mini-mental state examination. Arch Gen Psychiatry 40:812–812
Gentile A (1998) Movement science: implicit and explicit processes during acquisition of functional skills. Scand J Occup Ther 5:10. https://doi.org/10.3109/11038129809035723
Green D, Bunday K, Bowen J, Carter T, Bronstein A (2010) What does autonomic arousal tell us about locomotor learning? Neuroscience 170:42–53
Hogan N (1984) Adaptive control of mechanical impedance by coactivation of antagonist muscles. IEEE Trans Auto Control 29:9
Hsu CJ, Kim J, Roth EJ, Rymer WZ, Wu M (2017a) Forced use of the paretic leg induced by a constraint force applied to the nonparetic leg in individuals poststroke during walking. Neurorehabil Neural Repair 31:1042–1052. https://doi.org/10.1177/1545968317740972
Hsu CJ, Kim J, Tang R, Roth EJ, Rymer WZ, Wu M (2017b) Applying a pelvic corrective force induces forced use of the paretic leg and improves paretic leg EMG activities of individuals post-stroke during treadmill walking. Clin Neurophysiol 128:1915–1922. https://doi.org/10.1016/j.clinph.2017.07.409
Hsu CJ, Kim J, Roth EJ, Rymer WZ, Wu M (2019) Use of pelvic corrective force with visual feedback improves paretic leg muscle activities and gait performance after stroke. IEEE Trans Neural Syst Rehabil Eng 27:2353–2360. https://doi.org/10.1109/TNSRE.2019.2950226
Huang VS, Shadmehr R (2009) Persistence of motor memories reflects statistics of the learning event. J Neurophysiol 102:931–940
Kal E, Winters M, van der Kamp J, Houdijk H, Groet E, van Bennekom C, Scherder E (2016) Is implicit motor learning preserved after stroke? A systematic review with meta-analysis. PLoS ONE 11:e0166376. https://doi.org/10.1371/journal.pone.0166376
Kim M, Lee HH, Lee J (2020) Does isolated somatosensory impairment affect the balance and ambulation of patients with supratentorial stroke after the acute phase? J Clin Neurosci 74:109–114. https://doi.org/10.1016/j.jocn.2020.01.084
Kluzik J, Diedrichsen J, Shadmehr R, Bastian AJ (2008) Reach adaptation: what determines whether we learn an internal model of the tool or adapt the model of our arm? J Neurophysiol 100:1455–1464
Lin JT, Hsu CJ, Dee W, Chen D, Rymer WZ, Wu M (2019) Motor adaptation to weight shifting assistance transfers to overground walking in people with spinal cord injury. PM&R 11:1200–1209. https://doi.org/10.1002/pmrj.12132
Malone LA, Bastian AJ (2014) Spatial and temporal asymmetries in gait predict split-belt adaptation behavior in stroke. Neurorehabil Neural Repair 28:230–240. https://doi.org/10.1177/1545968313505912
Mehrholz J, Thomas S, Elsner B (2017) Treadmill training and body weight support for walking after stroke. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD002840.pub4
Michielsen ME, Selles RW, Stam HJ, Ribbers GM, Bussmann JB (2012) Quantifying nonuse in chronic stroke patients: a study into paretic, nonparetic, and bimanual upper-limb use in daily life. Arch Phys Med Rehabil 93:1975–1981. https://doi.org/10.1016/j.apmr.2012.03.016
Morton SM, Bastian AJ (2004) Prism adaptation during walking generalizes to reaching and requires the cerebellum. J Neurophysiol 92:2497–2509. https://doi.org/10.1152/jn.00129.2004
O’lney SJ, Richards C (1996) Hemiparetic gait following stroke. Part I: characteristics. Gait Posture 4:13. https://doi.org/10.1016/0966-6362(96)01063-6
Park SH, Lin JT, Dee W, Hsu CJ, Roth EJ, Rymer WZ, Wu M (2020) Targeted Pelvic Constraint Force Induces Enhanced Use of the Paretic Leg During Walking in Persons Post-Stroke. IEEE Trans Neural Syst Rehabil Eng 28:2184–2193. https://doi.org/10.1109/TNSRE.2020.3018397
Patrick SK, Musselman KE, Ta**o J, Ou HC, Bastian AJ, Yang JF (2014) Prior experience but not size of error improves motor learning on the split-belt treadmill in young children. PLoS ONE 9:e93349. https://doi.org/10.1371/journal.pone.0093349
Raja B, Neptune RR, Kautz SA (2012) Coordination of the non-paretic leg during hemiparetic gait: expected and novel compensatory patterns. Clin Biomech 27:1023–1030
Reisman DS, Wityk R, Silver K, Bastian AJ (2009) Split-belt treadmill adaptation transfers to overground walking in persons poststroke. Neurorehabil Neural Repair 23:735–744. https://doi.org/10.1177/1545968309332880
