Abstract
Rationale
on!® nicotine pouches (NPs) are oral tobacco-derived nicotine products that are tobacco-leaf free and are available in a variety of flavors and nicotine strengths. Switching completely to NPs from cigarettes may present the potential to reduce harm in adult smokers (AS) unable or unwilling to quit smoking. We characterized the abuse potential of six different flavor variants of NPs compared to cigarettes.
Objectives
The objective of this study was to evaluate the nicotine pharmacokinetics (PK) and subjective effects of different flavor variants of NPs compared to participants’ own brand cigarettes (OBCs) in AS.
Methods
In this single-blind, randomized, 7-way crossover study, we assessed nicotine PK, subjective measures (using well-established questionnaires), and product use behavior associated with six flavors of 4 mg NPs and OBCs in AS that remained in clinic for the duration of the test period.
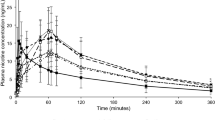
Results
Nicotine Cmax values ranged from 9.0 to 11.5 ng/mL for the NPs and 16.3 ng/mL for OBCs. The tmax ranged from 30.1 to 34.9 min for ONPs and 7.5 min for OBCs. Use of NPs resulted in lower ratings of urge to smoke or craving a cigarette. All the NPs were considered pleasant, but not as much as OBCs. Flavor did not appear to influence the nicotine PK or subjective responses.
Conclusions
Based on the nicotine PK parameters and subjective responses, we conclude that NPs, regardless of flavor, likely have lower abuse potential than cigarettes. Overall, this study suggests that the NPs may be potentially acceptable switching products for adult smokers.



Similar content being viewed by others
References
Atzori G, Lemmonds CA, Kotler ML, Durcan MJ, Boyle J (2008) Efficacy of a nicotine (4 mg)-containing lozenge on the cognitive impairment of nicotine withdrawal. J Clin Psychopharmacol 28(6):667–674. https://doi.org/10.1097/JCP.0b013e31818c9bb8
Blank MD, Eissenberg T (2010) Evaluating oral noncombustible potential-reduced exposure products for smokers. Nicotine Tob Res 12(4):336–343. https://doi.org/10.1093/ntr/ntq003
Blank MD, Sams C, Weaver MF, Eissenberg T (2008) Nicotine delivery, cardiovascular profile, and subjective effects of an oral tobacco product for smokers. Nicotine Tob Res 10(3):417–421. https://doi.org/10.1080/14622200801901880
Cappelleri JC, Bushmakin AG, Baker CL, Merikle E, Olufade AO, Gilbert DG (2007) Confirmatory factor analyses and reliability of the modified cigarette evaluation questionnaire. Addict Behav 32(5):912–923. https://doi.org/10.1016/j.addbeh.2006.06.028
Carter LP, Stitzer ML, Henningfield JE, O’Connor RJ, Cummings KM, Hatsukami D (2009) Abuse liability assessment of tobacco products including potential reduced exposure products. Cancer Epidemiol Biomark Prev 18(12):3241–3262. https://doi.org/10.1158/1055-9965.EPI-09-0948(NOTINFILE)
Cobb CO, Weaver MF, Eissenberg T (2010) Evaluating the acute effects of oral, non-combustible potential reduced exposure products marketed to smokers. Tob Control 19(5):367–373. https://doi.org/10.1136/tc.2008.028993
Cox LS, Tiffany ST, Christen AG (2001) Evaluation of the brief questionnaire of smoking urges (QSU-brief) in laboratory and clinical settings. Nicotine Tob Res 3(1):7–16. https://doi.org/10.1080/14622200020032051
Du D, Nides M, Borders J, Selmani A, Waverczak W (2014) Comparison of nicotine oral soluble film and nicotine lozenge on efficacy in relief of smoking cue-provoked acute craving after a single dose of treatment in low dependence smokers. Psychopharmacology 231(22):4383–4391. https://doi.org/10.1007/s00213-014-3586-2
