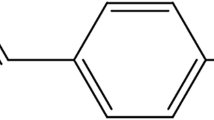
Abstract
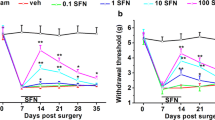
The current pharmacotherapy of neuropathic pain is inadequate as neuropathic pain involves varied clinical manifestations with multifactorial etiology, modulated by a cascade of physical and molecular events leading to different clinical presentations of pain. There is an accumulating evidence of the involvement of oxidative stress in neuropathy, and antioxidants have shown promise in mitigating neuropathic pain syndromes. To explore the evidence supporting this beneficial proclivity of antioxidants, this study investigated the antinociceptive effectiveness of N-(2-mercaptopropionyl)glycine or tiopronin, a well-recognized aminothiol antioxidant, in a refined chronic constriction injury (CCI) rat model of neuropathic pain. Tiopronin (10, 30, and 90 mg/kg, i.p.) and pregabalin (30 mg/kg, i.p.) were administered daily after CCI surgery. The neuropathic paradigms of mechanical/cold allodynia and mechanical/heat hyperalgesia were assessed on days 3, 7, 14, and 21 post-nerve ligation. At the end of study, malondialdehyde (MDA), superoxide dismutase (SOD), catalase (CAT), and glutathione (GSH) levels were estimated in the sciatic nerve, dorsal root ganglion, and spinal cord for assessing the extent of oxidative stress. The expression of neuropathic nociception was attenuated by tiopronin which was observed as a significant attenuation of CCI-induced allodynia and hyperalgesia. Tiopronin reversed the neuronal oxidative stress by significantly reducing MDA, and increasing SOD, CAT, and GSH levels. Pregabalin also showed similar beneficial propensity on CCI-induced neuropathic aberrations. These findings suggest prospective neuropathic pain attenuating efficacy of tiopronin and further corroborated the notion that antioxidants are effective in mitigating the development and expression of neuropathic pain and underlying neuronal oxidative stress.








Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the article (and its supplementary information file).
Change history
07 December 2020
A Correction to this paper has been published: https://doi.org/10.1007/s00210-020-02033-7
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/s0076-6879(84)05016-3
Ahmad N, Subhan F, Islam NU, Shahid M, Rahman FU, Fawad K (2017a) A novel pregabalin functionalized salicylaldehyde derivative afforded prospective pain, inflammation, and pyrexia alleviating propensities. Arch Pharm 350:e1600365. https://doi.org/10.1002/ardp.201600365
Ahmad N, Subhan F, Islam NU, Shahid M, Rahman FU, Sewell RD (2017b) Gabapentin and its salicylaldehyde derivative alleviate allodynia and hypoalgesia in a cisplatin-induced neuropathic pain model. Eur J Pharmacol 814:302–312. https://doi.org/10.1016/j.ejphar.2017.08.040
Akbar S, Subhan F, Karim N, Shahid M, Ahmad N, Ali G, Mahmood W, Fawad K (2016) 6-Methoxyflavanone attenuates mechanical allodynia and vulvodynia in the streptozotocin-induced diabetic neuropathic pain. Biomed Pharmacother 84:962–971. https://doi.org/10.1016/j.biopha.2016.10.017
Akbar S, Subhan F, Karim N, Aman U, Ullah S, Shahid M, Ahmad N, Fawad K, Sewell RD (2017) Characterization of 6-methoxyflavanone as a novel anxiolytic agent: a behavioral and pharmacokinetic approach. Eur J Pharmacol 801:19–27. https://doi.org/10.1016/j.ejphar.2017.02.047
Akbar S, Subhan F, Shahid M, Wadood A, Shahbaz N, Farooq U, Ayaz M, Raziq N (2020) 6-Methoxyflavanone abates cisplatin-induced neuropathic pain apropos anti-inflammatory mechanisms: a behavioral and molecular simulation study. Eur J Pharmacol: 172972. https://doi.org/10.1016/j.ejphar.2020.172972
