Abstract
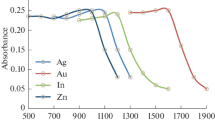
The atomization processes involved in the Electrothermal Atomization-Atomic Absorption Spectrometric (ETA-AAS) determination of Ag, Be, Cd, Li, Na, Sn and Zn in the presence of an uranium-plutonium matrix containing 25% Pu have been investigated. The significant fall in the absorbance signal for Ag, Cd, Na and Zn in an uranium matrix and its restoration in the presence of plutonium is probably correlated with the change in the partial pressure of oxygen released from the matrix at/below the signal appearance temperature (Tapp). In case of Li and Sn, the signal remains unaffected irrespective of the U-Pu matrix which might be due to a high Tapp for these analytes. Regarding Be, the steep suppression of the signal in the presence of the plutonium matrix compared to an uranium matrix can be ascribed to the formation of stable Pu-Be compounds (PuBe13). Based on these studies, analytical procedures have been developed for the direct determination of nanogram amounts of these analytes in an U-Pu matrix with an average precision of 9% RSD.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Goyal, N., Purohit, P.J., Page, A.G. et al. Atomization mechanism and determination of Ag, Be, Cd, Li, Na, Sn and Zn in uranium-plutonium matrices by ETA-AAS. Fresenius J Anal Chem 354, 311–315 (1996). https://doi.org/10.1007/s0021663540311
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s0021663540311