Abstract
Lung cancer is the most common cause of cancer-related mortality, chemo-resistance, and toxicity limit treatment. The focus is on innovative combined phytotherapy to improve treatment outcomes. Our aim was to investigate the potential effects of daidzein nanosuspension (DZ-NS) and its combination with cisplatin (CIS) on A549 non-small lung cancer cells. Cytotoxicity was investigated using MTT and Chou-Talalay methods. Oxidative, apoptotic, and inflammatory markers were analyzed by ELISA and qRT-PCR. The IC50 value for DZ-NS was 25.23 µM for 24 h and was lower than pure DZ (IC50 = 835 µM for pure DZ). DZ-NS (at IC50x2 and IC50 values) showed synergistic cytotoxicity with CIS. The cells treated with DZ-NS had low TOS and OSI levels. However, DZ-NS failed to regulate Cas3 and TGF-β1 activation in A549 cells. MMP-9 gene expression was significantly suppressed in DZ-NS-treated cells, especially in combination therapy. DZ represents a potential combination option for the treatment of lung cancer, and its poor toxicokinetic properties limit its clinical use. To overcome these limitations, the effects of the nanosuspension formulation were tested. DZ-NS showed a cytotoxic effect on A549 cells and optimized the therapeutic effect of CIS. This in vitro synergistic effect was mediated by suppression of MMP-9 and not by oxidative stress or Cas3-activated apoptosis. This study provides the basis for an in vivo and clinical trial of DZ-NS with concurrent chemotherapy.
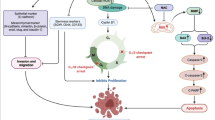
Graphical Abstract




Similar content being viewed by others
Data availability
Data available on request from the authors. The data that support the findings of this study are available from the corresponding author, Merve Becit-Kizilkaya, upon reasonable request.
Abbreviations
- Cas-3:
-
Caspase-3
- CI:
-
Combination index
- CIS:
-
Cisplatin
- DZ-NS:
-
Daidzein nanosuspension
- DZ:
-
Daidzein
- ELISA:
-
Enzyme-linked immunosorbent assay
- MMP:
-
Matrix metalloproteinase-9
- MTT:
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- NS:
-
Nanosuspension formulations
- NSCLC:
-
Non-small cell lung cancer
- OSI:
-
Oxidative stress index
- qRT-PCR:
-
Quantitative real time polymerase chain reaction
- TAS:
-
Total antioxidant status
- TGF-β1:
-
Transforming growth factor-β1
- TOS:
-
Total oxidant status
References
Abu Rous F, Singhi EK, Sridhar A, Faisal MS, Desai A (2023) Lung cancer treatment advances in 2022. Cancer Invest. 41(1):12–24. https://doi.org/10.1080/07357907.2022.2119479
Alshehri MM, Sharifi-Rad J, Herrera-Bravo J, Jara EL, Salazar LA, Kregiel D, Uprety Y, Akram M, Iqbal M, Martorell M, Torrens-Mas M, Pons DG, Daştan SD, Cruz-Martins N, Ozdemir FA, Kumar M, Cho WC (2021) Therapeutic potential of isoflavones with an emphasis on daidzein. Oxid Med Cell Longev. 2021:6331630. https://doi.org/10.1155/2021/6331630
Barta JA, Powell CA, Wisnivesky JP (2019) Global epidemiology of lung cancer. Ann Glob Health. 85(1):8. https://doi.org/10.5334/aogh.2419
BayatMokhtari R, Homayouni TS, Baluch N, Morgatskaya E, Kumar S, Das B, Yeger H (2017) Combination therapy in combating cancer. Oncotarget. 8(23):38022–38043. https://doi.org/10.18632/oncotarget.16723