Akrivis C, Vezyraki P, Kiortsis D, Fotopoulos A, Evangelou A (2000) Inhibition of puerperal lactation with 2-mercaptopropionyl-glycine. Eur J Clin Pharmacol 56:621–623. https://doi.org/10.1007/s002280000228
Alam J, Subhan F, Ullah I, Shahid M, Ali G, Sewell RD (2017) Synthetic and natural antioxidants attenuate cisplatin-induced vomiting. BMC Pharmacol Toxicol 18:4. https://doi.org/10.1186/s40360-016-0110-9
Ali G, Subhan F, Abbas M, Zeb J, Shahid M, Sewell RD (2015) A streptozotocin-induced diabetic neuropathic pain model for static or dynamic mechanical allodynia and vulvodynia: validation using topical and systemic gabapentin. Naunyn Schmiedeberg's Arch Pharmacol 388:1129–1140. https://doi.org/10.1007/s00210-015-1145-y
Aman U, Subhan F, Shahid M, Akbar S, Ahmad N, Ali G, Fawad K, Sewell RD (2016) Passiflora incarnata attenuation of neuropathic allodynia and vulvodynia apropos GABA-ergic and opioidergic antinociceptive and behavioural mechanisms. BMC Complement Altern Med 16:77. https://doi.org/10.1186/s12906-016-1048-6
Ametov AS, Barinov A, Dyck PJ, Hermann R, Kozlova N, Litchy WJ, Low PA, Nehrdich D, Novosadova M, O’Brien PC (2003) The sensory symptoms of diabetic polyneuropathy are improved with α-lipoic acid. Diabetes Care 26:770–776. https://doi.org/10.2337/diacare.26.3.770
Amor B, Mery C, Gery AD (1982) Tiopronin (N-[2-mercaptopropionyl] glycin) in rheumatoid arthritis. Arthritis Rheum 25:698–703. https://doi.org/10.1002/art.1780250614
Arcos M, Palanca JM, Montes F, Barrios C (2013) Antioxidants and gabapentin prevent heat hypersensitivity in a neuropathic pain model. J Investig Surg 26:109–117. https://doi.org/10.3109/08941939.2012.713444
Areti A, Yerra VG, Naidu V, Kumar A (2014) Oxidative stress and nerve damage: role in chemotherapy induced peripheral neuropathy. Redox Biol 2:289–295. https://doi.org/10.1016/j.redox.2014.01.006
Aşcı S, Demirci S, Aşcı H, Doğuç DK, Onaran İ (2016) Neuroprotective effects of pregabalin on cerebral ischemia and reperfusion. Balkan Med J 33:221–227. https://doi.org/10.5152/balkanmedj.2015.15742
Attal N (2019) Pharmacological treatments of neuropathic pain: the latest recommendations. Rev Neurol (Paris) 175:46–50. https://doi.org/10.1016/j.neurol.2018.08.005
Austin PJ, Wu A, Moalem-Taylor G (2012) Chronic constriction of the sciatic nerve and pain hypersensitivity testing in rats. J Vis Exp Mar 13(61):e3393. https://doi.org/10.3791/3393
Ayaz M, Junaid M, Ullah F, Subhan F, Sadiq A, Ali G, Ovais M, Shahid M, Ahmad A, Wadood A (2017) Anti-Alzheimer’s studies on β-sitosterol isolated from Polygonum hydropiper L. Front Pharmacol 8:697. https://doi.org/10.3389/fphar.2017.00697
Ayaz M, Sadiq A, Junaid M, Ullah F, Ovais M, Ullah I, Ahmed J, Shahid M (2019) Flavonoids as prospective neuroprotectants and their therapeutic propensity in aging associated neurological disorders. Front Aging Neurosci 11:155. https://doi.org/10.3389/fnagi.2019.00155
Bennett GJ, **e Y-K (1988) A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 33:87–107. https://doi.org/10.1016/0304-3959(88)90209-6
Bertolotto F, Massone A (2012) Combination of alpha lipoic acid and superoxide dismutase leads to physiological and symptomatic improvements in diabetic neuropathy. Drugs R D 12:29–34. https://doi.org/10.2165/11599200-000000000-00000
Bhat RA, Lingaraju MC, Pathak NN, Kalra J, Kumar D, Kumar D, Tandan SK (2016) Effect of ursolic acid in attenuating chronic constriction injury-induced neuropathic pain in rats. Fundam Clin Pharmacol 30:517–528. https://doi.org/10.1111/fcp.12223
