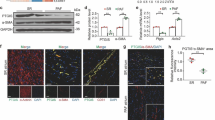
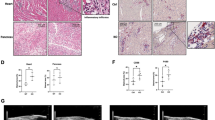
Abstract
Myocardial infarction is a worldwide disease with high morbidity and mortality and a major cause of chronic heart failure, seriously affecting patients’ quality of life. Natural medicine has been used to cure or prevent cardiovascular disease for decades. As a natural flavonoid, anthocyanidin has been used to treat many diseases due to its antioxidative, anti-inflammatory, and other properties. A mouse model (C57BL/6) weighing 30–40 g was utilized to induce myocardial infarction by ligating the left anterior descending coronary artery. Cyanidin (30 mg/kg) was administered orally to mice for four weeks. A variety of assessments were used to evaluate cardiac function. The gene expression was measured using RNAseq and Western blot. Histological changes in myocardial tissue were assessed using staining techniques, including Masson, Hematoxylin Eosin (HE), and transmission electron microscopy. Tunnel staining was implemented as a method to detect cellular apoptosis. For the quantification of B-type natriuretic peptide (BNP) and atrial natriuretic peptide (ANP) in the serum, an enzyme-linked immunosorbent assay (ELISA) was employed. Furthermore, autodock simulation was executed in order to assess the interaction between cyanidin and a subset of genes. Cyanidin treatment inhibited myocardial cell apoptosis, improved cardiac function, and reduced serum concentrations of BNP and atrial natriuretic peptide ANP, as well as mitigated histological cardiac tissue damage. Cyanidin also inhibited the activity of matrix metalloproteinases (MMP2/9) and Fibronectin 1 (Fn1). Cyanidin improves heart function and reduces myocardial damage in mice after MI. Furthermore, cyanidin can prevent cardiomyocyte apoptosis. These effects are most likely caused by suppression of MMP9/2 and control of the Akt signaling pathway, suggesting an appropriate therapeutic target.






Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
Abbreviations
- Akt:
-
Protein kinase B
- Col:
-
Collagen
- Cyn:
-
Cyanidin
- DEGs:
-
Differentially expressed genes
- ECG:
-
Electrocardiogram
- FN:
-
Fibronectin
- GO:
-
Gene ontology
- HE:
-
Hematoxylin and eosin
- qPCR:
-
Quantitative real-time quantitative
- LVEF:
-
Left ventricular ejection fraction
- MMP:
-
Matrix metalloproteinases
- RMSD:
-
Means square deviation
- MI:
-
Myocardial infarction
- OCT:
-
Optical coherence tomography
- PPI:
-
Protein-protein interaction
- TGF-β:
-
Transforming growth factor beta
- UV:
-
Ultraviolet
References
Audic S, Claverie JM (1997) The significance of digital gene expression profiles. Genome Res 7(10):986–995
Cheng N, Wang MY, Wu YB, Cui HM, Wei SX, Liu B, Wang R (2020) Circular RNA POSTN promotes myocardial infarction-induced myocardial injury and cardiac remodeling by regulating miR-96-5p/BNIP3 axis. Front Cell Dev Biol 8:618574
Cui N, Hu M, Khalil RA (2017) Biochemical and biological attributes of matrix metalloproteinases. Prog Mol Biol Transl Sci 147:1–73
Daseke MJ 2nd, Valerio FM, Kalusche WJ, Ma Y, DeLeon-Pennell KY, Lindsey ML (2019) Neutrophil proteome shifts over the myocardial infarction time continuum. Basic Res Cardiol 114(5):37
DeFilippis AP, Chapman AR, Mills NL, de Lemos JA, Arbab-Zadeh A, Newby LK, Morrow DA (2019a) Assessment and treatment of patients with type 2 myocardial infarction and acute nonischemic myocardial injury. Circulation 140(20):1661–1678
DeFilippis AP, Nasir K, Blaha MJ (2019b) Myocardial infarction as a clinical end point in research. Circ Res 124(12):1701–1703