Becit M, Aydin S (2020) An in vitro study on the interactions of Pycnogenol® with cisplatin in human cervical cancer cells. Turk J Pharm Sci. 17(1):1–6. https://doi.org/10.4274/tjps.galenos.2018.97759
Becit-Kızılkaya M, Öncü Ş, Şen S, Çelik S (2023) Berberine synergizes with cisplatin via inducing apoptosis on A549 non-small cell lung cancer cells. Eur J Ther. https://doi.org/10.58600/eurjther1703
Berrouet C, Dorilas N, Rejniak KA, Tuncer N (2020) Comparison of drug inhibitory effects ([Formula: see text]) in monolayer and spheroid cultures. Bull Math Biol. 82(6):68. https://doi.org/10.1007/s11538-020-00746-7
Bhat AA, Thapa R, Afzal O, Agrawal N, Almalki WH, Kazmi I, Alzarea SI, Altamimi ASA, Prasher P, Singh SK, Dua K, Gupta G (2023) The pyroptotic role of Caspase-3/GSDME signalling pathway among various cancer: a Review. Int J Biol Macromol. 242(Pt 2):124832. https://doi.org/10.1016/j.ijbiomac.2023.124832
Bi C, Miao XQ, Chow SF, Wu WJ, Yan R, Liao YH, Chow AH, Zheng Y (2017) Particle size effect of curcumin nanosuspensions on cytotoxicity, cellular internalization, in vivo pharmacokinetics and biodistribution. Nanomedicine. 13(3):943–953. https://doi.org/10.1016/j.nano.2016.11.004
Bi YL, Min M, Shen W, Liu Y (2018) Genistein induced anticancer effects on pancreatic cancer cell lines involves mitochondrial apoptosis, G0/G1cell cycle arrest and regulation of STAT3 signalling pathway. Phytomedicine. 39:10–16. https://doi.org/10.1016/j.phymed.2017.12.001
Boyacıoğlu Ö, Bilgiç E, Varan C, Bilensoy E, Nemutlu E, Sevim D, Kocaefe Ç, Korkusuz P (2021) ACPA decreases non-small cell lung cancer line growth through Akt/PI3K and JNK pathways in vitro. Cell Death Dis. 12(1):56. https://doi.org/10.1038/s41419-020-03274-3
Castañeda AM, Meléndez CM, Uribe D, Pedroza-Díaz J (2022) Synergistic effects of natural compounds and conventional chemotherapeutic agents: recent insights for the development of cancer treatment strategies. Heliyon. 8(6):e09519. https://doi.org/10.1016/j.heliyon.2022.e09519
Cayetano-Salazar L, Olea-Flores M, Zuñiga-Eulogio MD, Weinstein-Oppenheimer C, Fernández-Tilapa G, Mendoza-Catalán MA, Zacapala-Gómez AE, Ortiz-Ortiz J, Ortuño-Pineda C, Navarro-Tito N (2021) Natural isoflavonoids in invasive cancer therapy: from bench to bedside. Phytother Res. 35(8):4092–4110. https://doi.org/10.1002/ptr.7072
Chen X, Anderson JJ (2001) Isoflavones inhibit proliferation of ovarian cancer cells in vitro via an estrogen receptor-dependent pathway. Nutr Cancer. 41(1–2):165–71. https://doi.org/10.1080/01635581.2001.9680628
Chen Z, Miao H, Zhu Z, Zhang H, Huang H (2017) Daidzein induces apoptosis of non-small cell lung cancer cells by restoring STK4/YAP1 signaling. Int J Clin Exp Med 10(11):15205–15212 (IJCEM0022768)
Chou TC (2006) Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol Rev. 58(3):621–81. https://doi.org/10.1124/pr.58.3.10
Chou TC, Talalay P (1984) Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul. 22:27–55. https://doi.org/10.1016/0065-2571(84)90007-4
Cruz-Bermúdez A, Laza-Briviesca R, Vicente-Blanco RJ, García-Grande A, Coronado MJ, Laine-Menéndez S, Alfaro C, Sanchez JC, Franco F, Calvo V, Romero A, Martin-Acosta P, Salas C, Garcia JM, Provencio M (2019) Cancer-associated fibroblasts modify lung cancer metabolism involving ROS and TGF-β signaling. Free Radic Biol Med. 130:163–173. https://doi.org/10.1016/j.freeradbiomed.2018.10.450