Campbell JN, Meyer RA (2006) Mechanisms of neuropathic pain. Neuron 52:77–92. https://doi.org/10.1016/j.neuron.2006.09.021
Carrasco C, Naziroǧlu M, Rodríguez AB, Pariente JA (2018) Neuropathic pain: delving into the oxidative origin and the possible implication of transient receptor potential channels. Front Physiol 9:95. https://doi.org/10.3389/fphys.2018.00095
Chaplan S, Bach F, Pogrel J, Chung J, Yaksh T (1994) Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 53:55–63. https://doi.org/10.1016/0165-0270(94)90144-9
Choi DC, Lee JY, Lim EJ, Baik HH, Oh TH, Yune TY (2012) Inhibition of ROS-induced p38MAPK and ERK activation in microglia by acupuncture relieves neuropathic pain after spinal cord injury in rats. Exp Neurol 236:268–282. https://doi.org/10.1016/0165-0270(94)90144-9
Chong MS, Bajwa ZH (2003) Diagnosis and treatment of neuropathic pain. J Pain Symptom Manag 25:S4–S11. https://doi.org/10.1016/s0885-3924(03)00064-2
Chung W-s, Go Y-G, Lee W-h (2008) Antinociceptive effect of intraperitoneally administered 5, 5-dimethyl-1-pyrroline N-oxide on formalin induced nociception in rats. Korean J Anesthesiol 54:S35–S39. https://doi.org/10.4097/kjae.2008.54.3.S35
Çivi S, Emmez G, Dere ÜA, Börcek AÖ, Emmez H (2016) Effects of quercetin on chronic constriction nerve injury in an experimental rat model. Acta Neurochir 158:959–965. https://doi.org/10.1007/s00701-016-2761-0
Cojocaru IM, Cojocaru M, Silosi I, Vrabie CD (2014) Peripheral nervous system manifestations in systemic autoimmune diseases. Maedica (Buchar) 9:289–294
Date M-o, Morita T, Yamashita N, Nishida K, Yamaguchi O, Higuchi Y, Hirotani S, Matsumura Y, Hori M, Tada M (2002) The antioxidant N-2-mercaptopropionyl glycine attenuates left ventricular hypertrophy in in vivo murine pressure-overload model. J Am Coll Cardiol 39:907–912. https://doi.org/10.1016/S0735-1097(01)01826-5
De Vry J, Kuhl E, Franken-Kunkel P, Eckel G (2004) Pharmacological characterization of the chronic constriction injury model of neuropathic pain. Eur J Pharmacol 491:137–148. https://doi.org/10.1016/j.ejphar.2004.03.051
Dworkin RH, Backonja M, Rowbotham MC, Allen RR, Argoff CR, Bennett GJ, Bushnell MC, Farrar JT, Galer BS, Haythornthwaite JA (2003) Advances in neuropathic pain: diagnosis, mechanisms, and treatment recommendations. Arch Neurol 60:1524–1534. https://doi.org/10.1001/archneur.60.11.1524
Evangelou A, Karkabounas S, Kalpouzos G, Malamas M, Liasko R, Stefanou D, Vlahos AT, Kabanos TA (1997) Comparison of the therapeutic effects of two vanadium complexes administered at low doses on benzo [a] pyrene-induced malignant tumors in rats. Cancer Lett 119:221–225. https://doi.org/10.1016/S0304-3835(97)00278-4
Fujimoto T, Fuyuta M, Kiyofuji E, Hirata S (1979) Prevention by tiopronin (2-mercaptopropionyl glycine) of methylmercuric chloride-induced teratogenic and fetotoxic effects in mice. Teratology 20:297–301. https://doi.org/10.1002/tera.1420200213
Gilron I, Baron R, Jensen T (2015) Neuropathic pain: principles of diagnosis and treatment. Mayo Clin Proc 90:532–545. https://doi.org/10.1016/j.mayocp.2015.01.018
Gopalsamy B, Sambasevam Y, Zulazmi NA, Chia JSM, Farouk AAO, Sulaiman MR, Mohamad TAST, Perimal EK (2019) Experimental characterization of the chronic constriction injury-induced neuropathic pain model in mice. Neurochem Res 44:2123–2138. https://doi.org/10.1007/s11064-019-02850-0
Guedes RP, Dal Bosco L, Teixeira CM, Araújo AS, Llesuy S, Belló-Klein A, Ribeiro MFM, Partata WA (2006) Neuropathic pain modifies antioxidant activity in rat spinal cord. Neurochem Res 31:603–609. https://doi.org/10.1007/s11064-006-9058-2