Etoh T, Joffs C, Deschamps AM, Davis J, Dowdy K, Hendrick J, Baicu S, Mukherjee R, Manhaini M, Spinale FG (2001) Myocardial and interstitial matrix metalloproteinase activity after acute myocardial infarction in pigs. Am J Physiol Heart Circ Physiol 281(3):H987-994
Falk E, Nakano M, Bentzon JF, Finn AV, Virmani R (2013) Update on acute coronary syndromes: the pathologists’ view. Eur Heart J 34(10):719–728
Guo D, Kassiri Z, Basu R, Chow FL, Kandalam V, Damilano F, Liang W, Izumo S, Hirsch E, Penninger JM, Backx PH, Oudit GY (2010) Loss of PI3Kgamma enhances cAMP-dependent MMP remodeling of the myocardial N-cadherin adhesion complexes and extracellular matrix in response to early biomechanical stress. Circ Res 107(10):1275–1289
Hao J, Du H, Li W, Liu F, Lu J, Yang X, Cui W (2016) Anthocyanins protected hearts against ischemic injury by reducing MMP-2 activity via Akt/P38 pathways. Am J Transl Res 8(2):1100–1107
Hausenloy DJ, Chilian W, Crea F, Davidson SM, Ferdinandy P, Garcia-Dorado D, van Royen N, Schulz R, Heusch G (2019) The coronary circulation in acute myocardial ischaemia/reperfusion injury: a target for cardioprotection. Cardiovasc Res 115(7):1143–1155
Jana S, Patel D, Patel S, Upadhyay K, Thadani J, Mandal R, Das S, Devkar R (2017) Anthocyanin rich extract of Brassica oleracea L. alleviates experimentally induced myocardial infarction. PLoS ONE 12(8):e0182137
Jenca D, Melenovsky V, Stehlik J, Stanek V, Kettner J, Kautzner J, Adamkova V, Wohlfahrt P (2021) Heart failure after myocardial infarction: incidence and predictors. ESC Heart Fail 8(1):222–237
Jha S, Kanaujia SP, Limaye AM (2016) Direct inhibition of matrix metalloproteinase-2 (MMP-2) by (-)-epigallocatechin-3-gallate: a possible role for the fibronectin type II repeats. Gene 593(1):126–130
Ji J, Chen L, Zhuang Y, Han Y, Tang W, **a F (2020) Fibronectin 1 inhibits the apoptosis of human trophoblasts by activating the PI3K/Akt signaling pathway. Int J Mol Med 46(5):1908–1922
Johansson S, Rosengren A, Young K, Jennings E (2017) Mortality and morbidity trends after the first year in survivors of acute myocardial infarction: a systematic review. BMC Cardiovasc Disord 17(1):53
Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M, Itoh M, Katayama T, Kawashima S, Okuda S, Tokimatsu T, Yamanishi Y (2008) KEGG for linking genomes to life and the environment. Nucleic Acids Res 36(Database issue):D480-484
Konstandin MH, Toko H, Gastelum GM, Quijada P, De La Torre A, Quintana M, Collins B, Din S, Avitabile D, Volkers M, Gude N, Fassler R, Sussman MA (2013) Fibronectin is essential for reparative cardiac progenitor cell response after myocardial infarction. Circ Res 113(2):115–125
Kopustinskiene DM, Jakstas V, Savickas A, Bernatoniene J (2020) Flavonoids as anticancer agents. Nutrients 12(2):457
Kuhn TC, Knobel J, Burkert-Rettenmaier S, Li X, Meyer IS, Jungmann A, Sicklinger F, Backs J, Lasitschka F, Muller OJ, Katus HA, Krijgsveld J, Leuschner F (2020) Secretome analysis of cardiomyocytes identifies PCSK6 (proprotein convertase subtilisin/kexin type 6) as a novel player in cardiac remodeling after myocardial infarction. Circulation 141(20):1628–1644
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9(4):357–359
Lee YM, Yoon Y, Yoon H, Park HM, Song S, Yeum KJ (2017) Dietary anthocyanins against obesity and inflammation. Nutrients 9(10):1089
Li B, Dewey CN (2011) RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinformatics 12:323
Liobikas J, Skemiene K, Trumbeckaite S, Borutaite V (2016) Anthocyanins in cardioprotection: a path through mitochondria. Pharmacol Res 113(Pt B):808–815