Dagdemir A, Durif J, Ngollo M, Bignon YJ, Bernard-Gallon D (2013) Histone lysine trimethylation or acetylation can be modulated by phytoestrogen, estrogen or anti-HDAC in breast cancer cell lines. Epigenomics. 5(1):51–63. https://doi.org/10.2217/epi.12.74
Daware S, Patki M, Saraswat A, Palekar S, Patel K (2022) Development of a safe pediatric liquisolid self-nanoemulsifying system of triclabendazole for the treatment of fascioliasis. Int J Pharm. 15(626):122163. https://doi.org/10.1016/j.ijpharm.2022.122163
Demirtürk E, Ugur Kaplan AB, Cetin M, Akıllıoğlu K, DönmezKutlu M, Köse S, Aksu F (2022) Assessment of pharmacokinetic parameters of daidzein-containing nanosuspension and nanoemulsion formulations after oral administration to rats. Eur J Drug Metab Pharmacokinet. 47(2):247–257. https://doi.org/10.1007/s13318-021-00746-5
El-Badrawy MK, Yousef AM, Shaalan D, Elsamanoudy AZ (2014) Matrix metalloproteinase-9 expression in lung cancer patients and its relation to serum mmp-9 activity, pathologic type, and prognosis. J Bronchology Interv Pulmonol. 21(4):327–34. https://doi.org/10.1097/LBR.0000000000000094
Fu Y, Saraswat A, Wei Z, Agrawal MY, Dukhande VV, Reznik SE, Patel K (2021) Development of dual ARV-825 and nintedanib-loaded PEGylated nano-liposomes for synergistic efficacy in vemurafnib-resistant melanoma. Pharmaceutics. 13(7):1005. https://doi.org/10.3390/pharmaceutics13071005
Ganta S, Amiji M (2009) Coadministration of paclitaxel and curcumin in nanoemulsion formulations to overcome multidrug resistance in tumor cells. Mol Pharm. 6(3):928–39. https://doi.org/10.1021/mp800240j
Giampazolias E, Tait SWG (2018) Caspase-independent cell death: an anti-cancer double whammy. Cell Cycle. 17(3):269–270. https://doi.org/10.1080/15384101.2017.1408229
Gong L, Wu D, Zou J, Chen J, Chen L, Chen Y, Ni C, Yuan H (2016) Prognostic impact of serum and tissue MMP-9 in non-small cell lung cancer: a systematic review and meta-analysis. Oncotarget. 7(14):18458–68. https://doi.org/10.18632/oncotarget.7607
Gridelli C, Rossi A, Carbone DP, Guarize J, Karachaliou N, Mok T, Petrella F, Spaggiari L, Rosell R (2015) Non-small-cell lung cancer. Nat Rev Dis Primers. 1:15009. https://doi.org/10.1038/nrdp.2015.9
Guo J, Wang Q, Zhang Y, Sun W, Zhang S, Li Y, Wang J, Bao Y (2019) Functional daidzein enhances the anticancer effect of topotecan and reverses BCRP-mediated drug resistance in breast cancer. Pharmacol Res. 147:104387. https://doi.org/10.1016/j.phrs.2019.104387
Guo S, Wang Y, Li Y, Li Y, Feng C, Li Z (2020) Daidzein-rich isoflavones aglycone inhibits lung cancer growth through inhibition of NF-κB signaling pathway. Immunol Lett. 222:67–72. https://doi.org/10.1016/j.imlet.2020.03.004
He Y, Wu X, Cao Y, Hou Y, Chen H, Wu L, Lu L, Zhu W, Gu Y (2016) Daidzein exerts anti-tumor activity against bladder cancer cells via inhibition of FGFR3 pathway. Neoplasma. 63(4):523–31. https://doi.org/10.4149/neo_2016_405
Hu S, Li X, Xu R, Ye L, Kong H, Zeng X, Wang H, **e W (2016) The synergistic effect of resveratrol in combination with cisplatin on apoptosis via modulating autophagy in A549 cells. Acta Biochim Biophys Sin (Shanghai). 48(6):528–35. https://doi.org/10.1093/abbs/gmw026