Gustin S, Wrigley P, Siddall P, Henderson L (2010) Brain anatomy changes associated with persistent neuropathic pain following spinal cord injury. Cereb Cortex 20:1409–1419. https://doi.org/10.1093/cercor/bhp205
Hahm JR, Kim B-J, Kim K-W (2004) Clinical experience with thioctacid (thioctic acid) in the treatment of distal symmetric polyneuropathy in Korean diabetic patients. J Diabetes Complicat 18:79–85. https://doi.org/10.1016/s1056-8727(03)00033-3
Han CG, Han JK, Park KB, Kwak KH, Park SS, Lim DG (2012) Effect of superoxide on the development and maintenance of mechanical allodynia in a rat model of chronic post-ischemia pain. Korean J Anesthesiol 63:149–156. https://doi.org/10.4097/kjae.2012.63.2.149
Herzberg D, Strobel P, Chihuailaf R, Ramirez-Reveco A, Müller H, Werner M, Bustamante H (2019) Spinal reactive oxygen species and oxidative damage mediate chronic pain in lame dairy cows. Animals (Basel) 9:693. https://doi.org/10.3390/ani9090693
IASP (1994) Pain terms: a current list with definitions and notes on usage. Classification of Chronic Pain, 2nd edition, IASP Task Force on Taxonomy, eds H. Merskey, H., Bogduk, N. IASP Press, Washington DC, pp. 209-214. updated 2014. http://www.iasp-pain.org
Ichikawa H, Imaizumi K, Tazawa Y, Obara Y, Ishikawa Y, Tobari I, Tanabe Y (1980) Effect of tiopronin on senile cataracts. Ophthalmologica 180:293–298. https://doi.org/10.1159/000308990
Islam NU, Khan I, Rauf A, Muhammad N, Shahid M, Shah MR (2015) Antinociceptive, muscle relaxant and sedative activities of gold nanoparticles generated by methanolic extract of Euphorbia milii. BMC Complement Altern Med 15:160. https://doi.org/10.1186/s12906-015-0691-7
Islam NU, Amin R, Shahid M, Amin M, Zaib S, Iqbal J (2017) A multi-target therapeutic potential of Prunus domestica gum stabilized nanoparticles exhibited prospective anticancer, antibacterial, urease-inhibition, anti-inflammatory and analgesic properties. BMC Complement Altern Med 17:276. https://doi.org/10.1186/s12906-017-1791-3
Islam NU, Amin R, Shahid M, Amin M (2019a) Gummy gold and silver nanoparticles of apricot (Prunus armeniaca) confer high stability and biological activity. Arab J Chem 12:3977–3992. https://doi.org/10.1016/j.arabjc.2016.02.017
Islam NU, Jalil K, Shahid M, Muhammad N, Rauf A (2019b) Pistacia integerrima gall extract mediated green synthesis of gold nanoparticles and their biological activities. Arab J Chem 12:2310–2319. https://doi.org/10.1016/j.arabjc.2015.02.014
Islam NU, Jalil K, Shahid M, Rauf A, Muhammad N, Khan A, Shah MR, Khan MA (2019c) Green synthesis and biological activities of gold nanoparticles functionalized with Salix alba. Arab J Chem 12:2914–2925. https://doi.org/10.1016/j.arabjc.2015.06.025
Ivanova S, Batliwalla F, Mocco J, Kiss S, Huang J, Mack W, Coon A, Eaton JW, Al-Abed Y, Gregersen PK (2002) Neuroprotection in cerebral ischemia by neutralization of 3-aminopropanal. Proc Natl Acad Sci 99:5579–5584. https://doi.org/10.1073/pnas.082609299
Janes K, Neumann WL, Salvemini D (2012) Anti-superoxide and anti-peroxynitrite strategies in pain suppression. Biochim Biophys Acta 1822:815–821. https://doi.org/10.1016/j.bbadis.2011.12.008
Jensen TS, Finnerup NB (2014) Allodynia and hyperalgesia in neuropathic pain: clinical manifestations and mechanisms. Lancet Neurol 13:924–935. https://doi.org/10.1016/s1474-4422(14)70102-4
