Abstract
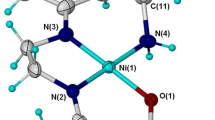
During the study of electrodeposition of nickel, natural coumarins were used as brighteners and caused an improvement in the surface of deposited plates. We thought about the mechanism by which the coumarins adhere to nickel and the most probable chemical mechanism would be an organometallic complex formation between these coumarins and the transition metal. In this study, the formation of a highly stable complex between a coumarin derivative, acetylumbelliferone and nickel, is investigated theoretically in aqueous solution, using the Gaussian 09 and DFT method. The energies of the HOMO and LUMO boundary orbitals, the atomic charges and the molecular electrostatic potentials of the acetylumbelliferone–nickel (Ni-AC4) complex were determined, analyzed and allowed us to locate the nucleophilic and electrophilic sites of attack for the free donor and the complex. Time-dependent density functional theory (TD-DFT) with PCM was used to calculate the electronic transitions of formed complex using the ground state geometry from optimized complexes. Charge transfer between metal and ligand is the most important factor for the stabilization of the complex (Ni-(AC) 4); NBO analysis confirmed this finding and indicated that the 3d orbital energy level order is dx2-y2 > dz2 > dxz, dyz > dxy.











Similar content being viewed by others
References
Dehari D, Jashari A, Dehari S, Shabani A (2012) New complexes of nickel (II) using 4-hydroxy-2-oxo-2H-chromene-3-carboxamide as ligand. World Acad Sci Eng Technol 6:1364–67
Anees Rahman KN, Haribabu J, Balachandran C, Bhuvanesh NSP, Ramasamy B, Karvembua R, Sreekanth A (2017) Copper, nickel and zinc complexes of 3-acetyl coumarin thiosemicarbazone: Synthesis, characterization and in vitro evaluation of cytotoxicity and DNA/protein binding properties. Polyhedron 135:26–35. https://doi.org/10.1016/j.poly.2017.06.044
Trendafilova N, Kostova I, Manolov I, Bauer G, Mihaylov T, Georgieva I (2004) Synthesis and spectroscopic study of a new lanthanum(III) complex of 3,3′-benzylidenedi-4-hydroxycoumarin. Synth React Inorg M 34:1635–1650. https://doi.org/10.1081/SIM-200026618
Grazula M, Budzisz E (2009) Biological activity of metal ions complexes of chromones, coumarins and flavones. Coord Chem Rev 25:32588–32598
Refat MS, El-Deen IM, Anwer ZM, El-Ghol S (2009) Bivalent transition metal complexes of coumarin-3-yl thiosemicarbazone derivatives: Spectroscopic, antibacterial activity and thermogravimetric studies, antibacterial activity and thermogravimetric studies. J Molecular Struct 920:149–162
Henkea H, Kandiollera W, Hanif M, Kepplera BK, Hartinger CG (2012) Organometallic ruthenium and osmium compounds of pyridin-2-and-4-ones as potential anticancer agents. Chem Biodivers 9:1718–1727. https://doi.org/10.1002/cbdv.201200005
Gasser G, Metzler-Nolte N (2012) The potential of organometallic complexes in medicinal chemistry. Curr Opin Chem Biol. 16:84–91. https://doi.org/10.1016/j.cbpa.2012.01.013
Guomundsson A, Bäckvall JE (2020) On the use of iron in organic chemistry. Molecules 25:1349. https://doi.org/10.3390/molecules25061349
Nicolaou KC (2014) Organic synthesis: the art and science of replicating the molecules of living nature and creating others like them in the laboratory. Proc R Soc 470:1–17. https://doi.org/10.1098/rspa.2013.0690
Boit TB, Bulger AS, Dander JE, Garg NK (2020) Activation of C−O and C−N bonds using non-precious-metal catalysis. ACS Catal 10:12109–12126. https://doi.org/10.1021/acscatal.0c03334
Wu L, Wang X, Xu W, Farzaneh F, Xu R (2009) The structure and pharmacological functions of coumarins and their derivatives. Curr Med Chem 16:4236–4260
Borges F, Roleira F, Milhazes N, Santana L, Uriarte E (2005) Simple coumarins and analogues in medicinal chemistry: occurrence, synthesis and biological activity. Curr Med Chem 12:887–916. https://doi.org/10.2174/0929867053507315
Matos MJ, Santana L, Uriarte E, Abreu OA, Molina E, Yordi EG (2015) Eds.; In Tech: Venketeshwer Rao. Vol. 1, Chapter 5, pp, 113–140. https://doi.org/10.5772/59982.