Huang T, Wang Y, Shen Y, Ao H, Guo Y, Han M, Wang X (2020) Preparation of high drug-loading celastrol nanosuspensions and their anti-breast cancer activities in vitro and in vivo. Sci Rep. 10(1):8851. https://doi.org/10.1038/s41598-020-65773-9
Jafari A, Teymouri M, Ebrahimi Nik M, Abbasi A, Iranshahi M, Hanafi-Bojd MY, Jafari MR (2019) Interactive anticancer effect of nanomicellar curcumin and galbanic acid combination therapy with some common chemotherapeutics in colon carcinoma cells. Avicenna J Phytomed. 9(3):237–247
Kashaw SK, Agarwal S, Mishra M, Sau S, Iyer AK (2019) Molecular docking analysis of caspase-3 activators as potential anticancer agents. Curr Comput Aided Drug Des. 15(1):55–66. https://doi.org/10.2174/1573409914666181015150731
Kim BN, Ahn DH, Kang N, Yeo CD, Kim YK, Lee KY, Kim TJ, Lee SH, Park MS, Yim HW, Park JY, Park CK, Kim SJ (2020) TGF-β induced EMT and stemness characteristics are associated with epigenetic regulation in lung cancer. Sci Rep. 10(1):10597. https://doi.org/10.1038/s41598-020-67325-7
Korrodi-Gregório L, Soto-Cerrato V, Vitorino R, Fardilha M, Pérez-Tomás R (2016) From proteomic analysis to potential therapeutic targets: functional profile of two lung cancer cell lines, A549 and SW900, widely studied in pre-clinical research. PLoS One. 11(11):e0165973. https://doi.org/10.1371/journal.pone.0165973
Laddha AP, Kulkarni YA (2023) Pharmacokinetics, pharmacodynamics, toxicity, and formulations of daidzein: an important isoflavone. Phytother Res. 37(6):2578–2604. https://doi.org/10.1002/ptr.7852
Lee JS, Son KH, Sung MK, Kim YK, Yu R, Kim JS (2003) Anticarcinogenic properties of a daidzein-rich fraction isolated from soybean. J Med Food. 6(3):175–81. https://doi.org/10.1089/10966200360716580
Li J, Shen C, Wang X, Lai Y, Zhou K, Li P, Liu L, Che G (2019) Prognostic value of TGF-β in lung cancer: systematic review and meta-analysis. BMC Cancer. 19(1):691. https://doi.org/10.1186/s12885-019-5917-5
Liu R, Xu X, Liang C, Chen X, Yu X, Zhong H, Xu W, Cheng Y, Wang W, Wu Y, Yu L, Hu X (2019) ERβ modulates genistein’s cisplatin-enhancing activities in breast cancer MDA-MB-231 cells via P53-independent pathway. Mol Cell Biochem. 456(1–2):205–216. https://doi.org/10.1007/s11010-019-03505-y
Liu Y, Wu F, Ding Y, Zhu B, Su Y, Zhu X (2019) Preparation and characterization of paclitaxel/chitosan nanosuspensions for drug delivery system and cytotoxicity evaluation in vitro. Advanced Fiber Materials. 1:152–162
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 25(4):402–8. https://doi.org/10.1006/meth.2001.1262
Luo M, Lu Z, Sun H, Yuan K, Zhang Q, Meng S, Wang F, Guo H, Ju X, Liu Y, Ye T, Lu Z, Zhai Z (2010) Nuclear entry of active caspase-3 is facilitated by its p3-recognition-based specific cleavage activity. Cell Res. 20(2):211–22. https://doi.org/10.1038/cr.2010.9
Meng H, Fu G, Shen J, Shen K, Xu Z, Wang Y, ** B, Pan H (2017) Ameliorative effect of daidzein on cisplatin-induced nephrotoxicity in mice via modulation of inflammation, oxidative stress, and cell death. Oxid Med Cell Longev. 2017:3140680. https://doi.org/10.1155/2017/3140680
Mhone TG, Chen MC, Kuo CH, Shih TC, Yeh CM, Wang TF, Chen RJ, Chang YC, Kuo WW, Huang CY (2022) Daidzein synergizes with gefitinib to induce ROS/JNK/c-Jun activation and inhibit EGFR-STAT/AKT/ERK pathways to enhance lung adenocarcinoma cells chemosensitivity. Int J Biol Sci. 18(9):3636–3652. https://doi.org/10.7150/ijbs.71870