Ji C, Xu Y, Han F, Sun D, Zhang H, Li X, Yao X, Wang H (2017) Quercetin alleviates thermal and cold hyperalgesia in a rat neuropathic pain model by inhibiting Toll-like receptor signaling. Biomed Pharmacother 94:652–658. https://doi.org/10.1016/j.biopha.2017.07.145
Jia D, Wang H, Han B, Zhang L, Guo J (2019) Tempol attenuates neuropathic pain by inhibiting nitric oxide production. Anal Cell Pathol 2019: 8253850. https://doi.org/10.1155/2019/8253850 8253855
Jorige A, Annapurna A (2016) Neuroprotective and antioxidant role of pregabalin in streptozotocin induced neurotoxicity. Int J Pharm Sci Res 7:4494. https://doi.org/10.13040/IJPSR.0975-8232.7
Kaulaskar S, Bhutada P, Rahigude A, Jain D, Harle U (2012) Effects of naringenin on allodynia and hyperalgesia in rats with chronic constriction injury-induced neuropathic pain. Zhong ** Yi Jie He Xue Bao 10:1482–1489. https://doi.org/10.3736/jcim20121223
Khan A, Naz S, Farooq U, Shahid M, Ullah I, Ali I, Rauf A, Mabkhot YN (2018) Bioactive chromone constituents from Vitex negundo alleviate pain and inflammation. J Pain Res 11:95–102. https://doi.org/10.2147/JPR.S145551
Kim HJ (2015) Berberine ameliorates allodynia induced by chronic constriction injury of the sciatic nerve in rats. J Med Food 18:909–915. https://doi.org/10.1089/jmf.2014.3346
Kim HK, Park SK, Zhou J-L, Taglialatela G, Chung K, Coggeshall RE, Chung JM (2004) Reactive oxygen species (ROS) play an important role in a rat model of neuropathic pain. Pain 111:116–124. https://doi.org/10.1016/j.pain.2004.06.008
Kim HK, Kim JH, Gao X, Zhou J-L, Lee I, Chung K, Chung JM (2006) Analgesic effect of vitamin E is mediated by reducing central sensitization in neuropathic pain. Pain 122:53–62. https://doi.org/10.1016/j.pain.2006.01.013
Kim D, You B, Jo E-K, Han S-K, Simon MI, Lee SJ (2010a) NADPH oxidase 2-derived reactive oxygen species in spinal cord microglia contribute to peripheral nerve injury-induced neuropathic pain. Proc Natl Acad Sci U S A 107:14851–14856. https://doi.org/10.1073/pnas.1009926107
Kim GH, Kellner CP, Hickman ZL, Zacharia BE, Starke RM, Hwang BY, Ducruet AF, Fernandez L, Mayer SA, Tracey KJ (2010b) A phase I clinical trial of tiopronin, a putative neuroprotective agent, in aneurysmal subarachnoid hemorrhage. Neurosurgery 67:182–186. https://doi.org/10.1227/01.NEU.0000370919.93259.3C
Kim HK, Zhang YP, Gwak YS, Abdi S (2010c) Phenyl N-tert-butylnitrone, a free radical scavenger, reduces mechanical allodynia in chemotherapy-induced neuropathic pain in rats. Anesthesiology 112:432–439. https://doi.org/10.1097/aln.0b013e3181ca31bd
Kim H, Thompson J, Ji G, Ganapathy V, Neugebauer V (2017) Monomethyl fumarate (MMF) inhibits pain behaviors and amygdala activity in a rat arthritis model. Pain 158:2376–2385. https://doi.org/10.1097/j.pain.0000000000001042
Komirishetty P, Areti A, Gogoi R, Sistla R, Kumar A (2017) Combination strategy of PARP inhibitor with antioxidant prevent bioenergetic deficits and inflammatory changes in CCI-induced neuropathy. Neuropharmacology 113:137–147. https://doi.org/10.1016/j.neuropharm.2016.09.027
Kono Y (1978) Generation of superoxide radical during autoxidation of hydroxylamine and an assay for superoxide dismutase. Arch Biochem Biophys 186:189–195. https://doi.org/10.1016/0003-9861(78)90479-4
Kumar A, Kaur H, Singh A (2018) Neuropathic pain models caused by damage to central or peripheral nervous system. Pharmacol Rep 70:206–216. https://doi.org/10.1016/j.pharep.2017.09.009
Lammel B, Pilz S, Freisleben H-J, Schraven E, Zimmer G (1987) Free radical scavenging drugs, assessed by ESR studies: influence of hemoglobin. Free Radic Res Commun 3:331–335. https://doi.org/10.3109/10715768709088073