Du D, Borders J, Selmani A, Waverczak W (2015) A pilot study to investigate the efficacy of nicotine oral soluble film, lozenge and gum in relief of acute smoking cue-provoked craving for cigarette in low dependence smokers. J Smok Cessat 10(2):87–95. https://doi.org/10.1017/jsc.2014.5
Gottlieb S, Zeller M (2017) A nicotine-focused framework for public health. N Engl J Med 377(12):1111–1114. https://doi.org/10.1056/NEJMp1707409
Gray JN, Breland AB, Weaver M, Eissenberg T (2008) Potential reduced exposure products (PREPs) for smokeless tobacco users: clinical evaluation methodology. Nicotine Tob Res 10(9):1441–1448. https://doi.org/10.1080/14622200802323258
Griffiths RR, Bigelow GE, Ator NA (2003) Principles of initial experimental drug abuse liability assessment in humans. Drug Alcohol Depend 70(3 Suppl):S41-54. https://doi.org/10.1016/s0376-8716(03)00098-x
Hanson K, O’Connor R, Hatsukami D (2009) Measures for assessing subjective effects of potential reduced-exposure products [18/12/3209 pii ;10.1158/1055-9965.EPI-09-0971 doi]. Cancer Epidemiol Biomark Prevent 18(12):3209–3224. https://doi.org/10.1158/1055-9965.EPI-09-0971
Hatsukami D, Lemmonds C, Tomar SL (2004) Smokeless tobacco use: harm reduction or induction approach? Prev Med 38(3):309–317. https://doi.org/10.1016/j.ypmed.2003.10.006
Hatsukami DK, Zhang Y, O’Connor RJ, Severson HH (2013) Subjective responses to oral tobacco products: scale validation. Nicotine Tob Res 15(7):1259–1264. https://doi.org/10.1093/ntr/nts265
Henningfield JE, Keenan RM (1993) Nicotine delivery kinetics and abuse liability. J Consult Clin Psychol 61(5):743–750. https://doi.org/10.1037//0022-006x.61.5.743
Institute of Medicine (2012) Scientific standards for studies on modified risk tobacco products. The National Academies Press. http://www.nap.edu/catalog/13294/scientific-standards-for-studies-on-modified-risk-tobacco-products. Accessed 30 April 2021
Kotlyar M, Mendoza-Baumgart MI, Li ZZ, Pentel PR, Barnett BC, Feuer RM, Smith EA, Hatsukami DK (2007) Nicotine pharmacokinetics and subjective effects of three potential reduced exposure products, moist snuff and nicotine lozenge. Tob Control 16(2):138–142. https://doi.org/10.1136/tc.2006.018440
Krautter GR, Borgerding MF (2014) Comparison of consumption patterns, biomarkers of exposure, and subjective effects in cigarette smokers who switched to dissolvable tobacco (Camel Orbs), dual use, or tobacco abstinence. Nicotine Tob Res 16(10):1336–1347. https://doi.org/10.1093/ntr/ntu082
Krautter GR, Chen PX, Borgerding MF (2015) Consumption patterns and biomarkers of exposure in cigarette smokers switched to Snus, various dissolvable tobacco products, dual use, or tobacco abstinence. Regul Toxicol Pharmacol 71(2):186–197. https://doi.org/10.1016/j.yrtph.2014.12.016
Lunell E, Curvall M (2011) Nicotine delivery and subjective effects of Swedish portion snus compared with 4 mg nicotine polacrilex chewing gum. Nicotine Tob Res 13(7):573–578. https://doi.org/10.1093/ntr/ntr044
Mendoza-Baumgart MI, Tulunay OE, Hecht SS, Zhang Y, Murphy S, Le C, Jensen J, Hatsukami DK (2007) Pilot study on lower nitrosamine smokeless tobacco products compared with medicinal nicotine. Nicotine Tob Res 9(12):1309–1323. https://doi.org/10.1080/14622200701704228
Perkins KA, Grobe JE, Caggiula A, Wilson AS, Stiller RL (1997) Acute reinforcing effects of low-dose nicotine nasal spray in humans. Pharmacol Biochem Behav 56(2):235–241. https://doi.org/10.1016/s0091-3057(96)00216-x
Pickworth WB, Rosenberry ZR, Gold W, Koszowski B (2014) Nicotine absorption from smokeless tobacco modified to adjust pH. J Addict Res Ther 5(3):1000184. https://doi.org/10.4172/2155-6105.1000184