Montalesi E, Cipolletti M, Cracco P, Fiocchetti M, Marino M (2020) Divergent effects of daidzein and its metabolites on estrogen-induced survival of breast cancer cells. Cancers (Basel). 12(1):167. https://doi.org/10.3390/cancers12010167
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 65(1–2):55–63. https://doi.org/10.1016/0022-1759(83)90303-4
Negrette-Guzmán M (2019) Combinations of the antioxidants sulforaphane or curcumin and the conventional antineoplastics cisplatin or doxorubicin as prospects for anticancer chemotherapy. Eur J Pharmacol. 859:172513. https://doi.org/10.1016/j.ejphar.2019.172513
Rawat S, Pathak S, Gupta G, Singh SK, Singh H, Mishra A, Gilhotra R (2019) Recent updates on daidzein against oxidative stress and cancer. EXCLI J. 18:950–954. https://doi.org/10.17179/excli2019-1847
Salama AAA, Allam RM (2021) Promising targets of chrysin and daidzein in colorectal cancer: amphiregulin, CXCL1, and MMP-9. Eur J Pharmacol. 892:173763. https://doi.org/10.1016/j.ejphar.2020.173763
Saraswat AL, Maher TJ (2020) Development and optimization of stealth liposomal system for enhanced in vitro cytotoxic effect of quercetin. J Drug Deliv Sci Technol 55:101477. https://doi.org/10.1016/j.jddst.2019.101477
Saraswat A, Patki M, Fu Y, Barot S, Dukhande VV, Patel K (2020) Nanoformulation of proteolysis targeting chimera targeting “undruggable” c-Myc for the treatment of pancreatic cancer. Nanomedicine (Lond). 15(18):1761–1777. https://doi.org/10.2217/nnm-2020-0156
Saraswat A, Vemana HP, Dukhande VV, Patel K (2022) Galactose-decorated liver tumor-specific nanoliposomes incorporating selective BRD4-targeted PROTAC for hepatocellular carcinoma therapy. Heliyon. 8(1):e08702. https://doi.org/10.1016/j.heliyon.2021.e08702
Sauler M, Bazan IS, Lee PJ (2019) Cell death in the lung: the apoptosis-necroptosis axis. Annu Rev Physiol. 81:375–402. https://doi.org/10.1146/annurev-physiol-020518-114320
Schnekenburger M, Diederich M (2015) Chapter 18 - nutritional epigenetic regulators in the field of cancer: new avenues for chemopreventive approaches, Editor(s): Steven G. Gray, Epigenetic Cancer Therapy,Academic Press, Pages 393-425, https://doi.org/10.1016/B978-0-12-800206-3.00018-5
Singh S, Grewal S, Sharma N, Behl T, Gupta S, Anwer MK, Vargas-De-La-Cruz C, Mohan S, Bungau SG, Bumbu A (2023) Unveiling the pharmacological and nanotechnological facets of daidzein: present state-of-the-art and future perspectives. Molecules. 28(4):1765. https://doi.org/10.3390/molecules28041765
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 71(3):209–249. https://doi.org/10.3322/caac.21660
Thadkala K, Nanam PK, Rambabu B, Sailu C, Aukunuru J (2014) Preparation and characterization of amorphous ezetimibe nanosuspensions intended for enhancement of oral bioavailability. Int J Pharm Investig. 4(3):131–7. https://doi.org/10.4103/2230-973X.138344
Uğur Kaplan AB, Öztürk N, Çetin M, Vural İ, ÖznülüerÖzer T (2022) The nanosuspension formulations of daidzein: preparation and in vitro characterization. Turk J Pharm Sci. 19(1):84–92. https://doi.org/10.4274/tjps.galenos.2021.81905
Uifălean A, Schneider S, Ionescu C, Lalk M, Iuga CA (2015) Soy isoflavones and breast cancer cell lines: molecular mechanisms and future perspectives. Molecules. 21(1):E13. https://doi.org/10.3390/molecules21010013