Robinson FR, Noto CT, Bevans SE (2003) Effect of visual error size on saccade adaptation in monkey. J Neurophysiol 90:1235–1244
Roelker SA, Bowden MG, Kautz SA, Neptune RR (2019) Paretic propulsion as a measure of walking performance and functional motor recovery post-stroke: a review. Gait Posture 68:6–14. https://doi.org/10.1016/j.gaitpost.2018.10.027
Roerdink M, Geurts AC, de Haart M, Beek PJ (2009) On the relative contribution of the paretic leg to the control of posture after stroke. Neurorehabil Neural Repair 23:267–274
Savin DN, Morton SM (2008) Asymmetric generalization between the arm and leg following prism-induced visuomotor adaptation. Exp Brain Res 186:175–182. https://doi.org/10.1007/s00221-007-1220-9
Savin DN, Morton SM, Whitall J (2014) Generalization of improved step length symmetry from treadmill to overground walking in persons with stroke and hemiparesis. Clin Neurophysiol 125:1012–1020. https://doi.org/10.1016/j.clinph.2013.10.044
Shadmehr R, Holcomb HH (1997) Neural correlates of motor memory consolidation. Science 277:821–825. https://doi.org/10.1126/science.277.5327.821
Smith MA, Ghazizadeh A, Shadmehr R (2006) Interacting adaptive processes with different timescales underlie short-term motor learning. PLoS Biol 4:e179. https://doi.org/10.1371/journal.pbio.0040179
Tang R, Kim J, Gaebler-Spira DJ, Wu M (2019) Gradual increase of perturbation load induces a longer retention of locomotor adaptation in children with cerebral palsy. Hum Mov Sci 63:20–33. https://doi.org/10.1016/j.humov.2018.11.006
Taub E, Crago JE, Burgio LD, Groomes TE, Cook EW 3rd, DeLuca SC, Miller NE (1994) An operant approach to rehabilitation medicine: overcoming learned nonuse by sha**. J Exp Anal Behav 61:281–293. https://doi.org/10.1901/jeab.1994.61-281
Taub E, Uswatte G, King DK, Morris D, Crago JE, Chatterjee A (2006) A placebo-controlled trial of constraint-induced movement therapy for upper extremity after stroke. Stroke 37:1045–1049
Torres-Oviedo G, Bastian AJ (2012) Natural error patterns enable transfer of motor learning to novel contexts. J Neurophysiol 107:346–356
Wei K, Kording K (2009) Relevance of error: what drives motor adaptation? J Neurophysiol 101:655–664
Wei Y, Patton J, Bajaj P, Scheidt R (2005) A real-time haptic/graphic demonstration of how error augmentation can enhance learning. In: Proceedings of the 2005 IEEE international conference on robotics and automation. IEEE, pp 4406–4411
Winter DA, MacKinnon CD, Ruder GK, Wieman C (1993) An integrated EMG/biomechanical model of upper body balance and posture during human gait. Prog Brain Res 97:359–367. https://doi.org/10.1016/s0079-6123(08)62295-5
Wolf SL, Winstein CJ, Miller JP et al (2008) The EXCITE trial: retention of improved upper extremity function among stroke survivors receiving CI movement therapy. Lancet Neurol 7:33
Wu M, Hornby TG, Landry JM, Roth H, Schmit BD (2011) A cable-driven locomotor training system for restoration of gait in human SCI. Gait Posture 33:256–260. https://doi.org/10.1016/j.gaitpost.2010.11.016
Wu M, Hsu CJ, Kim J (2019) Forced use of paretic leg induced by constraining the non-paretic leg leads to motor learning in individuals post-stroke. Exp Brain Res 237:2691–2703. https://doi.org/10.1007/s00221-019-05624-w
Yang Y-R, Wang R-Y, Lin K-H, Chu M-Y, Chan R-C (2006) Task-oriented progressive resistance strength training improves muscle strength and functional performance in individuals with stroke. Clin Rehabil 20:860–870
Yen S-C, Schmit BD, Landry JM, Roth H, Wu M (2012) Locomotor adaptation to resistance during treadmill training transfers to overground walking in human SCI. Exp Brain Res 216:473–482
Funding
This work was supported by the National Institute of Health (R01HD082216 to Ming Wu).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Additional information
Communicated by Francesco Lacquaniti.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Park, S.H., Hsu, CJ., Dee, W. et al. Gradual adaptation to pelvis perturbation during walking reinforces motor learning of weight shift toward the paretic side in individuals post-stroke. Exp Brain Res 239, 1701–1713 (2021). https://doi.org/10.1007/s00221-021-06092-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-021-06092-x