Mao Q, Liang X, Wu Y, Lu Y (2019) Nobiletin protects against myocardial injury and myocardial apoptosis following coronary microembolization via activating PI3K/Akt pathway in rats. Naunyn Schmiedebergs Arch Pharmacol 392(9):1121–1130
Melchior-Becker A, Dai G, Ding Z, Schafer L, Schrader J, Young MF, Fischer JW (2011) Deficiency of biglycan causes cardiac fibroblasts to differentiate into a myofibroblast phenotype. J Biol Chem 286(19):17365–17375
Minicucci MF, Azevedo PS, Polegato BF, Paiva SA, Zornoff LA (2011) Heart failure after myocardial infarction: clinical implications and treatment. Clin Cardiol 34(7):410–414
Moriya C, **nin M, Yamane K, Maruo K, Muchemwa FC, Igata T, Makino T, Fukushima S, Ihn H (2011) Expression of matrix metalloproteinase-13 is controlled by IL-13 via PI3K/Akt3 and PKC-delta in normal human dermal fibroblasts. J Invest Dermatol 131(3):655–661
Pedersen LR, Frestad D, Michelsen MM, Mygind ND, Rasmusen H, Suhrs HE, Prescott E (2016) Risk factors for myocardial infarction in women and men: a review of the current literature. Curr Pharm Des 22(25):3835–3852
Petroni K, Trinei M, Fornari M, Calvenzani V, Marinelli A, Micheli LA, Pilu R, Matros A, Mock HP, Tonelli C, Giorgio M (2017) Dietary cyanidin 3-glucoside from purple corn ameliorates doxorubicin-induced cardiotoxicity in mice. Nutr Metab Cardiovasc Dis 27(5):462–469
Porter KE, Turner NA (2009) Cardiac fibroblasts: at the heart of myocardial remodeling. Pharmacol Ther 123(2):255–278
Samad M, Malempati S, Restini CBA (2023) Natriuretic peptides as biomarkers: narrative review and considerations in cardiovascular and respiratory dysfunctions. Yale J Biol Med 96(1):137–149
Sarzani R, Allevi M, Di Pentima C, Schiavi P, Spannella F, Giulietti F (2022) Role of cardiac natriuretic peptides in heart structure and function. Int J Mol Sci 23(22):14415
Serraino I, Dugo L, Dugo P, Mondello L, Mazzon E, Dugo G, Caputi AP, Cuzzocrea S (2003) Protective effects of cyanidin-3-O-glucoside from blackberry extract against peroxynitrite-induced endothelial dysfunction and vascular failure. Life Sci 73(9):1097–1114
Shi H, Zhou P, Gao G, Liu PP, Wang SS, Song R, Zou YY, Yin G, Wang L (2021) Astragaloside IV prevents acute myocardial infarction by inhibiting the TLR4/MyD88/NF-kappaB signaling pathway. J Food Biochem 45(7):e13757
Sullivan RD, Houng AK, Gladysheva IP, Fan TM, Tripathi R, Reed GL, Wang D (2020) Corin overexpression reduces myocardial infarct size and modulates cardiomyocyte apoptotic cell death. Int J Mol Sci 21(10):3456
Ushakov A, Ivanchenko V, Gagarina A (2020) Regulation of myocardial extracellular matrix dynamic changes in myocardial infarction and postinfarct remodeling. Curr Cardiol Rev 16(1):11–24
Wang C, Bai X, Liu S, Wang J, Su Z, Zhang W, Bu D, Yan Y, Song X (2018) RNA-seq based transcriptome analysis of the protective effect of compound longmaining decoction on acute myocardial infarction. J Pharm Biomed Anal 158:339–345
Wang Y, Gao H, Cao X, Li Z, Kuang Y, Ji Y, Li Y (2022) Role of GADD45A in myocardial ischemia/reperfusion through mediation of the JNK/p38 MAPK and STAT3/VEGF pathways. Int J Mol Med 50(6):1–11
Wu RX, Laser M, Han H, Varadarajulu J, Schuh K, Hallhuber M, Hu K, Ertl G, Hauck CR, Ritter O (2006) Fibroblast migration after myocardial infarction is regulated by transient SPARC expression. J Mol Med (Berl) 84(3):241–252
Xu I, Theriault M, Brunette I, Rochette PJ, Proulx S (2021) Matrix metalloproteinases and their inhibitors in Fuchs endothelial corneal dystrophy. Exp Eye Res 205:108500