Manjunatha M, Naik VH, Kulkarni AD, Patil SA (2011) DNA cleavage, antimicrobial, antiinflammatory anthelmintic activities, and spectroscopic studies of Co(II), Ni(II), and Cu(II) complexes of biologically potential coumarin schiff bases. J Coord Chem 64:4264–4275
Hu Y, Chen W, Shen Y, Zhu B, Wang GX (2019) Synthesis and antiviral activity of coumarin derivatives against infectious hematopoietic necrosis virus. Bioorg Med Chem Lett 29:1749–1755. https://doi.org/10.5772/59982
Nofal ZM, El-Zahar MI, El-Karim SS (2000) Novel coumarin derivatives with expected biological activity. Molecules 5:99–113. https://doi.org/10.3390/50200099
Raev LD, Voinov E, Ivanov IC, Popov D (1990) Antitumor activity of some coumarin derivatives. Pharmazie 45:696–698
El-Sayed A, Abd-Allah O (2001) Synthetic and biological studies on coumarinhydrazone derivatives. Phosporus Sulfur Silicon Relat Elem 170:75–86
Giota AE, Fylaktakidou K, Litina D, Litinas K, Nicolaides D (2001) Synthesis and biological evaluation of several 3-(coumarin4-yl)tetrahydroisoxazole and 3-(coumarin-4-yl)dihydropyrazole derivatives. J Heterocyclic Chem 38:717–722. https://doi.org/10.1002/jhet.5570380329
Balcıoğlu S, lgun Karataş MO, Ateş B, Alıcı B, Özdemir İ (2020) Therapeutic potential of coumarin bearing metal complexes: where are we headed? Bioorg Med Chem Lett 30:15–30. https://doi.org/10.1016/j.bmcl.2019.126805
Carneiro A, Matos MJ, Uriarte E, Santana L (2021) Topics on coumarin and Its derivatives. Molecules 26:501. https://doi.org/10.3390/molecules26020501
Balewski Ł, Szulta S, Jalińska A, Kornicka A (2021) Recent advances in coumarin-metal complexes with biological properties. Front Chem 9:781779. https://doi.org/10.3389/fchem.2021.781779
Wang M, Wang LF, Li YZ, Li QX (2001) Antitumour activity of transition metal complexes with the thiosemicarbazone derived from 3-acetylumbelliferone. Transit Met Chem 26:307–310. https://doi.org/10.1023/A:1007159301849
Kulkarni A, Avaji PG, Bagihalli GB, Patil SA, Badami PS (2009) Synthesis, spectral, electrochemical and biological studies of Co(II), Ni(II) and Cu(II) complexes with Schiff bases of 8-formyl-7-hydroxy-4- methyl coumarin. J Coord Chem 62:481–492
Ashrafuzzaman MD, Camellia FK, Al Mahmud A, Joy Pramanik MD, Nahar K, Masuqul Haque MD, Kudrat-E-Zahan MD (2021) Bioactive mixed ligand metal complexes Of Cu(II), Ni(II), And Zn(II) Ions: synthesis, characterization, antimicrobial and antioxidant properties. J Chil Chem Soc 66:5295–5299. https://doi.org/10.4067/S0717-97072021000305295
Saddam Hossain Md, Zakaria CM, Roushown A, Kudrat-E-Zahan Md (2017) Selected pharmacological applications of 1st row transition metal complexes. Clin Med Res 6:177–191. https://doi.org/10.11648/j.cmr.20170606.13
Chohan H, Pervez H, Rauf A, Khan KM, Supuran CT (2004) Isatin-derived antibacterial and antifungal compounds and their transition metal complexes. J Enzyme Inhib Med Chem 19:417–423. https://doi.org/10.1080/14756360410001710383
Hashimoto T, Funatsu K, Ohtani A, Asano E, Yamaguchi Y (2019) Cross-coupling reaction of allylic ethers with aryl grignard reagents catalyzed by a nickel pincer complex. Molecules 24:2296. https://doi.org/10.3390/molecules24122296