Ma F, Zhang L, Westlund KN (2009) Reactive oxygen species mediate TNFR1 increase after TRPV1 activation in mouse DRG neurons. Mol Pain 5:31. https://doi.org/10.1186/1744-8069-5-31
Mbiya W, Adigun R, Tran T, Htwe Y, Simoyi RH (2015) Oxyhalogen–sulfur chemistry: kinetics and mechanism of oxidation of tiopronin by acidified bromate and aqueous bromine. Aust J Chem 68:262–272. https://doi.org/10.1071/CH14126
Montague K, Malcangio M (2017) The therapeutic potential of monocyte/macrophage manipulation in the treatment of chemotherapy-induced painful neuropathy. Front Mol Neurosci 10:397. https://doi.org/10.3389/fnmol.2017.00397
Moron MS, Depierre JW, Mannervik B (1979) Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. Biochim Biophys Acta 582:67–78. https://doi.org/10.1016/0304-4165(79)90289-7
Naik AK, Tandan SK, Dudhgaonkar SP, Jadhav SH, Kataria M, Prakash VR, Kumar D (2006) Role of oxidative stress in pathophysiology of peripheral neuropathy and modulation by N-acetyl-L-cysteine in rats. Eur J Pain 10:573–579. https://doi.org/10.1016/j.ejpain.2005.08.006
Nawaz NUA, Saeed M, Rauf K, Usman M, Arif M, Ullah Z, Raziq N (2018) Antinociceptive effectiveness of Tithonia tubaeformis in a vincristine model of chemotherapy-induced painful neuropathy in mice. Biomed Pharmacother 103:1043–1051. https://doi.org/10.1016/j.biopha.2018.04.115
Nishigori H, Hayashi R, Lee JW, Iwatsuru M (1984) Effect of MPG on glucocorticoid-induced cataract formation in develo** chick embryo. Invest Ophthalmol Vis Sci 25:1051–1055
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Ohri S, Sharma D, Dixit A (2002) Modulation of c-myc and c-fos gene expression in regenerating rat liver by 2-mercaptopropionylglycine. Cell Biol Int 26:187–192. https://doi.org/10.1006/cbir.2001.0825
Oyenihi AB, Ayeleso AO, Mukwevho E, Masola B (2015) Antioxidant strategies in the management of diabetic neuropathy. Biomed Res Int 2015:515042–515015. https://doi.org/10.1155/2015/515042
Qin F-J, Hu X-H, Chen Z, Chen X, Shen Y-M (2019) Protective effects of tiopronin against oxidative stress in severely burned patients. Drug Des Devel Ther 13:2827–2832. https://doi.org/10.2147/DDDT.S215927
Rajanandh MG, Kosey S, Prathiksha G (2014) Assessment of antioxidant supplementation on the neuropathic pain score and quality of life in diabetic neuropathy patients—a randomized controlled study. Pharmacol Rep 66:44–48. https://doi.org/10.1016/j.pharep.2013.08.003
Raziq N, Saeed M, Shahid M, Muhammad N, Khan H, Gul F (2016) Pharmacological basis for the use of Hypericum oblongifolium as a medicinal plant in the management of pain, inflammation and pyrexia. BMC Complement Altern Med 16:41. https://doi.org/10.1186/s12906-016-1018-z
Riffel APK, de Souza JA, Maria do Carmo QS, Horst A, Scheid T, Kolberg C, Belló-Klein A, Partata WA (2016) Systemic administration of vitamins C and E attenuates nociception induced by chronic constriction injury of the sciatic nerve in rats. Brain Res Bull 121:169–177. https://doi.org/10.1016/j.brainresbull.2016.02.004
Riffel APK, Santos MCQ, de Souza J, Scheid T, Horst A, Kolberg C, Belló-Klein A, Partata WA (2018) Treatment with ascorbic acid and α-tocopherol modulates oxidative-stress markers in the spinal cord of rats with neuropathic pain. Braz J Med Biol Res 51:e7097. https://doi.org/10.1590/1414-431x20177097
Rokyta R, Holecek V, Pekárková I, Krejcová J, Racek J, Trefil L, Yamamotová A (2003) Free radicals after painful stimulation are influenced by antioxidants and analgesics. Neuro Endocrinol Lett 24:304–309