Rasmussen SC, Becker WD, Shanga GM (2018) Single-dose bioequivalence of two mini nicotine lozenge formulations. Clin Pharmacol Drug Dev 7(5):498–505. https://doi.org/10.1002/cpdd.415
Robichaud MO, Seidenberg AB, Byron MJ (2019) Tobacco companies introduce ‘tobacco-free’ nicotine pouches. Tobacco Control 29(E1):e145–e146. https://doi.org/10.1136/tobaccocontrol-2019-055321
Rose JE, Salley A, Behm FM, Bates JE, Westman EC (2010) Reinforcing effects of nicotine and non-nicotine components of cigarette smoke [10.1007/s00213-010-1810-2 doi]. Psychopharmacology 210(1):1–12. https://doi.org/10.1007/s00213-010-1810-2
Shiffman S (2008) Effect of nicotine lozenges on affective smoking withdrawal symptoms: secondary analysis of a randomized, double-blind, placebo-controlled clinical trial. Clin Ther 30(8):1461–1475. https://doi.org/10.1016/j.clinthera.2008.07.019
St. Helen G, Havel C, Dempsey DA, Jacob P, Benowitz NL (2016) Nicotine delivery, retention and pharmacokinetics from various electronic cigarettes. Addiction 111:535–544. https://doi.org/10.1111/add.13183
Stegmayr B, Eliasson M, Rodu B (2005) The decline of smoking in northern Sweden. Scand J Public Health 33(4):321–324. https://doi.org/10.1080/14034940510032301 (discussion 243)
Sukhija M, Srivastava R, Kaushik A (2018) Pharmacokinetic characterization of three novel 4-mg nicotine lozenges. Int J Clin Pharmacol Ther 56(3):113–119. https://doi.org/10.5414/CP203097
United States Department of Health and Human Services, Substance Abuse and Mental Health Services Administration (SAMHSA), Center for Behavioral Health Statistics and Quality. National Survey on Drug Use and Health, 2018. 2019; https://pdas.samhsa.gov/#/survey/NSDUH-2018-DS0001. Accessed 30 April 2021
Vansickel AR, Cobb CO, Weaver MF, Eissenberg TE (2010) A clinical laboratory model for evaluating the acute effects of electronic “cigarettes”: nicotine delivery profile and cardiovascular and subjective effects. Cancer Epidemiol Biomarkers Prev 19:1945–1953. https://doi.org/10.1158/1055-9965.EPI-10-0288
Acknowledgements
Scientific writing support was provided by Meghan L Thompson, PharmD, PhD and Cynthia S Cecil, both of Altria Client Services LLC.
Funding
This study was funded by Altria Client Services LLC. Authors JR, JL, JW, AV, JE, and MS are employees of Altria Client Services LLC.
The Advarra Institutional Review Board (IRB) (Columbia, MD) reviewed and approved this study. The study was conducted in accordance with Food and Drug Administration (FDA) regulations as described in the Code of Federal Regulations (CFR) 21 Parts 50 and 56, Department of Health and Human Services regulations as described in 45 CFR 46, guidelines resulting from the International Council for Harmonisation (ICH) E6 Good Clinical Practice (GCP), the European Union (EU) directive 2001/20/EC, and the ethical principles set forth in the Declaration of Helsinki. In addition, the IRB operates in compliance with the portions of the Health Insurance Portability and Accountability Act of 1996 (HIPAA Privacy Rule) that apply to research, as described in 45 CFR Parts 160 and 164. All participants in this study reviewed, signed, and dated the informed consent form prior to study initiation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rensch, J., Liu, J., Wang, J. et al. Nicotine pharmacokinetics and subjective response among adult smokers using different flavors of on!® nicotine pouches compared to combustible cigarettes. Psychopharmacology 238, 3325–3334 (2021). https://doi.org/10.1007/s00213-021-05948-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-021-05948-y