Asano E, Hatayama Y, Kurisu N, Ohtani A, Hashimoto T, Kurihara Y, Ueda K, Ishihara S, Nagano H, Yamaguchi Y (2018) Acetylacetonato-based pincer-type nickel(II) complexes: synthesis and catalysis in cross-couplings of aryl chlorides with aryl grignard reagents. Dalton Trans 47:8003–8012. https://doi.org/10.1039/c8dt01295d
Nasirov FA (2003) Bifunctional nickel- or cobalt-containing catalyst-stabilizers for polybutadiene production and stabilization: (part II)-antioxidative properties and mechanism of catalyst-stabilizer’s action in the processes of thermo- and photooxidative ageing of polybutadiene. Iran Polym J. 12:281–289
Arslan H, Külcü N (2003) Synthesis and characterization of copper (II), nickel (II) and cobalt (II) complexes with novel thiourea derivatives. Transit Met Chem 28:816–819. https://doi.org/10.1023/A:1026064232260
Venanzi LM (1958) Tetrahedral complexes of nickel (II) and the factors determining their formation. J inorg nucl chem 8:137–142. https://doi.org/10.1016/0022-1902(58)80175-X
Zhang A, Wang C, Lai X, Zhai X, Pang M, Tunga CH, Wang W (2018) Reactivity of the diphosphinodithio ligated nickel (0) complex toward alkyl halides and resultant nickel(i) and nickel(ii)–alkyl complexes. Dalton Trans 44:15757–15764
Belbah H, Amira-Guebailia H, Affoune AM, Djaghout I, Houache O, Al-Kindi MA (2016) Effect of a natural brightener, daphne gnidium L. on the quality of nickel electroplating from wattsbath. J New Mat Electrochem Systems 19:97–102
Park T, Park JS, Sim JH, Kim SY (2020) 7-Acetoxycoumarin inhibits LPS-induced inflammatory cytokine synthesis by IκBα degradation and MAPK activation in macrophage cells. Molecules 14:3124. https://doi.org/10.3390/molecules25143124
Vijayalakshmi A, Sindhu G (2017) Dose responsive efficacy of umbelliferone on lipid peroxidation, anti-oxidant, and xenobiotic metabolism in DMBA-induced oral carcinogenesis. Biomed Pharmacother 88:852–862. https://doi.org/10.1016/j.biopha.2017.01.064
Kumar V, Ahmed D, Verma A, Anwar F, Ali M, Mujeeb M (2013) Umbelliferone β-D-galactopyranoside from Aegle marmelos (L.) corr. an ethnomedicinal plant with antidiabetic, antihyperlipidemic and antioxidative activity. BMC Complement. Altern Med 13:273. https://doi.org/10.1186/1472-6882-13-273
Islam N, Choi RJ, ** SE, Kim YS, Ahn BR, Zhao D, Jung HA, Choi JS (2012) Mechanism of anti-inflammatory activity of umbelliferone 6-carboxylic acid isolated from Angelica decursiva. J Ethnopharmacol 144:175–181. https://doi.org/10.1016/j.jep.2012.08.048
Ng TB, Liu F, Wang ZT (2000) Facile antioxidative activity of natural products from plants. Life Sci 66:709–723. https://doi.org/10.1016/s0024-3205(99)00642-6
Seo HJ, Kim JC (2014) 7-Acetoxycoumarin dimer-incorporated and folate-decorated liposomes: photoresponsive release and in vitro targeting and efficacy. Bioconjugate Chem 25:533–542. https://doi.org/10.1021/bc400521r
Wallingford CT (2009) Gaussian; Gaussian 09, revision A.1. Inc. GaussView v 5.0.9. Visualizer and Builder.