Vartak R, Saraswat A, Yang Y, Chen ZS, Patel K (2022) Susceptibility of lung carcinoma cells to nanostructured lipid carrier of ARV-825, a BRD4 degrading proteolysis targeting chimera. Pharm Res. 39(11):2745–2759. https://doi.org/10.1007/s11095-022-03184-3
Wang H, **ao Y, Wang H, Sang Z, Han X, Ren S, Du R, Shi X, **e Y (2019) Development of daidzein nanosuspensions: preparation, characterization, in vitro evaluation, and pharmacokinetic analysis. Int J Pharm. 20(566):67–76. https://doi.org/10.1016/j.ijpharm.2019.05.051
Wang L, Correa CR, Hayman JA, Zhao L, Cease K, Brenner D, Arenberg D, Curtis J, Kalemkerian GP, Kong FM (2009) Time to treatment in patients with stage III non-small cell lung cancer. Int J Radiat Oncol Biol Phys. 74(3):790–5. https://doi.org/10.1016/j.ijrobp.2008.08.039
Wang X, Hua P, He C, Chen M (2022) Non-apoptotic cell death-based cancer therapy: molecular mechanism, pharmacological modulators, and nanomedicine. Acta Pharm Sin B. 12(9):3567–3593. https://doi.org/10.1016/j.apsb.2022.03.020
Zappa C, Mousa SA (2016) Non-small cell lung cancer: current treatment and future advances. Transl Lung Cancer Res 5(3):288–300. https://doi.org/10.21037/tlcr.2016.06.07
Zhang H, Zhao B, Zhai ZG, Zheng JD, Wang YK, Zhao YY (2021) Expression and clinical significance of MMP-9 and P53 in lung cancer. Eur Rev Med Pharmacol Sci. 25(3):1358–1365. https://doi.org/10.26355/eurrev_202102_24844
Zhang M, Zhang YY, Chen Y, Wang J, Wang Q, Lu H (2021) TGF-β signaling and resistance to cancer therapy. Front Cell Dev Biol. 9:786728. https://doi.org/10.3389/fcell.2021.786728
Zhao Y, Huan ML, Liu M, Cheng Y, Sun Y, Cui H, Liu DZ, Mei QB, Zhou SY (2016) Doxorubicin and resveratrol co-delivery nanoparticle to overcome doxorubicin resistance. Sci Rep. 6:35267. https://doi.org/10.1038/srep35267
Zheng W, Liu T, Sun R, Yang L, An R, Xue Y (2018) Daidzein induces choriocarcinoma cell apoptosis in a dose-dependent manner via the mitochondrial apoptotic pathway. Mol Med Rep. 17(4):6093–6099. https://doi.org/10.3892/mmr.2018.8604
Acknowledgements
The authors thank Afyonkarahisar Health Sciences University Scientific Research Projects Coordination Unit for financial support.
Funding
This study was supported by Afyonkarahisar Health Sciences University Scientific Research Projects Coordination Unit under grant number 22.GENERAL.009.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation by [Afife Busra Ugur-Kaplan] and [Meltem Cetin], data collection and analysis were performed by [Seyma Oncu], [Merve Becit-Kizilkaya], [Serkan Sen] and [Sefa Celik]. The first draft of the manuscript was written by [Seyma Oncu] and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This is an in vitro study. Afyonkarahisar University of Health Sciences clinical research ethics committee approved that no ethical approval is required.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Oncu, S., Becit-Kizilkaya, M., Sen, S. et al. Daidzein nanosuspension in combination with cisplatin to enhance therapeutic efficacy against A549 non-small lung cancer cells: an in vitro evaluation. Naunyn-Schmiedeberg's Arch Pharmacol 397, 4871–4881 (2024). https://doi.org/10.1007/s00210-023-02924-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02924-5