Rukh L, Ali G, Ullah R, Islam NU, Shahid M (2020) Efficacy assessment of salicylidene salicylhydrazide in chemotherapy associated peripheral neuropathy. Eur J Pharmacol 888:173481. https://doi.org/10.1016/j.ejphar.2020.173481
Saffarpour S, Nasirinezhad F (2019) Ascorbic acid eliminated pain-induced peripheral neuropathy by modulation of nitric oxide pathway in rats. Nutrire 44:8. https://doi.org/10.1186/s41110-019-0098-1
Sagalajev B, Wei H, Chen Z, Albayrak I, Koivisto A, Pertovaara A (2018) Oxidative stress in the amygdala contributes to neuropathic pain. Neuroscience 387:92–103. https://doi.org/10.1016/j.neuroscience.2017.12.009
Salvemini D, Little JW, Doyle T, Neumann WL (2011) Roles of reactive oxygen and nitrogen species in pain. Free Radic Biol Med 51:951–966. https://doi.org/10.1016/j.freeradbiomed.2011.01.026
Shahid M, Subhan F, Ullah I, Ali G, Alam J, Shah R (2016) Beneficial effects of Bacopa monnieri extract on opioid induced toxicity. Heliyon 2:e00068. https://doi.org/10.1016/j.heliyon.2016.e00068
Shahid M, Subhan F, Ahmad N, Ali G, Akbar S, Fawad K, Sewell RD (2017a) Topical gabapentin gel alleviates allodynia and hyperalgesia in the chronic sciatic nerve constriction injury neuropathic pain model. Eur J Pain 21:668–680. https://doi.org/10.1002/ejp.971
Shahid M, Subhan F, Ahmad N, Sewell RDE (2017b) The flavonoid 6-methoxyflavone allays cisplatin-induced neuropathic allodynia and hypoalgesia. Biomed Pharmacother 95:1725–1733. https://doi.org/10.1016/j.biopha.2017.09.108
Shahid M, Subhan F, Ahmad N, Ullah I (2017c) A bacosides containing Bacopa monnieri extract alleviates allodynia and hyperalgesia in the chronic constriction injury model of neuropathic pain in rats. BMC Complement Altern Med 17:293. https://doi.org/10.1186/s12906-017-1807-z
Shahid M, Subhan F, Ali G, Ullah I, Alam J, Ullah S, Rauf K (2017d) Neuroprotective effect of Bacopa monnieri against morphine-induced histopathological changes in the cerebellum of rats. Pak J Pharm Sci 30:2067–2074
Shahid M, Subhan F, Ahmad N, Sewell RDE (2019a) Efficacy of a topical gabapentin gel in a cisplatin paradigm of chemotherapy-induced peripheral neuropathy. BMC Pharmacol Toxicol 20:51. https://doi.org/10.1186/s40360-019-0329-3
Shahid M, Subhan F, Islam NU, Ullah I, Alam J, Ahmad N, Ali G (2019b) Phytochemical, pharmacological and therapeutic profile of Bacopa monnieri: a prospective complementary medicine. Clinical Efficacy, Safety and Quality. CRC Press, Taylor & Francis Group, Florida, USA, Natural Medicines, pp 447–476
Tal M (1996) A novel antioxidant alleviates heat hyperalgesia in rats with an experimental painful peripheral neuropathy. Neuroreport 7:1382–1384. https://doi.org/10.1097/00001756-199605310-00010
Talukdar R, Lakhtakia S, Reddy DN, Rao GV, Pradeep R, Banerjee R, Gupta R, Ramchandani M, Tandan M, Murthy HV (2016) Ameliorating effect of antioxidants and pregabalin combination in pain recurrence after ductal clearance in chronic pancreatitis: results of a randomized, double blind, placebo-controlled trial. J Gastroenterol Hepatol 31:1654–1662. https://doi.org/10.1111/jgh.13332
Tan EC, Bahrami S, Kozlov AV, Kurvers HA, Ter Laak HJ, Nohl H, Redl H, Goris RJA (2009) The oxidative response in the chronic constriction injury model of neuropathic pain. J Surg Res 152:84–88. https://doi.org/10.1016/j.jss.2008.03.035
Tanonaka K, Iwai T, Motegi K, Takeo S (2003) Effects of N-(2-mercaptopropionyl)-glycine on mitochondrial function in ischemic–reperfused heart. Cardiovasc Res 57:416–425. https://doi.org/10.1016/S0008-6363(02)00698-3