Torrent M, Solà M, Frenking G (2000) Theoretical studies of some transition-metal-mediated reactions of industrial and synthetic importance. Chem Rev 10:439–493. https://doi.org/10.1021/cr980452i
Adamo C, Jacquemin D (2013) The calculations of excited-state properties with time-dependent density functional theory. Chem Soc Rev 42:845–856. https://doi.org/10.1039/c2cs35394f
Ramasubbu N, Gnanaguru K, Venkatesan K, Ramamurt VN (1982) Topochemical photodimerization of 7-acetoxycoumarin: the acetoxy group as a steering agent. Can J Chem 60:2159–2161. https://doi.org/10.1139/v82-308
Jacquemin D, Perpète EA (2006) Time-dependent density functional theory investigation of the absorption, fluorescence, and phosphorescence spectra of solvated coumarins. J Chem Phys 125:164324. https://doi.org/10.1063/1.2361290
Had Al-Kadhemy MF, Rasheed ZS, Salim SR (2016) Fourier transform infrared spectroscopy for irradiation coumarin doped polystyrene polymer films by alpha ray. J Radiat Res Appl Sci 9:321–331. https://doi.org/10.1016/j.jrras.2016.02.004
Abbott AP, Ballantyne A, Harris RC, Juma JA, Ryder KS, Forrest G (2015) A comparative study of nickel electrodeposition using deep eutectic solvents and aqueous solutions. Electrochim Acta 176:718–726. https://doi.org/10.1016/j.electacta.2015.07.051
He W, Zhang BL, Zhou SY, Sun XL, Zhang SY (2007) Facile total synthesis of xanthotoxol. Synth Commun 37:361–367. https://doi.org/10.1080/00397910601038616
Seo HJ, Kim JC (2014) 7-acetoxycoumarin dimer-incorporated and folate-decorated liposomes: photoresponsive release and in vitro targeting and efficacy. Bioconjug Chem 25:533–542. https://doi.org/10.1021/bc400521r
Shakir M, Abbasi A, Khan AU, Khan SN (2011) Synthesis and spectroscopic studies on the Schiff base ligand derived from condensation of 2-furaldehyde and 3,3 ’-diaminobenzidene, L and its complexes with Co(II), Ni(II), Cu(II) and Zn(II): Comparative DNA binding studies of L and its Cu(II) and Zn(II) complexes. Spectrochim Acta Part A 78:29–35. https://doi.org/10.1016/j.saa.2010.02.034
Boulmokh Y, Belguidoum K, Meddour F, Amira-Guebailia H (2021) Investigation of antioxidant activity of epigallocatechin gallate and epicatechin as compared to resveratrol and ascorbic acid: experimental and theoretical insights. Struct Chem 32:1907–1923. https://doi.org/10.1007/s11224-021-01763-5
Macit M, Tanak H, Orbay M, Özdemir N (2017) Synthesis, crystal structure, spectroscopic characterization and DFT studies of bis[(1Z,2E)-N-(2,6-diethylphenyl)-N’-hydroxy-2-(hydroxyimino)acetimidamidato]nickel(II). Inorganica Chimica Acta 459:36–44. https://doi.org/10.1016/j.ica.2017.01.013
Chermette H (1999) Chemical reactivity indexes in density functional theory. J Comp Chem 20:129–154. https://doi.org/10.1002/(SICI)1096-987X(19990115)20:1%3c129::AID-JCC13%3e3.0.CO;2-A
Golding RM, Ahn S (1981) Calculation of dipole moments for transition met complexes. Bull Korean Chem Soc 2:48–55
Hübschle CB, van Smaalen S (2017) The electrostatic potential of dynamic charge densities. J Appl Crystallogr 50:1627–1636. https://doi.org/10.1107/S1600576717013802
Balachandran V, Karthick T, Perumal S, Nataraj A (2013) Comparative theoretical studies on natural atomic orbitals, natural bond orbitals and simulated UV-visible spectra of N-(methyl)phthalimide and N-(2 bromoethyl)phthalimide. IJPAP 51:178–184
Midoune A, Messaoudi A (2021) DFT/TDDFT studies of the structural, electronic and NBO properties of some complexes with the tetrathiafulvalene-1, 3-benzothiazole ligand. Inorganica Chim Acta 516:120151. https://doi.org/10.1016/j.ica.2020.120151
Mason WR, Gray HB (1968) Electronic structures of square-planar complexes. J Am Chem Soc 90:5721–5729. https://doi.org/10.1021/JA01023A012
Börgel J, Campbell MG, Ritter T (2016) Transition metal d-orbital splitting diagrams: an updated educational resource for square planar transition metal complexes. J Chem Educ 1(93):118–121. https://doi.org/10.1021/acs.jchemed.5b00542
Takashiro A, Seiko K, Akio U (2001) Assignment of d-d transitions of square planar [CuIIN4] complexes containing imidate and amine ligands by means of polarized crystal spectra. Bull Chem Soc Jpn. 74:851–860
Banciu C, Lepädatu CI (1975) Electronic transitions in square-planar complexes with Ni(d8)-N(sp2) bonds. Z Phys Chem (NF) 97:197–205
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Belguidoum, K., Boulmokh, Y., Hamamdia, F.Z. et al. Tetradentate square-planar acetylumbelliferone–nickel (II) complex formation: a DFT and TD-DFT study. Theor Chem Acc 141, 48 (2022). https://doi.org/10.1007/s00214-022-02903-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-022-02903-8