Yin Z, Wu Y, Chen Y, Qie X, Zeng M, Wang Z, Qin F, Chen J, He Z (2021) Analysis of the interaction between cyanidin-3-O-glucoside and casein hydrolysates and its effect on the antioxidant ability of the complexes. Food Chem 340:127915
Zeng Z, Wang Q, Yang X, Ren Y, Jiao S, Zhu Q, Guo D, **a K, Wang Y, Li C, Wang W (2019) Qishen granule attenuates cardiac fibrosis by regulating TGF-beta /Smad3 and GSK-3beta pathway. Phytomedicine 62:152949
Zhang Y, Lin X, Chu Y, Chen X, Du H, Zhang H, Xu C, **e H, Ruan Q, Lin J, Liu J, Zeng J, Ma K, Chai D (2021) Dapagliflozin: a sodium-glucose cotransporter 2 inhibitor, attenuates angiotensin II-induced cardiac fibrotic remodeling by regulating TGFbeta1/Smad signaling. Cardiovasc Diabetol 20(1):121
Zhao J, Miao G, Wang T, Li J, **e L (2020) Urantide attenuates myocardial damage in atherosclerotic rats by regulating the MAPK signalling pathway. Life Sci 262:118551
Funding
This research was funded by the National Key Research and Development Program of China (2017YFA010560, 22018YFA0108700), the NSFC Projects of International Cooperation and Exchanges (81720108004), the National Natural Science Foundation of China (81974019), the Research Team Project of Natural Science Foundation of Guangdong Province of China (2017A030312007), the Key Program of Guangzhou Science Research Plan (201904020047), the Special Projects of Dengfeng Program of Guangdong Provincial People’s Hospital (DFJH201812, KJ012019119, KJ012020630, KJ012019423, KY0120220133, DFJHBF202111, KJ012020630), and the National Natural Science Foundation of China (82001301).
Author information
Authors and Affiliations
Contributions
All authors declare that all data were generated in-house and that no paper mill was used. Conceptualization: MIN,DG; investigation MIN, DG, SZ, MM, YR, OY original draft preparation MIN, SX, LG, ZM, NL, GL, CL; review and editingMIN, MM, GL, CL; funding acquisition MIN, PZ.
Corresponding authors
Ethics declarations
Ethics approval
The experiment was conducted after an animal ethical review by the Guangdong Provincial People’s Hospital of the Guangdong Academy of Medical Sciences (2019492A(R2)).
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
210_2024_2975_MOESM1_ESM.jpg
Supplementary file1 (JPG 1241 KB) Supplementary Figure 1: mRNA expression of TGF-β, Col I, MMP-2/9, FN1 p-pi3k, p-Akt, p-mTOR compared to 18S. Each experience was repeated 3 times ****P<0.0005.
210_2024_2975_MOESM2_ESM.jpg
Supplementary file2 (JPG 1818 KB) Supplementary Figure 2: RMSF curve represents the fluctuation of protein amino acid residues. (A) The figure demonstrates that many smaller locations inside the core of the protein have more flexibility in comparison to other parts. (B) The graph demonstrates that some of these smaller regions within the core structure of the protein are more flexible than other areas. (C) As can be observed from the figure, the total residue of the protein changes less, and this is because the protein has a very compact barrel-shaped structure. Because of this, the amino acid is bound, and as a result, it swings less. (D) As the image on the right demonstrates, this region is dominated by a loop, which causes it to exhibit variability.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gang, D., Qing, O., Yang, Y. et al. Cyanidin prevents cardiomyocyte apoptosis in mice after myocardial infarction. Naunyn-Schmiedeberg's Arch Pharmacol (2024). https://doi.org/10.1007/s00210-024-02975-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00210-024-02975-2