Tiwari AK (2001) Imbalance in antioxidant defense and human diseases: multiple approach of natural antioxidants therapy. Curr Sci 81:1179–1187
Ullah I, Subhan F, Rudd JA, Rauf K, Alam J, Shahid M, Sewell R (2014) Attenuation of cisplatin-induced emetogenesis by standardized Bacopa monnieri extracts in the pigeon: behavioral and neurochemical correlations. Planta Med 80:1569–1579. https://doi.org/10.1055/s-0034-1383121
Yang S, Chang MC (2019) Chronic pain: structural and functional changes in brain structures and associated negative affective states. Int J Mol Sci 20:3130. https://doi.org/10.3390/ijms20133130
Yowtak J, Lee KY, Kim HY, Wang J, Kim HK, Chung K, Chung JM (2011) Reactive oxygen species contribute to neuropathic pain by reducing spinal GABA release. Pain 152:844–852. https://doi.org/10.1016/j.pain.2010.12.034
Yu X, Liu H, Hamel KA, Morvan MG, Yu S, Leff J, Guan Z, Braz JM, Basbaum AI (2020) Dorsal root ganglion macrophages contribute to both the initiation and persistence of neuropathic pain. Nat Commun 11:264. https://doi.org/10.1038/s41467-019-13839-2
Zhang J-G, Viale M, Esposito M, Lindup W (1999) Tiopronin protects against the nephrotoxicity of cisplatin in the rat. Hum Exp Toxicol 18:713–717. https://doi.org/10.1191/096032799678839644
Zhang G, Liu N, Zhu C, Ma L, Yang J, Du J, Zhang W, Sun T, Niu J, Yu J (2019) Antinociceptive effect of isoorientin against neuropathic pain induced by the chronic constriction injury of the sciatic nerve in mice. Int Immunopharmacol 75:105753. https://doi.org/10.1016/j.intimp.2019.105753
Ziegler D, Low PA, Litchy WJ, Boulton AJ, Vinik AI, Freeman R, Samigullin R, Tritschler H, Munzel U, Maus J (2011) Efficacy and safety of antioxidant treatment with α-lipoic acid over 4 years in diabetic polyneuropathy: the NATHAN 1 trial. Diabetes Care 34:2054–2060. https://doi.org/10.2337/dc11-0503
Acknowledgments
We are thankful to Wilshire Laboratories (Pvt.) Ltd., Lahore, Pakistan, for the supply of pregabalin active material.
Code availability
Tiopronin (PubChem CID: 5483), Pregabalin (PubChem CID: 5486971).
Author information
Authors and Affiliations
Contributions
FS as research supervisor guided the research group in planning and conducting experiments throughout the study duration. MS, NA, and UF performed the experiments, interpreted the data, and carried out necessary statistical analysis. SAb, SAk, IU, NR, and ZUD provided inputs on the research work at different stages. MS prepared the initial draft of the manuscript. NUI critically revised the manuscript for important intellectual content. All authors read and approved the manuscript and all data were generated in-house and that no paper mill was used.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
The experimental procedures on animals were performed in accordance with the UK Animals (Scientific Procedures) Act 1986 and were approved by the Ethical Committee of the Department of Pharmacy, University of Peshawar (Reference No. 13/EC-15/Pharm). Moreover, these experimental protocols also conformed to the guidelines of Animal Research: Reporting In Vivo experiments (ARRIVE) and International Association for the Study of Pain (IASP).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 29.2 kb)
Rights and permissions
About this article
Cite this article
Shahid, M., Subhan, F., Islam, N.U. et al. The antioxidant N-(2-mercaptopropionyl)-glycine (tiopronin) attenuates expression of neuropathic allodynia and hyperalgesia. Naunyn-Schmiedeberg's Arch Pharmacol 394, 603–617 (2021). https://doi.org/10.1007/s00210-020-01995-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-020-01995-y